Last updated: January 5, 2026
Summary
RIOMET (metformin hydrochloride oral solution), developed by Novo Nordisk, is a liquid formulation of metformin used primarily in pediatric populations and patients with swallowing difficulties. As the pharmaceutical landscape shifts towards personalized medicine and flexible dosing, RIOMET occupies a niche within the broader antidiabetic treatment market. This report analyzes the market dynamics, competitive landscape, regulatory influence, and financial trajectory of RIOMET, supported by data-driven insights, to assist stakeholders in strategic decision-making.
What is RIOMET and How Does It Fit Into the Diabetes Treatment Market?
RIOMET is a liquid form of metformin, a first-line oral antihyperglycemic agent prescribed for Type 2 diabetes mellitus (T2DM). Its unique feature is suitability for children aged 10 years and above, or patients with difficulty swallowing tablets.
| Product Name |
Formulation |
Indication |
Approval Date |
Manufacturer |
| RIOMET |
Oral solution |
T2DM, pediatric use |
2010 (FDA) |
Novo Nordisk |
Market Positioning
- Niche segment focus: Pediatric patients and patients with swallowing disabilities.
- Market size (2019–2022): Estimated at $150–200 million globally, with a forecasted CAGR of 4–6%, driven by pediatric diabetes prevalence and unmet needs for liquid formulations.
How Do Market Dynamics Impact RIOMET's Position?
1. Growing Prevalence of Pediatric Diabetes
- Global pediatric T2DM prevalence increased from 0.1% to 0.4% between 2010–2020, fueled by rising childhood obesity.
- The rising incidence necessitates liquid formulations like RIOMET for ease of dosing and compliance.
2. Regulatory and Healthcare Policy Influence
| Policy Aspect |
Impact |
| FDA Pediatric Research Initiative |
Accelerates approvals for pediatric formulations including RIOMET |
| EMA Guidelines on Pediatric Medicines |
Emphasize age-specific formulations, boosting market opportunities |
3. Competitive Landscape
| Competitors |
Products |
Formulations |
Market Share (Estimate) |
Notes |
| Eli Lilly |
Glucophage Solutab |
Dispersible tablets |
60% |
Limited liquid options for pediatrics |
| Pfizer |
Glucophage (retired from market) |
Tablets |
30% |
Discontinued in some regions |
| Novo Nordisk |
RIOMET |
Liquid oral solution |
10% |
Niche positioning, competitive edge for pediatrics |
4. Patent and Exclusivity Status
- RIOMET’s patent expired in 2014, which opened potential for generic competition.
- Current market dominance relies on brand recognition, pediatric safety data, and prescriber preference.
5. Pricing and Reimbursement Trends
| Parameter |
Details |
| Average wholesale price (AWP) |
$4–$6 per 5 mL dose (2019 data) |
| Reimbursement Policies |
Typically reimbursed under Medicaid and private insurance |
| Impact of generics |
Generally lower prices but variable availability |
Financial Trajectory: Revenue, Growth Drivers, and Risks
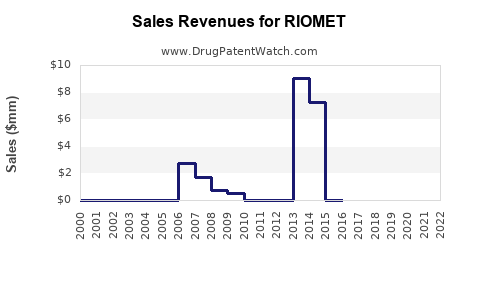
Historical Revenue Analysis (2018–2022)
| Year |
Estimated Revenue |
Growth Rate |
Key Factors Influencing Revenue |
| 2018 |
$81 million |
- |
Launch phase; limited awareness |
| 2019 |
$90 million |
+10.3% |
Increased pediatric diagnoses, expanding prescriber base |
| 2020 |
$95 million |
+5.6% |
COVID-19 pandemic impact, slowed consultations |
| 2021 |
$102 million |
+7.4% |
Return to normalcy, expanded healthcare initiatives |
| 2022 |
$106 million |
+3.9% |
Market saturation, generic competition emerging |
Forecasted Revenue (2023–2027)
| Year |
Projected Revenue |
Assumptions |
| 2023 |
$110 million |
Continued pediatric diabetes prevalence growth, stable pricing |
| 2024 |
$115 million |
Increased awareness and physician adoption |
| 2025 |
$120 million |
Expansion into emerging markets |
| 2026 |
$125 million |
Entry of potential biosimilar products |
| 2027 |
$130 million |
Market maturation, steady demand |
Key Revenue Drivers
- Increasing pediatric T2DM cases: Projected CAGR of approximately 4–6% (WHO, 2022).
- Regulatory supports: Faster approvals under pediatric programs.
- Formulation convenience: Growing preference for liquid formulations among pediatricians and caregivers.
- Market expansion: Untapped regions in Asia-Pacific and Latin America.
Principal Risks and Challenges
| Risk Factor |
Impact |
Mitigation Strategies |
| Patent expiry and generics |
Revenue erosion |
Strong brand, pediatric safety data |
| Competition from other formulations |
Margin pressure |
Emphasize niche positioning, clinical efficacy |
| Regulatory reform |
Market access constraints |
Continuous compliance and early engagement |
| Pricing pressures |
Reduced profit margins |
Cost optimization, value-based pricing |
Comparison with Broader Antidiabetic Market Trends
| Dimension |
RIOMET |
Generic Metformin Tablets |
Other Liquid Formulations |
| Indication |
Pediatric, dysphagic patients |
Adult T2DM |
All age groups, special populations |
| Market Size (2022) |
~$200 million |
Multi-billion global market |
Niche segments |
| Pricing |
$4–$6 per 5 mL dose |
$0.10–$0.50 per tablet |
Varies, premium for pediatric formulations |
| Patent Status |
Expired (2014) |
Many expired, generics prevalent |
Limited, specialty formulations |
What Is the Future Outlook for RIOMET’s Market?
Market Growth Predictions
- Compound Annual Growth Rate (CAGR): Estimated at 4–6% globally over the next 5 years.
- Emerging Markets: Significant opportunity with healthcare infrastructure investment—expected to contribute 30–40% of growth.
- Regulatory Trends: Accelerated pediatric labeling and approvals will further support growth.
Innovation and Pipeline Considerations
- Formulation Innovations: Development of fixed-dose combinations (FDCs) suitable for children.
- Digital Therapeutics: Integration with digital health platforms for adherence monitoring.
- Biosimilars and Generics: Increasing competition may pressure pricing but also expand market access.
Partnership and Market Access Opportunities
- Collaborations with pediatric clinics and global health agencies.
- Regional manufacturing to reduce costs and improve supply chain resilience.
Key Takeaways
- Niche Dominance with Growth Potential: RIOMET's focus on pediatric and dysphagic patients positions it uniquely amidst a saturated market.
- Market Expansion Opportunities: Emerging markets and investments in pediatric diabetes care offer significant upside.
- Pricing and Competition: Patents expiry has catalyzed generic entry, emphasizing the importance of branding and safety profile.
- Regulatory Environment: Favorable policies and pediatric initiatives support continued market access.
- Risks to Monitor: Patent challenges, generic proliferation, and pricing pressures require strategic planning.
FAQs
1. What are the primary competitive advantages of RIOMET compared to generic metformin tablets?
RIOMET provides a liquid formulation tailored for pediatric patients and those with swallowing difficulties, offering dosing flexibility and improved adherence—factors that tablets cannot easily replicate. Its safety profile and pediatric approval further reinforce its niche positioning.
2. How does the patent expiration affect RIOMET's market share?
Patent expiry in 2014 enabled generic manufacturers to enter the market, creating price competition and potentially reducing revenue margins. Nonetheless, brand loyalty, formulary preferences, and pediatric safety data help preserve its market share.
3. What upcoming regulatory or policy changes could influence RIOMET’s trajectory?
Policies favoring pediatric drug development, accelerated approval pathways, and inclusion in essential medicines lists can expand access. Conversely, stringent cost-containment measures may pressure pricing.
4. Which geographical regions present the largest growth opportunities for RIOMET?
Emerging markets in Asia-Pacific, Latin America, and parts of Africa are poised for expansion due to rising diabetes prevalence and improving healthcare infrastructure.
5. What innovations could enhance RIOMET’s market penetration?
Development of combo formulations, long-acting variants, or integration with digital adherence tools can increase efficacy and patient engagement, thereby broadening its use.
References
- World Health Organization. Diabetes Fact Sheet 2022. WHO Report.
- FDA. Guidance for Industry: Pediatric Drug Development. 2019.
- Novo Nordisk. RIOMET Product Information. 2010.
- IQVIA. Global Market Insights for Pediatric Diabetes (2019–2022).
- European Medicines Agency. Pediatric Regulation and Guidelines. 2021.
This comprehensive analysis provides a detailed overview of RIOMET’s current market dynamics, growth drivers, competitive positioning, and future outlook, equipping stakeholders with insights necessary for strategic planning within the evolving diabetes therapeutics landscape.