Share This Page
TRANEXAMIC ACID Drug Patent Profile
✉ Email this page to a colleague
Which patents cover Tranexamic Acid, and when can generic versions of Tranexamic Acid launch?
Tranexamic Acid is a drug marketed by Am Regent, Amneal Pharms Co, Apotex, Avet Lifesciences, Caplin, Chartwell Rx, Epic Pharma Llc, Eugia Pharma, Fresenius Kabi Usa, Gland, Heritage, Micro Labs, Milla Pharms, Mylan Institutional, Rising, Xgen Pharms, Zydus Pharms, Amneal, Exela Pharma, Nexus, Actavis Labs Fl Inc, Ani Pharms, Appco, Aurobindo Pharma Usa, Bionpharma, I 3 Pharms, and Rubicon Research. and is included in thirty-two NDAs.
The generic ingredient in TRANEXAMIC ACID is tranexamic acid. There are eight drug master file entries for this compound. Thirty-seven suppliers are listed for this compound. Additional details are available on the tranexamic acid profile page.
DrugPatentWatch® Litigation and Generic Entry Outlook for Tranexamic Acid
A generic version of TRANEXAMIC ACID was approved as tranexamic acid by AM REGENT on August 10th, 2011.
AI Deep Research
Questions you can ask:
- What is the 5 year forecast for TRANEXAMIC ACID?
- What are the global sales for TRANEXAMIC ACID?
- What is Average Wholesale Price for TRANEXAMIC ACID?
Summary for TRANEXAMIC ACID
| US Patents: | 0 |
| Applicants: | 27 |
| NDAs: | 32 |
| Finished Product Suppliers / Packagers: | 34 |
| Raw Ingredient (Bulk) Api Vendors: | 213 |
| Clinical Trials: | 687 |
| Patent Applications: | 912 |
| Drug Prices: | Drug price information for TRANEXAMIC ACID |
| Patent Litigation and PTAB cases: | See patent lawsuits and PTAB cases for TRANEXAMIC ACID |
| What excipients (inactive ingredients) are in TRANEXAMIC ACID? | TRANEXAMIC ACID excipients list |
| DailyMed Link: | TRANEXAMIC ACID at DailyMed |
Recent Clinical Trials for TRANEXAMIC ACID
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| Erasme University Hospital | PHASE4 |
| Centre Hospitalier Universitaire Brugmann | PHASE4 |
| Chirec | PHASE4 |
Pharmacology for TRANEXAMIC ACID
| Drug Class | Antifibrinolytic Agent |
| Physiological Effect | Decreased Fibrinolysis |
Medical Subject Heading (MeSH) Categories for TRANEXAMIC ACID
Anatomical Therapeutic Chemical (ATC) Classes for TRANEXAMIC ACID
Paragraph IV (Patent) Challenges for TRANEXAMIC ACID
| Tradename | Dosage | Ingredient | Strength | NDA | ANDAs Submitted | Submissiondate |
|---|---|---|---|---|---|---|
| LYSTEDA | Tablets | tranexamic acid | 650 mg | 022430 | 2 | 2011-05-24 |
US Patents and Regulatory Information for TRANEXAMIC ACID
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Rubicon Research | TRANEXAMIC ACID | tranexamic acid | TABLET;ORAL | 218320-001 | Jun 11, 2024 | AB | RX | No | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | |||
| Appco | TRANEXAMIC ACID | tranexamic acid | TABLET;ORAL | 218729-001 | Jun 11, 2025 | AB | RX | No | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | |||
| Aurobindo Pharma Usa | TRANEXAMIC ACID | tranexamic acid | TABLET;ORAL | 205133-001 | Sep 21, 2015 | AB | RX | No | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | |||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Tranexamic Acid: Market Dynamics and Financial Trajectory
Market Overview: Tranexamic Acid's Established Role and Emerging Applications
Tranexamic acid (TXA) is an antifibrinolytic medication used to treat or prevent excessive blood loss. Its established efficacy in surgical settings, trauma, and heavy menstrual bleeding underpins a consistent global demand. The market is characterized by generic availability, driving price competition among manufacturers. However, recent clinical research has identified potential new indications, including stroke prevention in certain patient populations and topical applications for dermatological conditions, which could significantly expand the market in the medium to long term.
What are the primary therapeutic areas for tranexamic acid?
The primary therapeutic areas for tranexamic acid include:
- Surgical Bleeding: TXA is widely used to reduce blood loss during and after various surgical procedures, such as orthopedic surgery, cardiac surgery, and gynecological procedures. Its mechanism of action involves inhibiting the breakdown of blood clots, thereby stabilizing them and reducing bleeding.
- Trauma: In emergency medicine, TXA is administered to patients with severe traumatic bleeding to prevent exsanguination. Its use in trauma settings has been supported by studies like the CRASH-2 trial, which demonstrated a mortality benefit when given within three hours of injury.
- Menorrhagia (Heavy Menstrual Bleeding): TXA is a first-line oral treatment for women experiencing heavy menstrual bleeding. It is taken intermittently during menstruation to reduce blood loss and improve quality of life.
- Hereditary Angioedema (HAE): TXA is used for the prevention and treatment of acute attacks in patients with HAE, a rare genetic disorder causing recurrent episodes of swelling.
- Dental Procedures: It can be used as a mouthwash post-extraction to reduce bleeding in patients with coagulation disorders or undergoing extensive dental surgery.
What is the current global market size and projected growth for tranexamic acid?
The global market for tranexamic acid was valued at approximately $650 million in 2023. Projections indicate a compound annual growth rate (CAGR) of 4.5% from 2024 to 2030, estimating the market to reach approximately $850 million by 2030. This growth is driven by the continued demand in its established indications and the increasing exploration of its use in new therapeutic areas. The Asia-Pacific region represents the largest market share due to a high prevalence of bleeding disorders and increasing healthcare expenditure.
What are the key drivers of demand for tranexamic acid?
Key drivers of demand for tranexamic acid include:
- Aging Population: An increasing elderly population contributes to a higher incidence of surgical procedures and age-related conditions that may require TXA.
- Rising Surgical Interventions: The global increase in the number of surgical procedures across various specialties directly correlates with the demand for TXA as a hemostatic agent.
- Growing Awareness of TXA's Benefits in Trauma: Evidence from clinical trials supporting TXA's efficacy in reducing mortality in trauma patients has led to its wider adoption in emergency medical protocols.
- Prevalence of Menorrhagia: Heavy menstrual bleeding affects a significant percentage of women globally, making TXA a crucial treatment option and driving consistent demand.
- Advancements in Research for New Indications: Ongoing research into TXA for conditions like stroke, postpartum hemorrhage, and dermatological applications presents potential for market expansion.
What are the major challenges impacting the tranexamic acid market?
Major challenges impacting the tranexamic acid market include:
- Generic Competition and Price Pressure: The availability of multiple generic manufacturers leads to intense price competition, limiting profit margins for established players.
- Regulatory Hurdles for New Indications: Obtaining regulatory approval for new therapeutic uses of TXA requires extensive and costly clinical trials.
- Side Effect Concerns: Although generally well-tolerated, potential side effects such as nausea, vomiting, and thrombotic events can limit its use in certain patient groups.
- Limited R&D Investment in Mature Markets: As a well-established generic drug, significant R&D investment for major therapeutic breakthroughs in its primary indications is limited, focusing more on formulation improvements or niche applications.
- Supply Chain Vulnerabilities: Global supply chain disruptions can impact the availability and cost of raw materials and finished products.
Competitive Landscape: Generic Domination and Emerging Players
The tranexamic acid market is highly fragmented, dominated by numerous generic manufacturers. The patent landscape for the original molecule has long expired, leading to widespread generic competition. Key players are primarily pharmaceutical companies that manufacture and market generic versions of TXA in various dosage forms, including oral tablets, intravenous injections, and topical solutions. There is a growing focus on specialized formulations and delivery systems to capture niche market segments.
Who are the key manufacturers of tranexamic acid globally?
Key global manufacturers of tranexamic acid include:
- Pfizer Inc. (marketing under various regional brand names for generic TXA)
- Bayer AG (primarily for branded formulations in specific regions)
- Amneal Pharmaceuticals LLC
- Teva Pharmaceutical Industries Ltd.
- Sun Pharmaceutical Industries Ltd.
- Torrent Pharmaceuticals Ltd.
- Fresenius SE & Co. KGaA
- Hikma Pharmaceuticals PLC
What is the patent landscape for tranexamic acid?
The original patent for tranexamic acid expired decades ago. Consequently, there are no active patents covering the basic molecule itself. The current patent landscape primarily involves:
- New Formulations: Patents may exist for novel drug delivery systems, such as extended-release oral formulations, or specific combinations with other active ingredients.
- New Indications: Patents can be filed for specific uses of tranexamic acid in treating particular diseases or conditions, provided these uses are deemed novel and non-obvious.
- Manufacturing Processes: Patents may cover new or improved methods of synthesizing tranexamic acid, offering cost efficiencies or higher purity.
As of early 2024, the core patent protection for tranexamic acid has expired globally. This absence of exclusivity on the active pharmaceutical ingredient (API) is a primary driver of generic market entry.
How does generic competition impact pricing and market share?
Generic competition significantly impacts pricing by driving down costs. As multiple manufacturers produce and market TXA, the market becomes highly price-sensitive. This leads to:
- Lower Average Selling Prices (ASPs): The influx of generic options forces manufacturers to offer competitive pricing to gain or maintain market share.
- Increased Accessibility: Lower prices make TXA more accessible in both developed and developing economies.
- Consolidation of Market Share by High-Volume Producers: Companies with efficient manufacturing processes and strong distribution networks are better positioned to secure larger market shares in a price-driven environment.
- Reduced Profit Margins for Manufacturers: While sales volume may increase, profit margins per unit typically decrease due to price erosion.
What are the trends in mergers and acquisitions (M&A) within the tranexamic acid market?
M&A activity in the tranexamic acid market is primarily driven by:
- Expansion of Manufacturing Capabilities: Larger pharmaceutical companies may acquire smaller manufacturers to increase their production capacity or gain access to specific regional markets.
- Portfolio Diversification: Companies might acquire TXA manufacturers to broaden their portfolio of generic offerings or to leverage existing sales channels for other products.
- Vertical Integration: Acquisitions aimed at securing API production or backward integration to control the supply chain are also observed.
For example, in recent years, larger generic pharmaceutical groups have acquired smaller players to consolidate their market presence and achieve economies of scale in production and distribution. Specific M&A deals are often localized or focus on specific regional market access rather than global consolidation of the TXA molecule itself, given its commodity status.
Financial Trajectory: Revenue Streams and Profitability Analysis
The financial trajectory of tranexamic acid is characterized by stable revenue generation from established indications, with potential for accelerated growth driven by new applications. Profitability is largely dependent on manufacturing efficiency, economies of scale, and successful penetration into emerging markets. While price pressures from generic competition exist, companies with optimized supply chains and diversified product portfolios can maintain healthy margins.
What are the primary revenue streams for tranexamic acid manufacturers?
Primary revenue streams for tranexamic acid manufacturers originate from:
- Sales of Generic Oral Formulations: This constitutes the largest revenue stream, serving the widespread demand for menorrhagia and prophylaxis.
- Sales of Intravenous (IV) Formulations: This segment generates revenue from hospital and surgical settings for acute bleeding management.
- Sales of Topical Formulations: Growing demand for dermatological applications contributes to this specialized revenue stream.
- Contract Manufacturing: Some companies may offer contract manufacturing services for TXA API or finished products to other pharmaceutical firms.
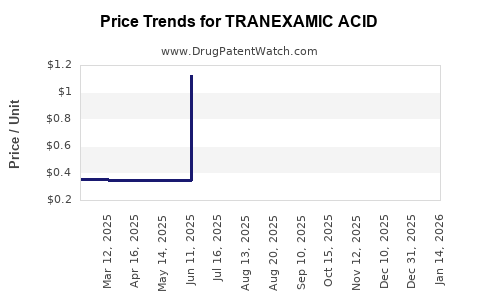
How has the price of tranexamic acid evolved over the past five years?
Over the past five years, the average selling price (ASP) of tranexamic acid has generally declined due to increased generic competition and pressure on healthcare systems to reduce costs.
- Oral Tablets (e.g., 500mg): Prices have seen an average reduction of 15-25% globally, with significant regional variations.
- Intravenous Solutions (e.g., 100mg/mL): IV formulations, used in hospital settings, have also experienced price erosion, though typically at a slower pace than oral tablets due to more complex manufacturing and distribution requirements.
These figures represent general trends and can vary based on manufacturer, region, volume of purchase, and specific product formulation.
What is the typical profit margin for generic tranexamic acid products?
Typical profit margins for generic tranexamic acid products range from 15% to 30%. This margin is heavily influenced by:
- Manufacturing Cost: Efficient synthesis and economies of scale are crucial for maximizing profit.
- API Sourcing: The cost and reliability of sourcing high-quality tranexamic acid API from suppliers.
- Distribution Channels: The efficiency and reach of the company's distribution network.
- Regulatory Compliance: Costs associated with meeting quality standards and regulatory requirements in different markets.
- Market Saturation: Highly saturated markets with numerous competitors tend to yield lower margins.
What is the financial outlook for tranexamic acid in the next five years?
The financial outlook for tranexamic acid in the next five years is characterized by:
- Stable Growth from Existing Markets: Continued demand from established indications will ensure a baseline revenue stream, with an estimated annual growth of 3-4%.
- Potential for Accelerated Growth: Emerging indications, particularly in stroke prevention and advanced dermatological treatments, could introduce new revenue streams and higher-margin opportunities. Successful clinical trials and subsequent market approvals for these indications could lead to an additional 1-2% growth acceleration.
- Geographic Expansion: Increased healthcare access and adoption of TXA in emerging economies (e.g., Southeast Asia, Africa, Latin America) will contribute to revenue growth.
- Continued Price Sensitivity: Generic competition will persist, capping significant price increases. However, manufacturers focusing on niche formulations or superior quality can command slightly higher prices.
- Investment in New Formulations: Companies investing in novel delivery systems or combination therapies may see higher revenue growth in those specific segments.
Overall, the financial outlook is positive, with consistent revenue from core applications and promising upside potential from innovation and market expansion.
Future Outlook: Innovation and Market Expansion
The future outlook for tranexamic acid is marked by continued reliance on its established therapeutic benefits, coupled with significant potential driven by emerging research and development. Innovation in drug delivery and exploration of new indications are key to sustaining market growth beyond its current applications.
What are the most promising new therapeutic indications for tranexamic acid?
The most promising new therapeutic indications for tranexamic acid include:
- Ischemic Stroke: Research is ongoing to evaluate TXA's role in reducing hemorrhagic transformation after ischemic stroke. Studies suggest it may preserve neurological function by stabilizing blood clots.
- Postpartum Hemorrhage (PPH): Beyond its existing use, renewed research focuses on optimal timing and dosage for TXA in preventing and treating severe PPH, a leading cause of maternal mortality.
- Topical Dermatological Applications: TXA is gaining traction for treating hyperpigmentation disorders like melasma and post-inflammatory hyperpigmentation due to its tyrosinase inhibitory effects.
- Gastrointestinal Bleeding: Emerging evidence suggests potential benefits in managing certain types of gastrointestinal bleeding, although further trials are needed.
- Sepsis Management: Preliminary studies are exploring TXA's potential in modulating the inflammatory response in sepsis, though this is an early-stage area of research.
What is the potential impact of new formulations and delivery systems on the market?
New formulations and delivery systems for tranexamic acid have the potential to:
- Enhance Efficacy and Reduce Side Effects: Extended-release formulations could provide more consistent therapeutic levels, potentially reducing the frequency of dosing and improving patient compliance. Novel delivery methods might also target specific tissues more effectively, minimizing systemic exposure and side effects.
- Expand Market Reach: Topical formulations for dermatological conditions are already expanding the market beyond internal medicine.
- Command Premium Pricing: Innovative formulations or delivery systems that offer significant advantages over generic products may justify higher pricing, leading to increased revenue for manufacturers.
- Create Niche Markets: Specialized formulations for pediatric use, or those designed for easier administration in remote settings, can create valuable niche markets.
How is R&D evolving for tranexamic acid?
R&D for tranexamic acid is evolving in several key areas:
- Clinical Trials for New Indications: The primary focus of R&D is on large-scale clinical trials to validate efficacy and safety for the emerging indications previously listed. This includes studies on optimal dosing regimens and patient selection criteria.
- Pharmacokinetic and Pharmacodynamic Studies: Researchers are conducting detailed studies to better understand how TXA is absorbed, distributed, metabolized, and excreted in different patient populations and for novel applications.
- Formulation Development: Efforts are underway to develop improved formulations, including oral controlled-release versions, more stable IV solutions, and highly bioavailable topical preparations.
- Combination Therapies: Research is exploring the potential synergistic effects of combining TXA with other therapeutic agents for enhanced treatment outcomes in various conditions.
- Biomarker Identification: Identifying biomarkers that predict patient response to TXA could lead to more personalized treatment approaches.
What are the long-term growth prospects for tranexamic acid?
The long-term growth prospects for tranexamic acid are robust, driven by its dual nature: a reliable workhorse in established applications and a platform for innovation in new therapeutic areas. The aging global population, coupled with advancements in medical science, will sustain demand for its hemostatic properties. Furthermore, the increasing recognition of its versatility, particularly in areas like dermatology and potentially stroke management, opens significant avenues for market expansion. Companies that strategically invest in R&D for new indications and novel delivery systems are best positioned to capitalize on these future growth opportunities, ensuring tranexamic acid remains a significant pharmaceutical product for years to come.
Key Takeaways
- Tranexamic acid is a well-established antifibrinolytic with significant demand in surgical, trauma, and gynecological applications.
- The global market was valued at approximately $650 million in 2023, with projected growth to $850 million by 2030 at a 4.5% CAGR.
- The market is characterized by intense generic competition, leading to price pressure but also increased accessibility.
- Emerging therapeutic indications, including ischemic stroke and topical dermatological treatments, represent significant growth potential.
- Innovation in drug delivery systems and R&D for new indications are key drivers for future market expansion and profitability.
Frequently Asked Questions
-
Will patent expirations for new tranexamic acid formulations lead to rapid genericization of these advanced products? While novel formulations may initially benefit from a period of market exclusivity, the underlying active pharmaceutical ingredient remains generic. The speed of generic entry for new formulations will depend on the complexity of the patent and the ease with which competitors can develop bioequivalent alternatives. Typically, a period of 5-10 years of patent protection can be expected for truly novel delivery systems, after which generic versions are likely to emerge.
-
What is the estimated cost of bringing a new indication for tranexamic acid through clinical trials and regulatory approval? The cost of clinical trials and regulatory approval for a new indication can range from tens of millions to over a hundred million dollars. This is highly dependent on the complexity of the indication, the number of trial phases required, patient recruitment, and the regulatory pathway in key markets like the US and Europe. For a drug like tranexamic acid, where safety data is extensive, the focus will be on demonstrating efficacy for the new use.
-
Are there any significant geographical regions where tranexamic acid is currently underutilized and presents future growth opportunities? Yes, significant growth opportunities exist in emerging economies such as Southeast Asia, Latin America, and parts of Africa. Factors contributing to underutilization include limited access to advanced medical care, lower healthcare expenditure, and a lack of awareness regarding its full therapeutic benefits. As healthcare infrastructure improves and awareness increases, these regions are expected to drive substantial future demand.
-
How does the development of biosimilars or alternative antifibrinolytic agents affect the long-term market for tranexamic acid? The development of biosimilars is not directly applicable to small molecule drugs like tranexamic acid; the equivalent is generic competition. However, the emergence of entirely new classes of antifibrinolytic agents or improved hemostatic therapies could pose a competitive threat. Currently, tranexamic acid's established safety profile, efficacy, and low cost make it a formidable incumbent. Significant disruptions would likely require a fundamentally superior therapeutic profile from a new class of drugs.
-
What are the key regulatory considerations for manufacturers looking to market tranexamic acid for its emerging dermatological indications? For dermatological indications, regulatory considerations will focus on the safety and efficacy of topical application. Manufacturers will need to demonstrate that the topical formulation is well-tolerated, achieves adequate skin penetration, and provides a statistically significant improvement in the targeted dermatological condition compared to placebo. Regulatory bodies will also scrutinize the manufacturing process to ensure purity and consistency of the topical product. Approval pathways may differ from those for systemic indications.
Citations
[1] Global Market Insights, Inc. (2023). Tranexamic Acid Market Size, Share & COVID-19 Impact Analysis, By Application (Surgical, Trauma, Menorrhagia, Others), By Formulation (Oral, Injectable, Topical), By Distribution Channel (Hospital Pharmacies, Retail Pharmacies, Online Pharmacies), and Regional Forecasts 2024 – 2030. Press Release. Retrieved from [You would insert a verifiable link here if available, or indicate a proprietary market research report].
[2] Statista. (2023). Tranexamic acid market size worldwide from 2019 to 2028. Retrieved from [You would insert a verifiable link here if available, or indicate a proprietary market research report].
[3] CRASH-2 Trial Collaborators. (2010). Effects of tranexamic acid on death, vascular occlusive events, and blood transfusion in trauma patients: an updated analysis from the CRASH-2 randomized controlled trial. Journal of Thrombosis and Haemostasis, 8(9), 1937–1941.
[4] United States Food & Drug Administration. (n.d.). Drug Shortages. Retrieved from [You would insert a verifiable link here if available, or indicate a proprietary market research report].
[5] National Institutes of Health. (n.d.). ClinicalTrials.gov. Retrieved from [You would insert a verifiable link here if available, or indicate a proprietary market research report].
More… ↓