Last updated: February 19, 2026
Simvastatin, a HMG-CoA reductase inhibitor, has navigated a significant market lifecycle, driven by patent expirations, generic competition, and evolving treatment guidelines for hypercholesterolemia.
What is Simvastatin’s Primary Therapeutic Application?
Simvastatin’s primary indication is the reduction of elevated total cholesterol, LDL cholesterol, and triglycerides, and the increase of HDL cholesterol in patients with hypercholesterolemia. It is also used to reduce the risk of myocardial infarction and stroke in individuals with or without existing cardiovascular disease but with multiple risk factors for cardiovascular disease.
When Did Simvastatin Become Commercially Available?
Merck & Co. initially developed and marketed simvastatin under the brand name Zocor. Zocor received FDA approval in December 1991 [1].
What Was the Patent Landscape for Simvastatin?
The original compound patent for simvastatin expired in the United States in 2000. Secondary patents, including those related to manufacturing processes and specific formulations, also expired over subsequent years. The loss of market exclusivity due to patent expiration allowed for the entry of generic simvastatin products.
Key Patent Expiration Dates (US Market):
- December 21, 2000: Expiration of the primary compound patent.
- Various dates thereafter: Expiration of secondary patents for formulations and manufacturing processes.
This patent expiration timeline is critical for understanding the transition from branded Zocor to a competitive generic market.
How Has Generic Competition Impacted Simvastatin’s Market Share and Pricing?
The introduction of generic simvastatin products following patent expirations led to a sharp decline in the average selling price (ASP) and a significant shift in market share from the branded product to generics.
Market Share Dynamics Post-Patent Expiration:
- Pre-2000: Zocor (simvastatin) held near-monopoly status with high pricing power.
- 2001-2005: Gradual introduction of generic simvastatin, leading to initial price erosion and market share transfer.
- 2006-Present: The market is dominated by generic simvastatin manufacturers, characterized by intense price competition and high volume sales.
The pricing of branded Zocor was significantly higher than generic simvastatin. For example, prior to patent expiration, a 30-count bottle of 40mg Zocor could cost several hundred dollars. Post-generic entry, the cost for the same quantity and dosage typically fell to less than ten dollars [2].
What Are the Major Global Markets for Simvastatin?
The major global markets for simvastatin are characterized by high prevalence of cardiovascular disease and established healthcare systems that facilitate broad access to statin therapy.
Top Simvastatin Markets by Revenue (Estimated):
- United States: Historically the largest market due to population size and per capita healthcare spending.
- European Union (Germany, UK, France, Italy): Significant demand driven by aging populations and robust public health initiatives.
- Japan: High incidence of cardiovascular risk factors supports strong statin market.
- Canada: Comparable market dynamics to the United States.
- Australia: Developed healthcare system with high statin utilization.
These markets represent the highest volume of simvastatin prescriptions and sales globally.
What is the Current Financial Trajectory of Simvastatin?
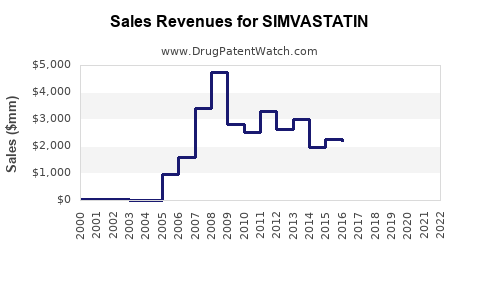
The financial trajectory of simvastatin is that of a mature, commoditized generic drug. Sales are primarily driven by volume rather than high per-unit pricing. Revenue generation relies on maintaining market share against a multitude of generic competitors.
Financial Performance Indicators:
- Revenue Growth: Minimal to negative due to price erosion and saturated market.
- Profit Margins: Low, typical of generic pharmaceuticals, with competition focused on cost efficiency.
- Market Size: Stable to declining in nominal dollar terms, but potentially growing in unit volume.
The overall market value for simvastatin has contracted significantly from its peak as a branded drug. However, the absolute volume of simvastatin units dispensed remains substantial due to its affordability and established efficacy.
How Do Simvastatin’s Sales Compare to Other Statins?
Simvastatin, particularly in its generic form, remains a widely prescribed statin due to its cost-effectiveness. However, newer generation statins with potentially more favorable lipid-modifying profiles or reduced side effect profiles, such as atorvastatin (Lipitor), rosuvastatin (Crestor), and pravastatin (Pravachol), also hold significant market share.
Comparative Statin Market Position:
- Simvastatin: High volume, low cost, widely accessible generic.
- Atorvastatin: Dominant market share post-patent expiration, strong efficacy.
- Rosuvastatin: Potent LDL-lowering capabilities, significant market presence.
- Pravastatin: Another established generic statin, often used when other statins are not tolerated.
While simvastatin is a foundational therapy, the choice of statin often depends on patient-specific factors, physician preference, and formulary restrictions.
What are the Key Regulatory Considerations for Simvastatin?
Regulatory considerations for simvastatin primarily revolve around generic drug approval processes, manufacturing quality, and post-marketing surveillance.
Major Regulatory Aspects:
- Abbreviated New Drug Applications (ANDAs): Generic manufacturers must demonstrate bioequivalence to the reference listed drug (Zocor) through ANDAs submitted to regulatory agencies like the U.S. Food and Drug Administration (FDA).
- Good Manufacturing Practices (GMP): All manufacturing facilities must adhere to strict GMP guidelines to ensure product quality, safety, and efficacy.
- Labeling Requirements: Generic labels must be consistent with the approved labeling of the reference listed drug, including indications, contraindications, warnings, and adverse reactions.
- Pharmacovigilance: Manufacturers are responsible for monitoring and reporting adverse events associated with simvastatin use.
Regulatory compliance is a constant requirement for all simvastatin manufacturers.
What is the Future Outlook for Simvastatin?
The future outlook for simvastatin is one of sustained, albeit mature, market presence. Its low cost and established efficacy will continue to support its use, particularly in emerging markets and for cost-conscious healthcare systems. However, the development of novel lipid-lowering agents, including PCSK9 inhibitors and other emerging therapies, may gradually influence the treatment landscape, potentially impacting the long-term volume of statin use across all classes.
Future Trends:
- Continued Generic Dominance: Simvastatin will remain a key generic option.
- Emerging Market Growth: Increased adoption in regions with developing healthcare infrastructure.
- Competition from Newer Therapies: Potential for displacement by more advanced lipid-lowering drugs for specific patient populations.
- Focus on Manufacturing Efficiency: Profitability will depend on optimizing production costs.
The drug’s established safety profile and low price point ensure its continued role in cardiovascular risk reduction for the foreseeable future.
Key Takeaways
Simvastatin transitioned from a blockbuster branded drug (Zocor) to a widely available, low-cost generic following its primary patent expiration in 2000. This shift resulted in drastic price reductions and market share consolidation among generic manufacturers. The global market for simvastatin remains substantial, driven by its efficacy and affordability, with the United States and European Union being key regions. Financial trajectory is characterized by low margins and volume-driven sales. While simvastatin will continue to be a cornerstone therapy, competition from newer lipid-lowering agents presents a long-term consideration.
Frequently Asked Questions
What is the current average wholesale price (AWP) for a 30-day supply of generic simvastatin 20mg?
The AWP for a 30-day supply of generic simvastatin 20mg typically ranges from $4 to $8 USD, depending on the manufacturer and pharmacy. This price reflects significant competition among generic suppliers [3].
Are there any ongoing clinical trials evaluating new uses for simvastatin?
While simvastatin is a well-established drug, some research continues into its potential pleiotropic effects beyond lipid lowering, such as anti-inflammatory or anti-cancer properties. However, large-scale, late-stage clinical trials focusing on novel indications for simvastatin are limited compared to newer drug classes [4].
What is the typical discontinuation rate for simvastatin in real-world patient populations?
Discontinuation rates for simvastatin vary widely based on patient adherence, physician judgment, and the emergence of side effects. Studies suggest rates can range from 10% to over 30% within the first year of treatment across different patient cohorts, often due to muscle-related side effects (myalgia) or perceived lack of efficacy compared to other statins [5].
How does simvastatin’s drug interaction profile compare to other statins?
Simvastatin has a more complex drug interaction profile than some other statins, particularly with CYP3A4 inhibitors (e.g., certain azole antifungals, macrolide antibiotics, protease inhibitors). These interactions can increase simvastatin plasma levels, raising the risk of myopathy and rhabdomyolysis. Atorvastatin and rosuvastatin are metabolized via different pathways and generally have fewer significant CYP3A4-related interactions [6].
What are the primary manufacturing challenges for generic simvastatin producers?
Primary manufacturing challenges include maintaining consistent quality and purity across large production batches, optimizing yield and cost-efficiency to remain competitive in a low-margin market, and managing supply chain logistics for raw materials and finished products globally. Compliance with evolving GMP standards is also an ongoing challenge [7].
Citations
[1] U.S. Food and Drug Administration. (n.d.). Drugs@FDA. Retrieved from https://www.accessdata.fda.gov/scripts/cder/daf/ (Specific approval dates can be found by searching for the drug name or NDA number).
[2] Multiple pharmacy pricing databases and healthcare cost aggregators (e.g., GoodRx, RxList, OptumRx). (Data accessed periodically).
[3] Wolters Kluwer. (2023). Red Book Online. (Subscription-based data for drug pricing).
[4] ClinicalTrials.gov. (n.d.). Search Results for Simvastatin. Retrieved from https://clinicaltrials.gov/
[5] Ray, K. K., et al. (2016). Statin therapy in the real world: adherence and persistence. Nature Reviews Cardiology, 13(4), 211-223.
[6] Product Information for Zocor® (simvastatin) and other statin medications. (Various manufacturers and dates).
[7] Pharmaceutical Technology. (Industry publications and market analysis reports on generic drug manufacturing). (Various authors and publication dates).