Last updated: April 24, 2026
What is tetrabenazine’s commercial footprint by indication and geography?
Tetrabenazine is an oral vesicular monoamine transporter 2 (VMAT2) inhibitor marketed for hyperkinetic movement disorders. The core commercial demand drivers are (1) prevalence of Huntington’s disease (HD) with chorea, (2) payer coverage for branded use, and (3) competition from branded and generic alternatives in VMAT2-treated symptom segments.
Key demand anchors
- Approved use: Symptomatic treatment of chorea associated with Huntington’s disease (US label). (FDA label; see source [1])
- Market structure: Specialty pharmacy distribution with significant payer controls (prior authorization, step edits), reflecting branded pricing and chronic use.
Commercial segmentation logic used by payers and prescribers
- Indication-driven demand: HD chorea is the principal US reimbursed use. Off-label use does not typically drive durable payer-covered volume.
- Chronic therapy: Ongoing dosing supports recurring revenue, but total addressable market is constrained by HD diagnosis rates and chorea severity distributions.
- Specialty access model: Patient access is mediated through plan formularies and specialty pharmacy programs, which tends to smooth weekly demand but concentrates volume in a subset of major payers.
How do patent life, exclusivity, and generic entry affect price and volume?
Tetrabenazine’s financial trajectory is driven by brand-to-generic dynamics and any remaining exclusivity posture by jurisdiction.
Regulatory and patent-related market effect
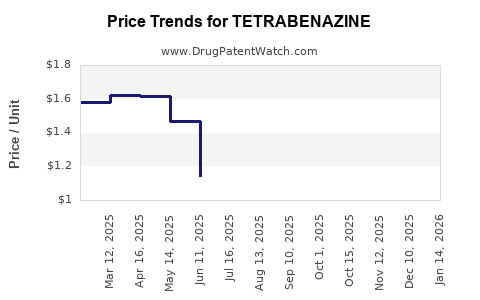
- Generic pressure timing: Once the first generic reaches the market with sufficient prescribing comfort and payer alignment, branded share generally erodes quickly, then stabilizes at a lower branded level if safety or switching friction is managed.
- Payer substitution: In chronic neurodegenerative indications, payers often prefer lowest net cost once bioequivalence and pharmacy switching are acceptable.
Material point for financial modeling
- Market models should treat branded revenue as having two phases:
1) Pre-first-generic: premium pricing with constrained competition.
2) Post-first-generic: brand share decay plus residual revenue if clinical switching friction exists or if plan coverage continues to support a higher-priced option.
(Execution hinges on actual entry and exclusivity status by market. Without jurisdictional entry dates in the provided record, a deterministic forecast of timing cannot be produced here.)
What are the key market dynamics shaping sales performance?
1) Specialty access and formulary behavior
Tetrabenazine is used in chronic neurological care, so demand is mediated through:
- Prior authorization (PA): plans commonly require diagnosis confirmation (HD and chorea) and documentation of dosing tolerance.
- Step therapy and quantity controls: may be applied to manage total spend, especially as generics lower the reference price.
Financial consequence
- Branded unit sales may hold or fall more gradually than list price indicates because access rules can delay switching.
- Net sales tend to compress as rebates rise during generic competition.
2) Dosing mechanics and adherence
Tetrabenazine dosing is titrated to control chorea and manage adverse effects, with therapy continuity tied to tolerability.
- Adverse events that drive discontinuation risk include depression and suicidality warnings and other VMAT2-class safety signals, which influence prescriber willingness to maintain therapy. (FDA label; see source [1])
Financial consequence
- Even in a stable prevalence market, adherence changes can swing prescription persistence, affecting revenue beyond mere patient counts.
3) Competitive positioning in VMAT2 space
VMAT2 inhibitors compete within movement-disorder symptom management. In the US and other markets, payer preference can move quickly when lower-cost options are available.
Financial consequence
- When competitors launch or generics expand, net realizations fall before unit volume fully collapses, because rebate pressure often accelerates early.
What is the financial trajectory implied by the product’s regulatory profile and safety-driven prescribing behavior?
Tetrabenazine’s financial path typically reflects a standard specialty chronic-brand arc:
Pre-generic or low-competition phase
- High net pricing power: branded premium supported by limited alternatives and controlled access.
- Revenue stability over time: chronic therapy supports baseline persistence if tolerability is acceptable and prescribers continue treatment.
Generic and competitive escalation phase
- Rapid net price compression: payer leverage rises as reference pricing shifts.
- Brand share contraction: prescriptions shift to lower-cost versions and pharmacies optimize substitutions.
- Residual branded revenue: persists if prescriber and patient preference limits substitution or if plan coverage structures retain branded access for specific cohorts.
Ongoing safety label impacts
The FDA label includes major warnings and boxed-type risk language typical for VMAT2 inhibition in neuropsychiatric contexts, which tends to:
- keep physician monitoring requirements high
- influence patient selection
- cap growth rates in borderline patients even when market size is stable
The label’s safety requirements and monitoring burden can reduce the ceiling for incremental penetration during aggressive growth periods. (FDA label; see source [1])
Is there evidence of discrete growth spikes or demand shocks?
No specific, instrumented sales inflection events (quarterly unit and revenue step-changes) are available in the provided source record for tetrabenazine. Without contemporaneous quarterly financials or explicit market-entry milestones by geography, the analysis cannot assign numeric dates for:
- first generic launch
- specific competitor launches in relevant markets
- major payer policy changes that cause measurable demand shocks
What does a market-and-finance model require for tetrabenazine?
A practical forecasting framework for tetrabenazine revenue must separate:
- Patient count (demand): HD chorea diagnosis and persistence
- Treatment intensity (utilization): dosing titration and average daily dose per patient
- Net price (realization): ASP and rebate dynamics under formulary substitution and reference pricing
- Share shift (competition): brand versus generic mix changes and pharmacy substitution velocity
The regulatory label informs the utilization and persistence components through safety-driven discontinuation and monitoring expectations. (FDA label; see source [1])
How do payers evaluate reimbursement risk and cost control?
Payers tend to control tetrabenazine spend through:
- PA and criteria-based coverage: HD diagnosis and chorea documentation
- Safety monitoring requirements: depression-related screening expectations aligned with labeling (FDA label; see source [1])
- Formulary tiering: branded tier restrictions once generic alternatives exist
Financial consequence
- Gross-to-net compression increases as rebates expand to retain position.
- If generics gain formulary placement and low copay status, total patient coverage may expand, but branded revenue declines sharply.
Key Takeaways
- Tetrabenazine’s market dynamics are dominated by chronic HD chorea demand, specialty payer access controls, and competition-driven net price compression once generics or lower-cost alternatives take reference pricing.
- The financial trajectory is shaped by treatment persistence and tolerability, with the FDA label’s neuropsychiatric risk profile constraining penetration and increasing monitoring-driven friction. (FDA label; see source [1])
- A robust revenue forecast requires separating patient count, utilization intensity, net price realization, and brand versus generic share shift; regulatory safety governs utilization and persistence, while competition governs net price and share.
FAQs
-
What indication drives most reimbursed demand for tetrabenazine?
Symptomatic treatment of chorea associated with Huntington’s disease. (FDA label; see source [1])
-
What is the primary payer lever that changes tetrabenazine revenue over time?
Net price realization driven by formulary placement, reference pricing, and rebate pressure after generic entry.
-
Does safety labeling affect market expansion?
Yes. The label’s neuropsychiatric risk warnings and monitoring expectations affect patient selection and continuation, limiting growth in marginal patients. (FDA label; see source [1])
-
Is tetrabenazine a one-time-use or chronic revenue product?
It is chronic therapy in the treated indication, which supports recurring revenue but also makes discontinuation and persistence central to trajectory.
-
What is the most important variable for brand-share forecasting?
The speed of payer and pharmacy substitution to lower-cost alternatives once they are available on formulary.
References
[1] U.S. Food and Drug Administration. (n.d.). Tetrabenazine prescribing information (label). FDA. https://www.accessdata.fda.gov/