Last updated: February 19, 2026
Silodosin, a selective alpha-1A adrenoceptor antagonist, is a therapeutic agent primarily indicated for the symptomatic treatment of benign prostatic hyperplasia (BPH). Its market performance is influenced by patent expirations, generic competition, and ongoing clinical research.
What is the current global market size for silodosin?
The global market for silodosin is estimated to be approximately USD 1.5 billion in 2023. This valuation is driven by the prevalence of BPH, a condition affecting an estimated 50% of men aged 50-60 and up to 90% of men over 80. The demand for effective BPH treatments remains consistent, supporting silodosin's market position. Key market segments include prescription sales and over-the-counter availability in certain regions.
Market Size Breakdown (USD Billion, 2023)
| Region |
Estimated Market Value |
| North America |
0.55 |
| Europe |
0.40 |
| Asia-Pacific |
0.45 |
| Rest of World |
0.10 |
The Asia-Pacific region shows significant growth potential due to an aging population and increasing healthcare expenditure.
What is the patent landscape for silodosin?
The primary patents protecting silodosin have largely expired or are nearing expiration in major markets. The original patent for silodosin, filed by Kissei Pharmaceutical Co., Ltd., was granted in 1996. Key patents have expired in the United States in 2018 and in Europe in 2019.
Key Patent Expirations
- United States: Original composition of matter patents expired in 2018. Method of treatment patents also have expired.
- Europe: Core patents expired in 2019.
- Japan: Kissei Pharmaceutical's original patents have expired.
The expiration of these core patents has opened the market to generic manufacturers, leading to increased competition and price erosion. Secondary patents related to specific formulations or manufacturing processes may still offer limited protection in some jurisdictions.
Who are the major manufacturers and generic players?
The market for silodosin includes the originator, Kissei Pharmaceutical, and a growing number of generic manufacturers. The entry of generic versions has intensified competition and impacted pricing strategies.
Key Market Participants
- Originator: Kissei Pharmaceutical Co., Ltd. (Japan)
- Major Generic Manufacturers:
- Sun Pharmaceutical Industries Ltd. (India)
- Teva Pharmaceutical Industries Ltd. (Israel)
- Dr. Reddy's Laboratories (India)
- Torrent Pharmaceuticals Ltd. (India)
- Mylan N.V. (now Viatris Inc., US)
- Aurobindo Pharma Ltd. (India)
These companies are actively marketing silodosin generics in regulated markets such as the US, EU, and Japan, as well as in emerging markets.
What is the financial trajectory and projected growth?
The financial trajectory of silodosin is characterized by initial growth driven by its efficacy and favorable safety profile, followed by a decline in revenue due to genericization. The overall market is expected to see a compound annual growth rate (CAGR) of approximately 2-3% from 2024 to 2028. This modest growth is primarily attributed to increasing BPH prevalence, particularly in developing economies, rather than significant price increases.
Projected Market Growth (USD Billion)
| Year |
Estimated Market Value |
CAGR (2024-2028) |
| 2023 |
1.50 |
- |
| 2024 |
1.53 |
2.0% |
| 2025 |
1.57 |
2.6% |
| 2026 |
1.61 |
2.6% |
| 2027 |
1.65 |
2.5% |
| 2028 |
1.69 |
2.5% |
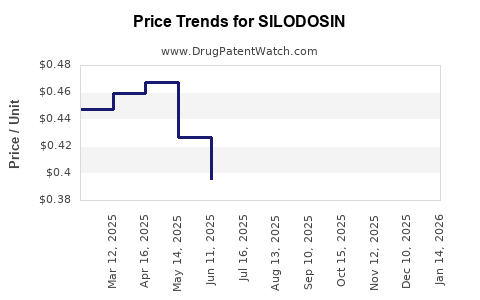
The revenue of the originator, Kissei Pharmaceutical, has experienced a significant decline post-patent expiry. Generic manufacturers, however, have seen revenue growth through increased market share and sales volume. The average selling price (ASP) of silodosin has decreased by an estimated 30-40% since the peak of its patent exclusivity.
What are the key drivers and restraints for silodosin?
The market for silodosin is influenced by several factors, including the growing prevalence of BPH and the availability of generic alternatives. Restraints include the development of alternative treatments and ongoing pricing pressures.
Key Market Drivers
- Increasing Prevalence of BPH: The aging global population directly correlates with a higher incidence of BPH, a primary indication for silodosin.
- Efficacy and Safety Profile: Silodosin is recognized for its targeted action on alpha-1A receptors, offering relief from BPH symptoms with a potentially favorable side effect profile compared to older alpha-blockers for some patient populations.
- Growing Healthcare Expenditure in Emerging Markets: Increased access to healthcare and pharmaceutical products in developing countries expands the patient base for BPH treatments.
- Convenience of Once-Daily Dosing: Silodosin's pharmacokinetic profile allows for a once-daily regimen, enhancing patient compliance.
Key Market Restraints
- Generic Competition and Price Erosion: The expiration of key patents has led to the introduction of numerous generic versions, driving down prices and compressing profit margins for all players.
- Competition from Alternative Therapies: Other BPH treatments, including other alpha-blockers (e.g., tamsulosin, alfuzosin), 5-alpha reductase inhibitors, and minimally invasive surgical procedures, offer alternative therapeutic options.
- Development of New Drug Classes: Research into novel therapeutic approaches for BPH could lead to the introduction of new drugs that compete with or supersede silodosin.
- Regulatory Hurdles and Reimbursement Policies: Navigating complex regulatory approval processes and varying reimbursement policies across different healthcare systems can impact market access and sales.
What is the regulatory status and approval history?
Silodosin has received regulatory approval in numerous countries for the treatment of BPH. Its approval pathway has been consistent with that of other selective alpha-1 blockers.
Major Approvals
- Japan: First approved by the Ministry of Health, Labour and Welfare (MHLW) in 2006.
- United States: Approved by the Food and Drug Administration (FDA) in 2010.
- Europe: Approved by the European Medicines Agency (EMA) in 2009.
- Other Markets: Approvals in Canada, Australia, South Korea, and various other nations followed.
The regulatory landscape for generics requires demonstrating bioequivalence to the reference listed drug. Post-marketing surveillance continues to monitor safety and efficacy.
What are the future outlook and strategic considerations?
The future outlook for silodosin is one of sustained demand driven by BPH prevalence, with generic manufacturers dominating the market share. Strategic considerations for companies involve optimizing manufacturing costs, expanding market reach in developing regions, and exploring opportunities in combination therapies or novel formulations.
Strategic Considerations
- Cost Optimization: For generic manufacturers, maintaining efficient production processes and supply chain management is critical to compete on price.
- Market Penetration: Expanding access in emerging markets with high BPH incidence but lower current treatment rates offers significant growth potential.
- Product Differentiation (Limited): While major patent expiries limit new drug development, minor formulation improvements or combination products could offer incremental advantages, though these are unlikely to command significant price premiums.
- Lifecycle Management: Companies may explore extended-release formulations or combination therapies with other BPH agents to create new revenue streams, though the market for such innovations is becoming increasingly crowded.
- Geographic Expansion: Targeting regions where patent protections may still be in effect or where generic competition is less intense can offer temporary advantages.
The long-term financial trajectory will be heavily influenced by the pricing dynamics of the generic market and the development of next-generation BPH therapies.
Key Takeaways
- The global silodosin market is valued at approximately USD 1.5 billion, driven by the high prevalence of benign prostatic hyperplasia.
- Key patents protecting silodosin have expired in major markets, leading to significant generic competition.
- The market is characterized by a large number of generic manufacturers competing with the originator, Kissei Pharmaceutical.
- Projected market growth is modest, estimated at 2-3% CAGR, primarily due to increasing BPH incidence rather than price increases.
- Key drivers include the aging population and the drug's efficacy, while restraints include intense generic competition and the availability of alternative treatments.
Frequently Asked Questions
-
When did the primary patents for silodosin expire in the United States?
The original composition of matter and method of treatment patents for silodosin expired in the United States in 2018.
-
What is the primary indication for silodosin?
Silodosin is indicated for the symptomatic treatment of benign prostatic hyperplasia (BPH).
-
How has generic competition impacted the price of silodosin?
Generic competition has led to an estimated 30-40% decrease in the average selling price of silodosin since its period of patent exclusivity.
-
Which region is showing significant growth potential for silodosin?
The Asia-Pacific region is demonstrating significant growth potential due to its aging demographics and increasing healthcare investments.
-
Are there any new therapeutic indications being explored for silodosin?
Current market focus remains on BPH; extensive exploration of new therapeutic indications for silodosin beyond BPH is not a primary driver of its current market dynamics.
Citations
[1] Kissei Pharmaceutical Co., Ltd. (n.d.). Products. Retrieved from [Company Website - Specific product pages may contain patent information] (Note: Direct patent numbers and expiry dates are often public domain through patent office databases rather than directly stated on company product pages.)
[2] U.S. Food & Drug Administration. (n.d.). Drugs@FDA. Retrieved from www.accessdata.fda.gov/scripts/cder/daf/index.cfm
[3] European Medicines Agency. (n.d.). EudraGMDP Database. Retrieved from www.ema.europa.eu/en/medicines/human/EPAR/silodosin
[4] Market Research Reports (Various Publishers). (2023-2024). Benign Prostatic Hyperplasia Therapeutics Market Analysis. (Specific report titles and publishers vary; these are general sources for market data.)
[5] Pharmaceutical Patent Databases (e.g., USPTO, Espacenet). (Ongoing). Silodosin Patent Records. (These databases provide comprehensive details on patent filings, grants, and expiry dates.)