Last updated: April 23, 2026
What does the RAMIPRIL commercial market look like by regulatory reality?
Ramipril is an ACE inhibitor with a long commercial history and broad global availability, which makes pricing and margins primarily a function of generic competition, tendering, and national reimbursement rules rather than ongoing patent-led exclusivity.
Regulatory status (commercial implication)
- Originator era has ended in most major markets, leaving the market to generic manufacturers.
- Brand-to-generic transition is typically mature, so growth is driven by population needs, hypertension prevalence, and formulary placement, not by new molecular innovation.
What the market structure means for pricing
- Generic-heavy supply compresses ex-manufacturer pricing.
- Tender and reimbursement controls dominate retail outcomes.
- Competitive intensity is driven by:
- availability of multiple ANDA/MAH supply chains,
- ability to pass quality and bioequivalence requirements at low cost,
- national prescribing habits and local formulary rules.
How does ramipril demand typically evolve in key therapeutic demand drivers?
Ramipril is used across chronic cardiovascular indications where adherence and long-term treatment persistence matter.
Main demand drivers
- Hypertension management: long-duration therapy supports baseline demand.
- Cardiovascular risk reduction: persistent patient pools from chronic disease and comorbidity management.
- Post-event treatment in appropriate labeled settings increases stickiness because discontinuation rates are low under guideline-driven care.
Demand elasticity and mix shifts
- Low price elasticity in chronic use: patients and clinicians typically accept switch-to-generic once tolerated.
- Mix shifts occur between:
- single-drug ACE inhibitors and fixed-dose combinations,
- ACE inhibitors versus ARBs in markets with higher ARB penetration.
- Ramipril tends to maintain share where it is embedded in long-standing guideline regimens and stable formularies.
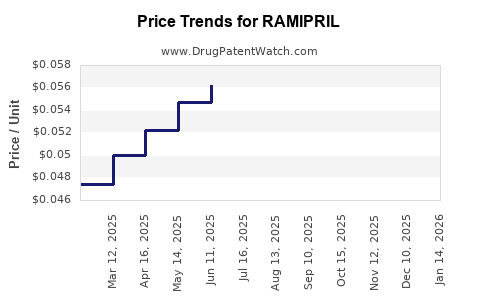
What does pricing pressure look like over time for ramipril?
For older ACE inhibitors, the pricing trajectory is shaped by stepwise generic entry rather than continuous erosion from a single event.
Typical financial pattern after generic entry
- Initial generic entry creates a rapid price drop from brand pricing to low-cost generic levels.
- Subsequent entries usually reduce prices further but at a slower rate as the market approaches a lower cost floor tied to:
- manufacturing scale,
- API sourcing costs,
- logistics and tender pricing dynamics,
- regulatory compliance costs.
Profit model under generic competition
- Sustained profitability depends on:
- scale manufacturing and stable API supply,
- tight cost structure and tender wins,
- minimal formulation differentiation risk (few meaningful product differentiators beyond bioequivalence, packaging, and supply reliability).
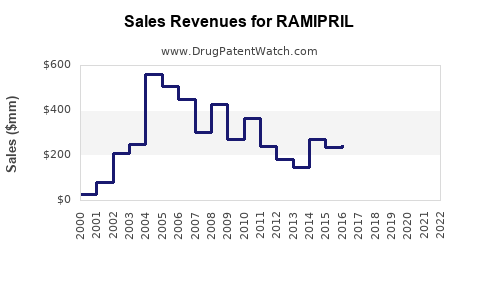
How does financial trajectory differ by business type: originator vs generics vs combination brands?
Ramipril’s financial story is mostly a story of business model, not molecule growth.
Originator P&L trajectory (brand)
- Post exclusivity end typically means:
- share loss to generics,
- gross margin compression,
- brand shrink unless the originator maintains defensive pricing in high-retention segments.
- Revenues track prescriptions and remaining brand share rather than unit growth in the molecule overall.
Generic manufacturers (core driver of current economics)
- Revenue grows with:
- volume capture in tenders and formularies,
- ability to supply consistently.
- Margin depends on:
- API cost, batch yield, and fixed cost absorption,
- tender pricing and contract duration.
Combination strategy (fixed-dose positioning)
- Where regulators and clinicians prefer fixed-dose combinations, ramipril can shift from standalone to combo products.
- Financial trajectory then depends on:
- the combo’s ability to win formulary placement,
- competitive combination ecosystems from multiple ACE inhibitor partners.
What is the likely trajectory for RAMIPRIL revenues and margins in the near-to-mid term?
Given the molecule’s maturity and broad generic availability, near-to-mid-term dynamics are typically characterized by:
- stable-to-slow revenue growth tied to underlying cardiovascular prevalence and adherence,
- continued margin compression where new entrants or aggressive tenders occur,
- offset from volume retention as switching stabilizes.
Revenue trajectory (directional)
- Volume: generally stable with growth tied to demographics and treatment rates.
- Value: can be pressured where payer systems push lowest-price supply.
- Share: shifts among generic suppliers based on tender cycles and supply continuity.
Margin trajectory (directional)
- Gross margin: pressured by price competition and API volatility.
- Operating margin: increasingly dependent on scale and manufacturing efficiency.
- Risk: disruptions in API supply can briefly improve prices for compliant suppliers, but the effect typically fades as alternative sources enter.
How do tenders, reimbursement, and switching policies drive quarter-by-quarter outcomes?
For mature ACE inhibitors, the main financial variability is operational and payer-driven.
Key levers
- Hospital and national tenders
- winners can lock in volume for months to a year,
- losers can experience sharp revenue declines even with stable underlying demand.
- Reimbursement rules
- reference pricing and substitution policies determine retail and payer reimbursement.
- Switching behavior
- once patients stabilize, switching is less frequent unless mandated by payer policy.
What financial analysts should watch
- tender award timing and contract expiries,
- supplier qualification changes,
- reference price updates,
- wholesale inventory cycles around contract changes.
What competitive landscape factors shape market share for ramipril tablets/capsules?
Competition is not only about price; it also includes quality, supply reliability, and packaging.
Market-share determinants
- Bioequivalence compliance and regulatory track record
- Supply reliability during tight tender timelines
- Manufacturing capacity and API sourcing resilience
- Packaging and labeling suitability for local formularies and substitution rules
Differentiation limits
- In a generic-heavy market, differentiation is mostly procedural:
- availability,
- stability and quality,
- logistics,
- documentation strength.
What macro and healthcare-policy factors can alter the financial trajectory?
Ramipril’s usage depends on cardiovascular care delivery and drug-policy enforcement.
Macro factors
- demographic growth in treated populations,
- changes in hypertension detection and treatment rates,
- economic pressure increasing payer sensitivity to lowest-cost options.
Policy factors
- generic substitution enforcement,
- price renegotiations,
- inclusion/exclusion in reimbursement lists,
- procurement reforms favoring multi-supplier tenders or sole-source contracts.
What does this imply for investment or R&D strategy tied to RAMIPRIL?
Ramipril’s commercial economics typically reward:
- supply-scale businesses,
- manufacturers with reliable API procurement,
- firms positioned to win tender-driven volume.
Where value can be created
- Manufacturing efficiency and cost absorption at high batch volumes.
- Geographic portfolio across multiple tender regimes to smooth revenue variability.
- Combination or lifecycle stewardship if the firm controls an in-formulary combination portfolio.
Where value leakage tends to occur
- reliance on single-country tender dependence,
- exposure to API supply constraints without alternative sourcing,
- pricing sensitivity without scale cost advantages.
Key Takeaways
- Ramipril is a mature, generic-dominated ACE inhibitor, so market dynamics are shaped by tendering, reimbursement, and substitution rather than patent exclusivity.
- Demand is structurally supported by chronic hypertension and cardiovascular risk reduction, producing relatively stable volume outcomes.
- Financial trajectory is dominated by pricing pressure, with margins determined by manufacturing scale, API cost control, and contract wins.
- Near-to-mid-term value growth is constrained; variability shows up in supplier-specific revenue swings around tenders and reference price updates.
- Strategic advantage concentrates in supply-scale and payer-facing execution, not in molecule-level differentiation.
FAQs
-
Is ramipril still growing as a molecule overall?
Typically yes in volume terms via population and treatment-rate dynamics, but value growth is limited by generic pricing and payer reference pricing.
-
What drives quarterly results for ramipril generic suppliers?
Tender outcomes, contract renewals, reference price updates, and supply continuity.
-
Does ramipril benefit from fixed-dose combinations?
In markets where combinations are preferred, ramipril often shifts from standalone to combo demand, changing unit economics and formulary exposure.
-
What is the main margin risk for ramipril manufacturers?
Continued price erosion from competitive entry and aggressive tenders, amplified by API or manufacturing cost volatility.
-
Is R&D the primary value lever for ramipril now?
Not usually. In mature ACE inhibitor classes, execution advantages in manufacturing, compliance, and procurement tend to matter more than molecule innovation.
References
[1] World Health Organization (WHO). WHO Model List of Essential Medicines. (Most recent edition accessible via WHO).