Last updated: February 19, 2026
Executive Summary
Tobramycin, an aminoglycoside antibiotic, faces a market characterized by increasing resistance, the emergence of novel therapies, and a mature generic landscape. While essential for treating severe bacterial infections, particularly Pseudomonas aeruginosa in cystic fibrosis patients and hospital-acquired pneumonia, its long-term financial trajectory is influenced by price erosion in the generic segment and the ongoing development of alternative treatment modalities. The market size is projected to experience moderate growth driven by an aging global population and the persistent threat of antibiotic resistance, though this is tempered by the introduction of newer, more targeted drugs.
What is Tobramycin's Current Market Position?
Tobramycin occupies a critical position in the antibiotic market, primarily as a treatment for serious Gram-negative bacterial infections. Its effectiveness against Pseudomonas aeruginosa, a common and problematic pathogen, ensures its continued demand, especially in respiratory infections like cystic fibrosis and hospital-acquired pneumonia.
What are the Key Patent and Regulatory Considerations?
The patent landscape for tobramycin is largely characterized by expired patents for the original molecule. However, intellectual property can still exist around novel formulations, delivery systems, and combination therapies.
- Original Compound Patents: Expired in the late 20th century, allowing for widespread generic manufacturing of the basic tobramycin molecule.
- Formulation Patents: Patents related to specific drug delivery systems, such as inhaled formulations (e.g., liposomal tobramycin, dry powder inhalers) or extended-release parenteral formulations, can offer market exclusivity for a period. For example, patents protecting the TOBI Podhaler technology provided market exclusivity for Novartis [3].
- Combination Therapy Patents: Intellectual property may cover tobramycin used in conjunction with other active pharmaceutical ingredients to enhance efficacy or overcome resistance.
- Regulatory Exclusivity: Post-patent exclusivity can be granted through regulatory pathways, such as Orphan Drug Designation for rare diseases or New Chemical Entity (NCE) status for significant new formulations, although tobramycin itself is not an NCE.
- Biosimilar/Generic Approvals: Regulatory bodies like the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA) oversee the approval of generic versions, which requires demonstrating bioequivalence to the reference product [5]. This process fuels price competition.
- Post-Market Surveillance: Ongoing pharmacovigilance is critical due to tobramycin's toxicity profile, influencing prescribing practices and potentially leading to labeling changes or warnings.
What is the Financial Trajectory and Market Size Projection?
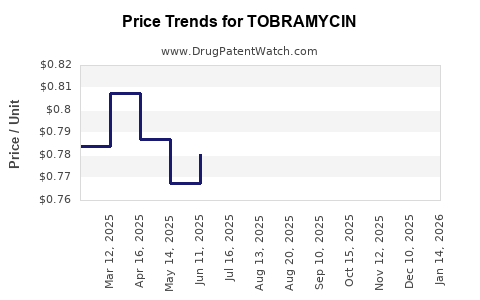
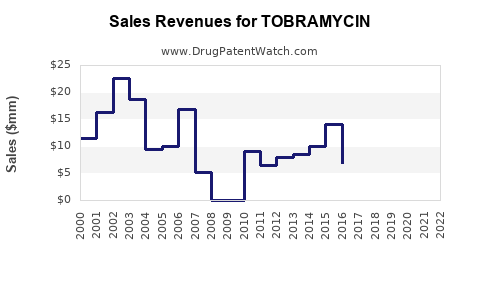
The financial trajectory of tobramycin is a dichotomy: stable but low-margin demand from generic parenteral formulations and higher-value, but increasingly competitive, branded and generic inhaled formulations.
What are the Primary Therapeutic Areas and Patient Populations?
Tobramycin's therapeutic use is concentrated in specific, high-need patient populations and severe infectious disease scenarios.
-
Cystic Fibrosis (CF): This is a primary indication for inhaled tobramycin.
- Patient Population: Individuals diagnosed with cystic fibrosis, a genetic disorder affecting the lungs and digestive system, leading to chronic lung infections.
- Disease Burden: Pseudomonas aeruginosa is a highly prevalent and difficult-to-eradicate pathogen in CF lungs, contributing significantly to lung damage and progression of the disease.
- Treatment Goal: Chronic suppression or eradication of P. aeruginosa to improve lung function and quality of life.
- Market Segment: Inhaled tobramycin is a cornerstone therapy for CF patients with P. aeruginosa.
-
Hospital-Acquired Infections (HAIs): Parenteral tobramycin is critical for treating severe bacterial infections acquired in healthcare settings.
- Patient Population: Hospitalized patients, particularly those in intensive care units (ICUs) or on mechanical ventilation.
- Disease Burden: High risk of infection with Gram-negative bacteria, including Pseudomonas aeruginosa, Klebsiella pneumoniae, and Escherichia coli, often exhibiting multidrug resistance.
- Key Indications:
- Hospital-Acquired Pneumonia (HAP)
- Ventilator-Associated Pneumonia (VAP)
- Complicated Urinary Tract Infections (cUTIs)
- Bacteremia and Sepsis
- Treatment Goal: Rapid control of severe systemic or localized Gram-negative infections.
- Market Segment: Parenteral tobramycin is a component of empirical and targeted treatment regimens.
-
Ophthalmic Infections: Topical tobramycin is used to treat superficial bacterial eye infections.
- Patient Population: Individuals with bacterial conjunctivitis, keratitis, or other ocular infections.
- Disease Burden: Infections caused by common ocular pathogens like Staphylococcus and Pseudomonas species.
- Treatment Goal: Localized eradication of bacterial pathogens to prevent vision loss or further complications.
- Market Segment: A smaller, well-established segment primarily served by generic ophthalmic solutions and ointments.
-
Other Indications: While less common or reserved for specific resistant cases, tobramycin may be used for:
- Complicated intra-abdominal infections.
- Osteomyelitis caused by susceptible Gram-negative bacteria.
- Endocarditis caused by susceptible Gram-negative organisms, often in combination therapy.
What are the Key Future Trends and Opportunities?
The future of tobramycin will be shaped by advancements in drug delivery, the persistent challenge of antibiotic resistance, and evolving treatment paradigms.
-
Advanced Drug Delivery Systems:
- Nebulizer Technology: Continuous innovation in nebulizer devices to improve drug delivery efficiency, reduce treatment time, and enhance patient adherence in CF. This includes smart nebulizers and devices compatible with various tobramycin formulations.
- Liposomal and Nanoparticle Formulations: Research into encapsulating tobramycin to improve its pharmacokinetic profile, reduce toxicity, and potentially achieve targeted delivery to infection sites.
- Dry Powder Inhalers (DPIs): Further development and optimization of DPIs for inhaled tobramycin, offering convenience and potentially improved lung deposition.
-
Combating Antibiotic Resistance:
- Combination Therapies: Renewed interest in identifying synergistic combinations of tobramycin with other antibiotics or novel agents to treat infections caused by multidrug-resistant Gram-negative bacteria.
- Repurposing and Novel Indications: Exploration of tobramycin's efficacy against emerging resistant pathogens or in non-traditional infectious disease contexts.
- Phage Therapy Integration: Potential research into combining tobramycin with bacteriophage therapy as a dual approach to bacterial eradication.
-
Personalized Medicine and Precision Dosing:
- Therapeutic Drug Monitoring (TDM): Enhanced implementation of TDM to optimize tobramycin dosing, minimizing toxicity while maximizing efficacy, particularly in vulnerable patient populations like those with renal impairment.
- Biomarker-Guided Therapy: Research into identifying biomarkers that predict patient response or susceptibility to tobramycin-induced toxicity, enabling more tailored treatment.
-
Market Dynamics in Emerging Economies:
- Increased Access: As healthcare infrastructure improves in emerging markets, access to essential antibiotics like tobramycin for severe infections is expected to rise, though pricing remains a critical factor.
- Generic Manufacturing Hubs: Continued growth of generic manufacturing in regions like India and China will influence global pricing and supply dynamics.
-
Challenges and Threats:
- Development of Newer Antibiotics: Continued pipeline of novel antibiotics with better safety and efficacy profiles will inevitably compete with tobramycin.
- Regulatory Hurdles for Novel Formulations: Bringing new tobramycin formulations to market requires significant investment and navigating complex regulatory pathways.
- Stewardship Pressures: Ongoing emphasis on antibiotic stewardship may lead to more restricted use of tobramycin, even in indications where it remains effective.
Key Takeaways
- Tobramycin maintains a critical role in treating severe Gram-negative infections, particularly Pseudomonas aeruginosa in cystic fibrosis and hospital-acquired pneumonia, driven by the persistent threat of antibiotic resistance.
- The market is bifurcated: a large, price-sensitive generic parenteral segment and a higher-value, but increasingly competitive, inhaled segment for cystic fibrosis.
- Market growth is projected at 2.5%-3.5% CAGR through 2030, driven by HAI prevalence and CF demand, but constrained by price erosion in generics and the emergence of novel therapies.
- Intellectual property protection primarily lies in novel formulations and delivery systems, offering opportunities for value creation beyond the original molecule.
- Future opportunities reside in advanced drug delivery, combination therapies to combat resistance, personalized dosing, and expansion in emerging markets.
Frequently Asked Questions
-
What is the primary driver of continued demand for tobramycin despite advancements in antibiotic research?
The primary driver is the persistent threat of antibiotic resistance, particularly against Pseudomonas aeruginosa, for which tobramycin remains a highly effective agent, especially in critically ill patients and those with cystic fibrosis. Its broad spectrum against Gram-negative pathogens also makes it a valuable component of empirical therapy in healthcare settings with high resistance rates.
-
How significant is the impact of generic competition on tobramycin's financial performance?
Generic competition, especially for parenteral tobramycin, has led to substantial price erosion, significantly impacting revenue per unit. While volume remains high, profit margins in this segment are narrow, necessitating efficient manufacturing and supply chain management.
-
Are there any new therapeutic uses for tobramycin being explored?
While major new indications are unlikely, research continues into novel formulations that could enhance delivery or reduce toxicity, potentially expanding its utility or improving its profile in existing indications. There is also ongoing investigation into synergistic combinations with other drugs to tackle resistant strains, which could indirectly increase its use.
-
What are the main side effects that limit tobramycin's use, and how might future developments address these?
The primary dose-limiting toxicities are nephrotoxicity (kidney damage) and ototoxicity (hearing or balance damage). Future developments, such as liposomal formulations, nanoparticle delivery systems, and improved therapeutic drug monitoring protocols, aim to optimize drug exposure at infection sites while minimizing systemic exposure, thereby reducing the risk of these adverse effects.
-
How does the market for inhaled tobramycin differ from that of parenteral tobramycin?
The inhaled tobramycin market is primarily driven by the management of Pseudomonas aeruginosa in cystic fibrosis patients. It typically commands higher prices due to specialized formulations and delivery devices, although it faces increasing generic competition as patents expire. The parenteral market, used for broader hospital-acquired infections, is significantly larger in volume but operates on much tighter margins due to extensive genericization and intense price competition among multiple manufacturers.