Last updated: February 19, 2026
This report analyzes the patent landscape and projected market trajectory for TrosPium Chloride, a muscarinic antagonist used to treat overactive bladder (OAB). The analysis focuses on existing patent exclusivity, generic competition, and the potential for market expansion based on clinical development and regulatory pathways.
WHAT IS TROSPIUM CHLORIDE AND ITS CURRENT MARKET POSITION?
TrosPium Chloride is a quaternary amine anticholinergic medication. It functions by blocking muscarinic receptors in the bladder detrusor muscle, reducing involuntary contractions and increasing bladder capacity. Approved indications include the treatment of detrusor overactivity, which manifests as urinary urgency, frequency, and urge incontinence.
The global market for OAB treatments is significant, driven by an aging population and increased prevalence of OAB symptoms. TrosPium Chloride competes with other antimuscarinic agents such as oxybutynin, tolterodine, solifenacin, and darifenacin, as well as beta-3 adrenergic agonists like mirabegron.
Key Market Indicators for TrosPium Chloride:
- Global Market Size: While specific current market size figures for TrosPium Chloride vary by report and forecast period, the OAB market overall is projected to grow. Estimates for the global OAB market range from USD 6 billion to USD 9 billion by 2027-2030, with antimuscarinics holding a substantial share [1, 2].
- Therapeutic Class Dominance: Antimuscarinics remain a cornerstone of OAB management due to their established efficacy and cost-effectiveness, particularly in the generic market.
- Competitive Landscape: The market is characterized by a mix of branded and generic products. TrosPium Chloride's market share is influenced by its availability in both immediate-release and extended-release formulations, as well as its side effect profile compared to competitors.
WHAT IS THE PATENT EXCLUSIVITY STATUS FOR TROSPIUM CHLORIDE?
The original patent protection for TrosPium Chloride has long expired, allowing for generic market entry. However, subsequent patent filings related to specific formulations, manufacturing processes, and new therapeutic uses can extend effective exclusivity periods.
Key Patent Milestones and Status:
- Original Composition of Matter Patent: The fundamental patent covering TrosPium Chloride expired significantly in the early 2000s.
- Formulation Patents: Several patents have been granted for extended-release (ER) formulations of TrosPium Chloride, which aim to improve patient compliance by reducing dosing frequency and potentially mitigating side effects like dry mouth. For instance, U.S. Patent No. 6,479,515, assigned to Astellas Pharma, covers a specific extended-release formulation [3]. The expiry dates of these formulation patents are crucial for determining the longevity of market exclusivity for specific product versions.
- Manufacturing Process Patents: Patents related to novel or improved manufacturing processes for TrosPium Chloride may exist. These can be challenged by generic manufacturers seeking to develop non-infringing processes.
- Method of Use Patents: Patents claiming new uses for TrosPium Chloride, such as in the treatment of conditions beyond OAB or in specific patient populations, could offer further protection if granted and valid.
Example of Formulation Patent Exclusivity:
Consider the Sanofi-Aventis (now Sanofi) product, Sanctura XR®. Patents covering this extended-release formulation have provided market protection. However, with the expiry of these key patents, generic versions of TrosPium Chloride ER have entered the market, significantly impacting pricing and market share dynamics for the originator product. The initial approval date for Sanctura XR® was in 2007.
HOW ARE GENERIC COMPETITORS IMPACTING THE TROSPIUM CHLORIDE MARKET?
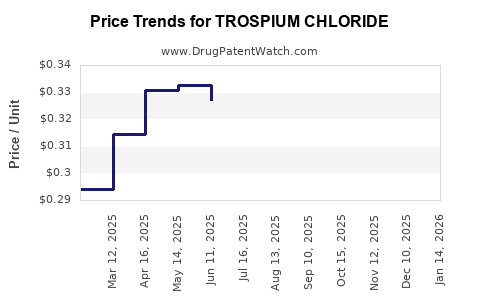
The entry of generic TrosPium Chloride products has led to a substantial decrease in pricing and an increase in market accessibility. Generic competition is a primary driver of market dynamics for off-patent drugs.
Impact of Generic Entry:
- Price Erosion: Upon patent expiry and the approval of generic versions, prices for TrosPium Chloride have declined significantly. This price erosion benefits payers and patients by making the treatment more affordable. For example, the average wholesale price (AWP) for generic TrosPium Chloride capsules can be less than 10% of the AWP for the branded equivalent prior to generic entry.
- Increased Market Share for Generics: Generic manufacturers quickly capture market share due to their lower price points. This shift is evident in prescription data, where generic TrosPium Chloride formulations often represent the majority of dispensed units.
- Competition Among Generic Manufacturers: The market for generic TrosPium Chloride is competitive, with multiple manufacturers vying for market share. This intense competition further drives down prices and can lead to supply chain dynamics influenced by production costs and capacity.
- Formulation Specialization: Generic companies often focus on developing bioequivalent generic versions of both immediate-release and extended-release formulations, further intensifying competition across different product types.
Key Generic Competitors and Their Market Presence:
While specific market share data for individual generic manufacturers is proprietary, major generic pharmaceutical companies that typically enter markets upon patent expiry of established drugs include:
- Teva Pharmaceuticals
- Mylan (now Viatris)
- Sandoz (a division of Novartis)
- Apotex
- Dr. Reddy's Laboratories
These companies often have robust generic portfolios and can leverage their manufacturing and distribution networks to gain significant market penetration.
WHAT ARE THE CURRENT AND FUTURE GROWTH DRIVERS FOR TROSPIUM CHLORIDE?
Despite being an off-patent drug, TrosPium Chloride can experience market growth through several avenues. These include its established efficacy, favorable safety profile relative to some older agents, continued physician prescribing habits, and potential for new indications or combination therapies.
Potential Growth Drivers:
- Established Efficacy and Tolerability: TrosPium Chloride offers a proven efficacy in managing OAB symptoms. Its quaternary amine structure results in limited penetration of the blood-brain barrier, which can lead to a lower incidence of central nervous system side effects (e.g., cognitive impairment, confusion) compared to some other antimuscarinics [4]. This favorable side effect profile can make it an attractive option for older patients or those sensitive to CNS side effects.
- Cost-Effectiveness: As a generic medication, TrosPium Chloride is a cost-effective treatment option for OAB, making it a preferred choice for healthcare systems and patients with cost constraints.
- Physician Familiarity and Prescribing Habits: Physicians are familiar with TrosPium Chloride's efficacy and tolerability, leading to continued prescription. Established prescribing patterns are slow to change, especially for medications with a good track record.
- Combination Therapy Potential: Research into combination therapies for OAB, potentially combining TrosPium Chloride with other agents that act via different mechanisms, could create new market opportunities.
- Expanded Indications: While currently approved for OAB, any future clinical trials demonstrating efficacy in other related conditions (e.g., irritable bowel syndrome with predominant diarrhea, hyperhidrosis) could lead to label expansions and market growth. However, significant clinical development for new indications of an off-patent drug is less common without strong pharmaceutical company investment.
- Market Penetration in Emerging Economies: As healthcare access expands in emerging economies, demand for established and affordable treatments like generic TrosPium Chloride is likely to increase.
Market Projections:
The market for TrosPium Chloride is expected to remain stable with modest growth, primarily driven by the continued prevalence of OAB and its cost-effectiveness as a generic option.
- CAGR (Compound Annual Growth Rate): Forecasts for the generic TrosPium Chloride market are generally in the low single digits, likely between 2% and 5% CAGR over the next five to seven years. This growth rate reflects the maturity of the drug but also the sustained demand for OAB treatments [1, 2].
- Geographic Expansion: Growth will be most pronounced in regions with increasing healthcare expenditure and a growing elderly population, such as Asia-Pacific and Latin America.
- Competition: The market will continue to be shaped by generic competition, with pricing remaining a key determinant of market share.
WHAT ARE THE REGULATORY CONSIDERATIONS AND CLINICAL DEVELOPMENT PATHWAYS?
Regulatory pathways for TrosPium Chloride are primarily focused on generic drug approvals and post-market surveillance. Significant new clinical development for novel indications would require substantial investment and follow standard regulatory approval processes.
Regulatory Landscape:
- Generic Drug Approval (ANDA): Generic versions of TrosPium Chloride are approved by regulatory agencies like the U.S. Food and Drug Administration (FDA) through the Abbreviated New Drug Application (ANDA) pathway. This requires demonstrating bioequivalence to the reference listed drug (RLD) [5].
- Post-Market Surveillance: Like all approved drugs, TrosPium Chloride is subject to pharmacovigilance, where adverse events are monitored and reported.
- Labeling Requirements: Generic drug labels must be equivalent to the RLD's approved labeling, including indications, contraindications, warnings, precautions, and adverse reactions.
- Manufacturing Standards: Generic manufacturers must adhere to Current Good Manufacturing Practices (cGMP) to ensure product quality and consistency.
Clinical Development Pathways (Hypothetical for New Indications):
Should any company pursue new clinical development for TrosPium Chloride, the pathway would involve:
- Pre-clinical Studies: In vitro and in vivo studies to assess the pharmacological activity and safety of TrosPium Chloride for a new proposed indication.
- Investigational New Drug (IND) Application: Submission to the FDA to request permission to conduct clinical trials in humans.
- Phase 1 Clinical Trials: Small studies in healthy volunteers to assess safety, tolerability, and pharmacokinetics.
- Phase 2 Clinical Trials: Studies in a larger group of patients with the target condition to evaluate efficacy and determine optimal dosage.
- Phase 3 Clinical Trials: Large-scale, randomized, controlled trials to confirm efficacy, monitor adverse reactions, and compare against existing treatments.
- New Drug Application (NDA) or Supplemental NDA (sNDA): Submission to the FDA to seek approval for the new indication.
Given that TrosPium Chloride is an established generic, significant investment in new clinical development for novel indications is unlikely without a strong commercial rationale, such as strong patent protection on a new formulation or a significant unmet medical need that existing treatments do not address.
WHAT ARE THE KEY FINANCIAL AND INVESTMENT CONSIDERATIONS?
For investors, TrosPium Chloride represents a mature, generic pharmaceutical asset. The financial implications are primarily related to volume sales, pricing pressures from generic competition, and operational efficiencies.
Financial Considerations for Investors:
- Revenue Streams: Revenue for TrosPium Chloride is generated through sales of generic formulations. This revenue is volume-driven rather than price-driven, especially following patent expiry.
- Profit Margins: Generic drug profit margins are typically lower than those of branded drugs but are maintained through high sales volumes and efficient manufacturing and distribution. Margins are subject to intense competition.
- Cost of Goods Sold (COGS): Efficient manufacturing processes, secure raw material sourcing, and economies of scale are critical for maintaining competitive COGS.
- Market Dynamics: The presence of multiple generic manufacturers leads to price negotiations with wholesalers, distributors, and pharmacy benefit managers (PBMs).
- Intellectual Property (IP) Landscape: While core patents have expired, ongoing monitoring of formulation or process patents is necessary to identify any potential for extended exclusivity or challenges to existing generic market entry.
- R&D Investment: For companies already manufacturing generic TrosPium Chloride, R&D investment is minimal, focused on process optimization or ensuring cGMP compliance. For companies considering entry, the primary investment is in obtaining regulatory approval and establishing manufacturing capacity.
- Risk Factors: Key risks include increased competition leading to further price erosion, regulatory changes affecting drug pricing or reimbursement, and potential supply chain disruptions.
Investment Strategy:
Investment in the TrosPium Chloride market is generally characterized by a focus on established generic manufacturers with strong production capabilities and established market access. The attractiveness lies in stable, albeit modest, revenue streams generated by a well-established therapeutic. Companies looking to acquire or develop generic TrosPium Chloride should focus on:
- Operational Efficiency: Streamlined manufacturing and supply chain management.
- Cost Leadership: Achieving the lowest cost of production to remain competitive on price.
- Market Access: Establishing strong relationships with key distributors and PBMs.
- Portfolio Diversification: TrosPium Chloride would typically be part of a broader generic portfolio, mitigating reliance on a single product.
KEY TAKEAWAYS
- TrosPium Chloride is a mature, off-patent medication for overactive bladder, with its primary market exclusivity long expired.
- Generic competition has significantly reduced pricing and increased market accessibility, making it a cost-effective OAB treatment.
- The OAB market is substantial and growing, with antimuscarinics like TrosPium Chloride remaining a significant segment.
- Future market growth is projected to be modest, driven by continued OAB prevalence, physician familiarity, and cost-effectiveness, with low single-digit CAGRs anticipated.
- Potential for new indications is limited due to the drug's generic status; current development focuses on generic formulation optimization and manufacturing efficiencies.
- Investment considerations center on operational efficiency, cost leadership in manufacturing, and securing broad market access within the competitive generic pharmaceutical landscape.
FREQUENTLY ASKED QUESTIONS
What is the average wholesale price difference between branded and generic TrosPium Chloride?
The average wholesale price (AWP) difference can be substantial, with generic TrosPium Chloride formulations typically costing 70% to 90% less than the originator product's price prior to generic entry. This significant reduction is a direct consequence of generic competition entering the market after patent expiry.
Are there any novel formulations of TrosPium Chloride currently under development for new indications?
As of current public disclosures, there is no significant, widely publicized pipeline of novel TrosPium Chloride formulations pursuing entirely new indications. Development efforts for off-patent drugs typically focus on lifecycle management, such as improved generic formulations or manufacturing process enhancements rather than novel therapeutic areas, unless a substantial unmet need and patentability exist.
How does the blood-brain barrier penetration of TrosPium Chloride compare to other antimuscarinics used for OAB?
TrosPium Chloride, being a quaternary amine, has limited lipid solubility and consequently exhibits minimal penetration across the blood-brain barrier. This contrasts with some other antimuscarinics, such as oxybutynin and tolterodine, which are tertiary amines and can cross the blood-brain barrier more readily, potentially leading to a higher incidence of central nervous system side effects like cognitive impairment or drowsiness.
What is the typical market share held by generic TrosPium Chloride compared to branded versions?
Following the expiry of key patents and the introduction of generic alternatives, generic TrosPium Chloride formulations collectively command a dominant market share, often exceeding 90% of dispensed prescriptions. The branded originator product's share diminishes rapidly post-generic entry.
Which regulatory agencies approve generic TrosPium Chloride in major markets?
Generic TrosPium Chloride is approved by major regulatory agencies globally. In the United States, this is the Food and Drug Administration (FDA) via the Abbreviated New Drug Application (ANDA) process. In Europe, it is the European Medicines Agency (EMA) and national competent authorities within member states. Other significant markets include Health Canada, Australia's Therapeutic Goods Administration (TGA), and Japan's Pharmaceuticals and Medical Devices Agency (PMDA).
Citations
[1] Grand View Research. (2023). Overactive Bladder Treatment Market Size, Share & Trends Analysis Report By Drug Class (Antimuscarinics, Beta-3 Adrenergic Agonists), By Region, And Segment Forecasts, 2023 - 2030.
[2] Mordor Intelligence. (2023). Overactive Bladder Treatment Market - Growth, Trends, COVID-19 Impact, and Forecasts (2024 - 2029).
[3] Astellas Pharma US, Inc. (n.d.). U.S. Patent No. 6,479,515. United States Patent and Trademark Office.
[4] Drake, M. J. (2011). TrosPium Chloride in the treatment of overactive bladder. Therapeutic Advances in Urology, 3(1), 25–34.
[5] U.S. Food and Drug Administration. (2020). Generics: Abbreviated New Drug Applications (ANDAs). Retrieved from https://www.fda.gov/drugs/abbreviated-new-drug-applications-and/generics-abbreviated-new-drug-applications-andas