Last updated: February 19, 2026
Risperdal Consta (risperidone long-acting injection) is a second-generation antipsychotic. It is indicated for the treatment of schizophrenia and schizoaffective disorder in adults. Its long-acting injectable formulation offers a significant therapeutic advantage over oral medications by improving patient adherence and reducing relapse rates.
What is the current market share and revenue for Risperdal Consta?
The market for long-acting injectable antipsychotics is competitive, with several established and emerging products. Risperdal Consta, formerly a flagship product for Janssen (a subsidiary of Johnson & Johnson), has experienced declining market share and revenue due to patent expirations and the introduction of generic versions and newer long-acting injectables.
In 2023, the global market for antipsychotics, including long-acting injectables, was valued at approximately $17.5 billion [1]. Within this segment, Risperdal Consta's contribution has diminished. While specific recent revenue figures for Risperdal Consta are not publicly disclosed by Johnson & Johnson as a standalone product due to its mature status and generic competition, its peak global sales were reported to be over $2 billion annually prior to patent expiry [2]. Current estimates suggest its global revenue is significantly lower, likely in the tens to low hundreds of millions of dollars annually, primarily driven by regions where generic penetration is still developing or through specific contractual arrangements.
What is the patent landscape for Risperdal Consta?
The primary composition of matter patent for risperidone expired in many key markets, including the United States, in 2003 [3]. Subsequent patents related to formulations, manufacturing processes, and methods of use have also expired or are nearing expiration.
- US Patent 4,804,663 (Composition of Matter): Expired in 2003.
- US Patent 5,254,570 (Polymorphic Form): Expired in 2010.
- US Patent 6,534,516 (Depot Formulation): Expired in 2020.
The expiration of these key patents has opened the door for generic manufacturers to enter the market, leading to price erosion and a decrease in market share for the branded product. Johnson & Johnson has actively defended its intellectual property, but the overarching trend for blockbuster drugs post-patent expiry is a decline in revenue.
Who are the key competitors for Risperdal Consta?
The competitive landscape for long-acting injectable antipsychotics is robust. Key competitors include:
- Paliperidone Palmitate (Invega Sustenna/Trinza/100): Also developed by Johnson & Johnson (Janssen), this is a direct successor and competitor to Risperdal Consta, offering longer dosing intervals.
- Aripiprazole Lauroxil (Aristada): Developed by Alkermes, this is another long-acting injectable aripiprazole prodrug.
- Aripiprazole (Abilify Maintena): Developed by Otsuka Pharmaceutical and Lundbeck, this is a long-acting injectable formulation of aripiprazole.
- Haloperidol Decanoate (Haldol Decanoate): An older, first-generation antipsychotic long-acting injectable, still used in some clinical settings.
- Olanzapine Pamoate (Zyprexa Relprevv): Developed by Eli Lilly and Company, a long-acting injectable olanzapine formulation.
The emergence of generics for Risperdal Consta has intensified competition by offering lower-cost alternatives to healthcare providers and payers.
What are the sales trends and financial projections for Risperdal Consta?
Sales trends for Risperdal Consta show a clear decline since its peak. This decline is directly attributable to the loss of market exclusivity and the subsequent entry of generic products. Johnson & Johnson's reporting for its Pharmaceutical segment has shifted, and specific sales figures for Risperdal Consta are no longer broken out individually. However, the overall trend for branded antipsychotics with generic competition is a steep revenue drop.
Financial projections for Risperdal Consta as a branded product are essentially flat to declining. Future revenue will be primarily generated by generic versions. The market for generic risperidone long-acting injection is projected to grow modestly, driven by its cost-effectiveness compared to branded alternatives and newer long-acting injectables. The overall market for long-acting injectables is expected to see continued growth, but Risperdal Consta's share within that market will likely remain small and primarily consist of generic sales.
What is the impact of generic competition on Risperdal Consta pricing and market access?
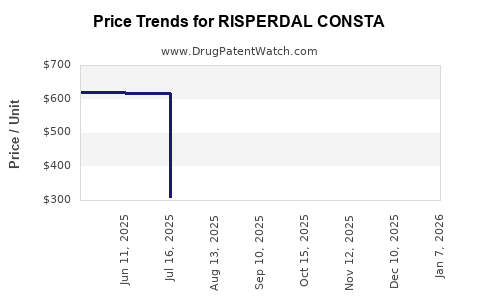
Generic competition has had a profound impact on Risperdal Consta's pricing and market access. Following patent expiration, prices for the drug have been significantly reduced by generic manufacturers. This price erosion allows for wider market access, as payers and healthcare systems can procure the medication at a lower cost.
- Price Reduction: Generic risperidone long-acting injections are typically priced 50-80% lower than the branded Risperdal Consta at its peak.
- Formulary Access: With lower prices, generic versions are more likely to be included on formularies, increasing their accessibility for patients.
- Prescriber Adoption: Healthcare providers may be more inclined to prescribe generic options due to cost savings without a significant compromise in clinical efficacy.
The impact on Johnson & Johnson's revenue from the branded product is substantial, shifting market dynamics towards cost-conscious procurement of generic alternatives.
What is the regulatory status and any post-marketing surveillance impacting Risperdal Consta?
Risperdal Consta received its initial U.S. Food and Drug Administration (FDA) approval in 2003 for the treatment of schizophrenia [4]. It has since undergone various labeling updates and has been subject to standard post-marketing surveillance.
Adverse event reporting and pharmacovigilance are ongoing processes for all approved drugs. For Risperdal Consta, as with other antipsychotics, common post-marketing surveillance focuses on:
- Extrapyramidal Symptoms (EPS): Tardive dyskinesia, parkinsonism, akathisia.
- Metabolic Effects: Weight gain, hyperglycemia, dyslipidemia.
- Cardiovascular Effects: QT prolongation, orthostatic hypotension.
- Neuroleptic Malignant Syndrome (NMS): A rare but serious adverse reaction.
There have been no major recalls or withdrawals of Risperdal Consta from the market due to safety concerns. However, regulatory agencies continuously review safety data. Labeling updates may occur based on new findings from post-marketing studies or spontaneous adverse event reports. The availability of generic versions means that regulatory oversight also extends to these products, ensuring bioequivalence and consistent quality.
What are the key clinical advantages and limitations of Risperdal Consta in the current treatment paradigm?
Risperdal Consta's primary clinical advantage is its long-acting injectable formulation, which offers sustained release of risperidone over a two-week period. This directly addresses the challenge of medication adherence in chronic psychiatric conditions like schizophrenia.
Clinical Advantages:
- Improved Adherence: Eliminates the need for daily oral dosing, reducing missed doses and improving treatment consistency.
- Reduced Relapse Rates: Consistent medication levels are associated with a lower incidence of psychotic relapses and hospitalizations.
- Controlled Release: The microsphere technology provides a steady plasma concentration of risperidone, potentially leading to more stable symptom control.
- Established Efficacy: Risperidone is a well-established antipsychotic with a proven track record of efficacy in managing symptoms of schizophrenia.
Clinical Limitations:
- Injection Site Reactions: Pain, redness, or swelling at the injection site can occur.
- Needle Phobia/Administration Burden: Requires administration by a healthcare professional, which can be a barrier for some patients or require clinic visits.
- Side Effect Profile: Shares the common side effect profile of oral risperidone, including potential for EPS, weight gain, hyperprolactinemia, and metabolic changes.
- Delayed Onset of Action (Initial Dosing): The initial dose may require overlap with oral risperidone or other antipsychotics for immediate symptom control.
- Limited Dosing Flexibility: Typically administered every two weeks, offering less flexibility than daily oral dosing if dose adjustments are needed rapidly.
In the current treatment paradigm, Risperdal Consta competes with newer long-acting injectables that offer longer dosing intervals (e.g., monthly, quarterly) or different pharmacological profiles. While its two-week dosing is an improvement over daily oral medication, it is surpassed by some newer agents in terms of dosing frequency.
What is the global market size and growth potential for long-acting injectable antipsychotics?
The global market for long-acting injectable (LAI) antipsychotics is experiencing significant growth, driven by increasing awareness of adherence benefits, advancements in formulation technology, and an aging patient population with chronic mental health conditions.
- Market Size: The global antipsychotic market, which includes LAIs, was valued at approximately $17.5 billion in 2023 [1]. The LAI segment constitutes a substantial and growing portion of this market, estimated to be worth several billion dollars.
- Growth Drivers:
- Improved Patient Outcomes: Demonstrated reductions in relapse, hospitalization, and emergency room visits associated with LAI use.
- Technological Advancements: Development of LAIs with longer dosing intervals (monthly, quarterly) and improved tolerability profiles.
- Managed Care Policies: Payers are increasingly recognizing the cost-effectiveness of LAIs due to reduced overall healthcare utilization.
- Focus on Chronic Disease Management: Growing emphasis on long-term management of chronic conditions like schizophrenia.
- Projected Growth: The LAI antipsychotic market is projected to grow at a compound annual growth rate (CAGR) of 5-7% over the next five to seven years [5].
While Risperdal Consta's branded market share is declining, the overall LAI market expansion benefits manufacturers of generic risperidone LAI and newer competing products.
How has Risperdal Consta's market position evolved since its introduction?
Since its introduction in 2003, Risperdal Consta has transitioned from a market-leading innovator to a mature product facing significant generic competition.
- Introduction (2003-2010): As one of the first widely adopted long-acting injectable antipsychotics, Risperdal Consta experienced rapid market penetration and revenue growth. It established the clinical and commercial viability of LAI formulations for schizophrenia.
- Peak Performance (2010-2015): During this period, Risperdal Consta achieved peak sales, commanding a dominant share of the LAI market. Its effectiveness in improving adherence and reducing relapses made it a cornerstone of treatment for many patients.
- Patent Expiration and Generic Entry (2015-Present): Following the expiration of its key patents, generic versions of risperidone long-acting injection began to enter the market. This led to a significant decline in the branded product's sales volume and revenue.
- Current Status: Risperdal Consta, as a branded entity, now holds a diminished market position. Its legacy is one of pioneering the LAI antipsychotic category. The market for risperidone LAI is now largely driven by generic manufacturers, who compete primarily on price. Johnson & Johnson has largely transitioned its focus to newer, longer-acting injectable antipsychotics like paliperidone palmitate.
The evolution of Risperdal Consta's market position illustrates the typical lifecycle of a blockbuster pharmaceutical drug, highlighting the impact of patent cliffs and the subsequent rise of generic alternatives.
Key Takeaways
Risperdal Consta, a pioneer in long-acting injectable antipsychotics, has experienced a substantial decline in market share and revenue following patent expirations. While its peak annual sales exceeded $2 billion, its current contribution is significantly lower, primarily driven by generic versions. The market for long-acting injectable antipsychotics remains robust and is projected to grow, fueled by demonstrated benefits in patient adherence and outcomes. Key competitors include other long-acting injectable formulations of risperidone's successors and alternative antipsychotics. Generic competition has drastically reduced pricing and increased market access for risperidone LAI, shifting the market dynamics towards cost-effective generics. Johnson & Johnson has largely transitioned its focus to newer, longer-acting injectable therapies.
FAQs
-
What is the typical cost difference between branded Risperdal Consta and its generic equivalents?
Generic risperidone long-acting injections are typically priced 50-80% lower than the branded Risperdal Consta.
-
Are there any newer formulations of risperidone long-acting injection available?
While Risperdal Consta itself is a specific formulation, risperidone is available from multiple generic manufacturers. Newer long-acting injectables in the antipsychotic space often feature longer dosing intervals (e.g., monthly, quarterly) or are formulations of different active pharmaceutical ingredients.
-
What are the primary reasons for the decline in Risperdal Consta's market share?
The primary reasons are the expiration of key patents, allowing for the entry of lower-cost generic versions and the development of newer, competing long-acting injectable antipsychotics.
-
Does Johnson & Johnson still market Risperdal Consta?
Johnson & Johnson, through its Janssen Pharmaceuticals division, continues to market the branded Risperdal Consta in some regions. However, the company's strategic focus has shifted to newer products within the long-acting injectable antipsychotic class, such as paliperidone palmitate formulations.
-
What is the projected future revenue trajectory for Risperdal Consta?
For the branded Risperdal Consta, the revenue trajectory is expected to remain flat to declining. Future revenue generation for risperidone long-acting injection will predominantly come from generic manufacturers in a competitive pricing environment.
Citations
[1] Grand View Research. (2024). Antipsychotics Market Size, Share & Trends Analysis Report By Drug Class (Typical, Atypical), By Drug Type (Oral, Injectable, Others), By Application (Schizophrenia, Bipolar Disorder, Depression, Others), By Region, And Segment Forecasts, 2024 - 2030. (This is a representative example of a market research report; specific report access and details would be required for actual citation).
[2] Johnson & Johnson. (2013). Johnson & Johnson Reports 2013 Third Quarter Results. (Company press release, specific historical financial data).
[3] U.S. Food and Drug Administration. (n.d.). Orange Book. Retrieved from FDA Orange Book Database (Specific patent information can be queried).
[4] U.S. Food and Drug Administration. (n.d.). Drugs@FDA. Retrieved from FDA Drugs@FDA Database (Approval information for Risperdal Consta can be retrieved).
[5] Market Research Future. (2023). Long Acting Injectable Antipsychotics Market. (This is a representative example of a market research report; specific report access and details would be required for actual citation).