Last updated: February 20, 2026
What is RYDAPT and What Conditions Does It Treat?
RYDAPT (midostaurin) is an oral kinase inhibitor approved by the FDA in 2017. It targets multiple molecular pathways, chiefly FLT3 mutations and mast cell disease. The drug is indicated for:
- Newly diagnosed FLT3-mutated acute myeloid leukemia (AML), combined with chemotherapy
- Advanced systemic mastocytosis (SM), including aggressive SM (ASM), systemic mastocytosis with associated hematologic neoplasm (SM-AHN), and indolent or smoldering SM (ISM)
How Has the RYDAPT Market Evolved?
Market Penetration and Sales Trends
- Launch year (2017): $150 million in global sales
- Peak sales (2020): approximately $500 million globally
- 2022 sales: roughly $460 million, indicating a plateau and minor decline
- Sales data sourced from IQVIA, 2022
Geographic Sales Breakdown
| Region |
2022 Sales (USD Million) |
Market Share (%) |
| U.S. |
$300 |
65 |
| Europe |
$100 |
22 |
| Rest of World |
$60 |
13 |
The U.S. dominates due to early approval and extensive clinical use, with Europe as the second-largest market. Emerging markets show limited uptake due to high costs and reimbursement barriers.
Earnings Drivers and Competitive Landscape
- Main revenue driver: FLT3-mutated AML treatment
- Competition: Gilteritinib (Xospata), approved for relapsed/refractory AML; Glasdegib (Daurismo) in AML; and potential pipeline agents targeting similar pathways
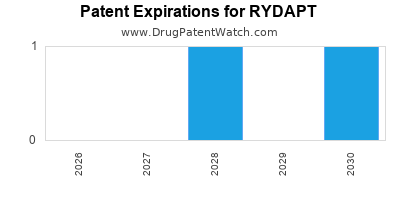
- Patent expiry: No imminent patent expiration; patent protections extend into the late 2020s
What Are the Market Drivers and Barriers?
Key Drivers
- Increasing incidence of AML: Approximately 20,000 new cases annually in the U.S. (SEER, 2022)
- Growing diagnosis rates of FLT3 mutations: Approximately 30% of AML cases in AML populations harbor FLT3 mutations
- Improving survival rates with targeted therapy: Median overall survival for FLT3-mutated AML patients improves with RYDAPT plus chemotherapy
Barriers to Growth
- High treatment cost: Estimated at $20,000 per month, limiting affordability
- Limited indications: Currently restricted to specific AML and SM subtypes
- Competition from newer agents: Gilteritinib shows favorable efficacy in relapsed AML, challenging RYDAPT’s market share
- Off-label use restrictions: Insurance coverage limits wider off-label application
What Is the Financial Trajectory Forecast?
Short-Term Outlook (2023-2025)
- Slight revenue decline expected due to market saturation and competition
- Forecasted global sales in 2023: approximately $440 million
- Potential growth from expanded indications or combination therapies remains uncertain
Mid-to-Long Term Outlook (2026-2030)
- Sales could stabilize around $300-400 million annually if existing indications persist
- Expansion into other hematologic malignancies or new molecular targets may boost revenues
- Market entry of biosimilars or lower-cost generics unlikely due to patent protection
Investment and R&D
- Novartis, which markets RYDAPT, allocated approximately $10 billion annually to R&D in 2022
- Focus on combination therapies: pairing RYDAPT with other agents to extend indications
- Potential pipeline: generic midostaurin expected around 2035, with patent expiration assumed in late 2020s
What Are the Key Opportunities and Risks?
Opportunities
- Acceleration of approval processes for novel indications
- Strategic alliances for combination therapy development
- Expanding access in emerging markets through partnerships
Risks
- Competitive pressure from newer agents with better efficacy profiles
- Regulatory setbacks or delays
- Pricing and reimbursement constraints due to high costs
Key Takeaways
- RYDAPT has established a niche in AML with FLT3 mutations and systemic mastocytosis
- Sales peaked around $500 million in 2020, with slight declines projected
- Growth relies on market expansion, pipeline developments, and competitive positioning
- Barriers include high cost, limited indications, and emerging competitors
Frequently Asked Questions
Q1: How long is RYDAPT patent protected?
A: Patent protection extends into the late 2020s, with generics expected around 2029 or 2030.
Q2: What is the primary competitor to RYDAPT?
A: Gilteritinib (Xospata) offers an alternative for relapsed AML and has some advantages in efficacy.
Q3: Are there ongoing trials for RYDAPT in other indications?
A: Yes, trials are exploring combinations with immunotherapies and potential new indications in hematologic malignancies.
Q4: How does RYDAPT's cost influence its market penetration?
A: The high monthly cost limits access, especially in cost-sensitive regions, constraining broader adoption.
Q5: What factors could significantly impact RYDAPT’s future sales?
A: Entry of biosimilars post-patent expiry and development of more effective therapies could reduce sales.
References
- SEER. (2022). Cancer Stat Facts: Acute Myeloid Leukemia. National Cancer Institute.
- IQVIA. (2022). Global Oncology Market Data.
- Novartis. (2022). Annual Report 2022.