Last updated: February 19, 2026
Tamoxifen citrate, a selective estrogen receptor modulator (SERM), holds a significant position in the pharmaceutical market primarily for its role in breast cancer treatment and prevention. Its market trajectory is shaped by patent expirations, the emergence of generics, and ongoing clinical research for new indications.
What is the current market size and projected growth for Tamoxifen Citrate?
The global tamoxifen citrate market is substantial, driven by its established efficacy and broad patient base. Market size estimates vary, but generally fall within the range of $200 million to $300 million annually, with projections indicating moderate but steady growth of 3-5% per annum over the next five to seven years. This growth is underpinned by the increasing incidence of breast cancer globally, particularly in aging populations, and the drug's inclusion in standard treatment protocols.
Key Market Drivers:
- Breast Cancer Incidence: Rising global breast cancer rates are the primary demand driver. The World Health Organization (WHO) reported over 2.3 million new cases of breast cancer in 2020 [1].
- Generic Availability: Patent expiries have led to widespread generic availability, making tamoxifen citrate an accessible and cost-effective treatment option. This affordability supports high prescription volumes.
- Clinical Utility: Tamoxifen citrate remains a cornerstone therapy for hormone receptor-positive (HR+) breast cancer, accounting for approximately 70-80% of all breast cancer cases. Its role in both adjuvant and metastatic settings, as well as for risk reduction in high-risk individuals, ensures consistent demand.
- Emerging Indications: Ongoing research into tamoxifen citrate's efficacy for other conditions, such as uterine fibroids and infertility, could potentially expand its market share.
Market Restraints:
- Competition from Newer Therapies: Newer generations of SERMs, aromatase inhibitors (AIs), and targeted therapies offer alternative or adjunctive treatment options, albeit often at higher price points.
- Side Effect Profile: While generally well-tolerated, tamoxifen citrate's known side effects, including hot flashes, increased risk of endometrial cancer, and thromboembolic events, can lead to patient non-adherence or selection for alternative treatments.
- Generic Price Erosion: Intense competition within the generic market can lead to significant price erosion, limiting revenue growth for manufacturers.
What is the patent landscape for Tamoxifen Citrate?
The primary patents for tamoxifen citrate have long expired. The original patent for tamoxifen was filed by Imperial Chemical Industries (ICI), which later became AstraZeneca. These foundational patents granted market exclusivity for decades.
- Original Patents: Expired. The initial composition of matter and method of use patents have been out of force for many years.
- Process Patents: Various patents related to specific manufacturing processes or formulations of tamoxifen citrate may still exist or have recently expired. However, these typically do not grant broad market exclusivity akin to composition of matter patents.
- Exclusivity Periods: Given the drug's age, the primary market exclusivity has long since concluded. The drug is widely available as a generic.
Impact of Patent Expiration:
The expiration of key patents has led to a fully genericized market. This has resulted in:
- Increased Competition: Numerous pharmaceutical companies manufacture and market generic versions of tamoxifen citrate.
- Price Reduction: Significant downward pressure on pricing due to generic competition.
- Market Access: Widespread availability and affordability across different healthcare systems and geographies.
Who are the key players in the Tamoxifen Citrate market?
The market for tamoxifen citrate is characterized by a fragmented landscape of generic manufacturers. Leading players are those with robust generic drug portfolios and established distribution networks.
Major Generic Manufacturers:
- Teva Pharmaceutical Industries: A significant global supplier of generic pharmaceuticals.
- Mylan N.V. (now part of Viatris): Another major player in the generic drug market.
- Sun Pharmaceutical Industries: A large Indian multinational pharmaceutical company with a broad generic offering.
- Dr. Reddy's Laboratories: A prominent Indian pharmaceutical company engaged in generic drug manufacturing.
- Cipla Limited: An Indian multinational pharmaceutical company with a strong presence in emerging markets.
Brand Name Originator:
- AstraZeneca: While the original innovator, AstraZeneca's primary market share in tamoxifen citrate is now in the context of its historical role and potential for branded generics or differentiated formulations if any were to emerge. However, the bulk of the market is dominated by generic versions.
What are the global sales and revenue trends for Tamoxifen Citrate?
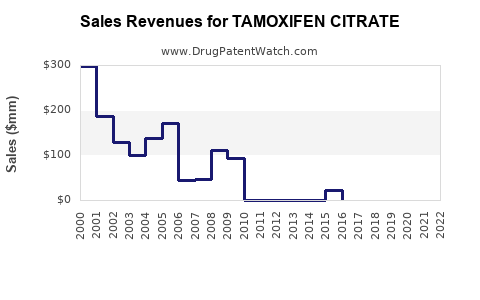
Global sales revenue for tamoxifen citrate has stabilized and is characterized by modest growth, largely driven by volume rather than significant price increases. The total revenue is influenced by pricing strategies of various generic manufacturers and regional market dynamics.
Estimated Annual Global Revenue:
- 2023: Approximately $250 million.
- Projected 2024: $255 million.
- Projected 2025: $260 million.
Regional Sales Distribution (Approximate):
- North America: 35%
- Europe: 30%
- Asia-Pacific: 20%
- Rest of the World: 15%
The revenue figures reflect sales from both branded (where applicable, though limited) and generic versions. The vast majority of sales volume is attributed to generic products.
What is the clinical utility and regulatory status of Tamoxifen Citrate?
Tamoxifen citrate is approved by major regulatory agencies worldwide for specific indications, predominantly in breast cancer management. Its established efficacy and safety profile, documented over decades of use, solidify its regulatory standing.
Approved Indications:
- Treatment of early-stage breast cancer: Adjuvant therapy in postmenopausal and premenopausal women.
- Treatment of metastatic breast cancer: In postmenopausal and premenopausal women.
- Ductal Carcinoma In Situ (DCIS): Reduction of the risk of developing invasive breast cancer in women with DCIS.
- Risk Reduction of Breast Cancer: In women at high risk for developing breast cancer.
Regulatory Agencies:
- U.S. Food and Drug Administration (FDA): Approved for the above indications.
- European Medicines Agency (EMA): Approved for similar indications.
- Other National Regulatory Bodies: Approval status aligns with major international agencies.
Notable Side Effects Requiring Monitoring:
- Endometrial Cancer: Increased risk, particularly in postmenopausal women.
- Thromboembolic Events: Increased risk of deep vein thrombosis (DVT) and pulmonary embolism (PE).
- Hot Flashes and Vaginal Discharge: Common menopausal symptoms.
- Cataracts and Visual Disturbances: Less common but reported.
The regulatory status remains strong for its established indications. Any expansion into new therapeutic areas would require new clinical trials and subsequent regulatory approvals.
What are the future market outlook and potential growth opportunities for Tamoxifen Citrate?
The future market for tamoxifen citrate is expected to remain stable, with growth driven by an expanding patient population requiring breast cancer treatment and prevention. Opportunities for growth, though limited, may arise from specific market segments and potential new applications.
Key Future Market Factors:
- Aging Global Population: Increased prevalence of breast cancer with age.
- Improved Diagnostics: Earlier detection leading to higher treatment volumes.
- Cost-Effectiveness in Developing Economies: Tamoxifen citrate's affordability makes it a critical treatment option in resource-limited settings.
- Potential for Combination Therapies: Further research into using tamoxifen citrate alongside newer agents could create niche markets.
Potential Growth Opportunities:
- Expanded Indications Research:
- Gynecological Conditions: Investigations into its use for uterine fibroids, endometriosis, and infertility continue. While these are not primary drivers currently, positive trial outcomes could open new market segments.
- Other Cancers: Limited research has explored its role in other estrogen-sensitive cancers, though with less robust evidence.
- Geographic Expansion: Increased market penetration in developing regions where access to newer, more expensive therapies is limited.
- Formulation Innovation: While unlikely to disrupt the generic market significantly, any development of novel delivery systems or improved formulations with reduced side effect profiles could capture a small premium market share, though this is highly speculative given the drug's age and cost constraints.
The market for tamoxifen citrate is mature. Sustained revenue will largely depend on maintaining high prescription volumes for its established breast cancer indications and potentially realizing incremental gains from niche applications or geographical penetration.
Key Takeaways
- The global tamoxifen citrate market is valued between $200-$300 million annually, with projected growth of 3-5% due to increasing breast cancer incidence.
- Original patents for tamoxifen citrate have expired, resulting in a fully genericized market dominated by multiple manufacturers.
- Key market drivers include rising breast cancer rates and the drug's cost-effectiveness, while competition from newer therapies and side effects act as restraints.
- The market is fragmented, with Teva Pharmaceutical Industries, Mylan (Viatris), Sun Pharmaceutical Industries, Dr. Reddy's Laboratories, and Cipla Limited being significant generic players.
- Future growth is expected to be modest, driven by demographic shifts and potential, albeit speculative, expansion into new indications.
Frequently Asked Questions
-
What is the primary therapeutic use of tamoxifen citrate?
Tamoxifen citrate is primarily used for the treatment and prevention of breast cancer, particularly hormone receptor-positive (HR+) breast cancer.
-
Has tamoxifen citrate lost patent protection?
Yes, the foundational patents for tamoxifen citrate have long expired, allowing for the widespread production and sale of generic versions by numerous pharmaceutical companies.
-
What are the major side effects associated with tamoxifen citrate?
Significant side effects include an increased risk of endometrial cancer, thromboembolic events (like blood clots), hot flashes, and vaginal discharge.
-
Are there any new indications being explored for tamoxifen citrate?
Ongoing research is investigating its potential use in treating gynecological conditions such as uterine fibroids, though these are not yet established indications.
-
How does the market for tamoxifen citrate compare to newer breast cancer therapies?
Tamoxifen citrate competes with newer therapies such as aromatase inhibitors and targeted drugs. While these may offer different efficacy profiles or side effect management, tamoxifen citrate remains a cornerstone due to its established efficacy, affordability, and broad applicability, especially in generic form.