QSYMIA Drug Patent Profile
✉ Email this page to a colleague
When do Qsymia patents expire, and what generic alternatives are available?
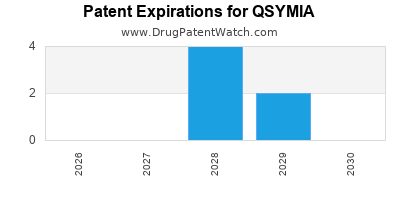
Qsymia is a drug marketed by Vivus Llc and is included in one NDA. There are six patents protecting this drug and one Paragraph IV challenge.
This drug has forty patent family members in seventeen countries.
The generic ingredient in QSYMIA is phentermine hydrochloride; topiramate. There are seventeen drug master file entries for this compound. Four suppliers are listed for this compound. Additional details are available on the phentermine hydrochloride; topiramate profile page.
DrugPatentWatch® Litigation and Generic Entry Outlook for Qsymia
A generic version of QSYMIA was approved as phentermine hydrochloride; topiramate by ACTAVIS LABS FL INC on June 25th, 2024.
AI Deep Research
Questions you can ask:
- What is the 5 year forecast for QSYMIA?
- What are the global sales for QSYMIA?
- What is Average Wholesale Price for QSYMIA?
Summary for QSYMIA
| International Patents: | 40 |
| US Patents: | 6 |
| Applicants: | 1 |
| NDAs: | 1 |
| Finished Product Suppliers / Packagers: | 2 |
| Raw Ingredient (Bulk) Api Vendors: | 4 |
| Clinical Trials: | 20 |
| Drug Prices: | Drug price information for QSYMIA |
| Patent Litigation and PTAB cases: | See patent lawsuits and PTAB cases for QSYMIA |
| What excipients (inactive ingredients) are in QSYMIA? | QSYMIA excipients list |
| DailyMed Link: | QSYMIA at DailyMed |
Recent Clinical Trials for QSYMIA
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| University of Minnesota | PHASE3 |
| University of Toronto | PHASE4 |
| Mayo Clinic | PHASE4 |
Pharmacology for QSYMIA
| Drug Class | Sympathomimetic Amine Anorectic |
| Mechanism of Action | Cytochrome P450 2C19 Inhibitors Cytochrome P450 3A4 Inducers |
| Physiological Effect | Appetite Suppression Decreased Central Nervous System Disorganized Electrical Activity Increased Sympathetic Activity |
Paragraph IV (Patent) Challenges for QSYMIA
| Tradename | Dosage | Ingredient | Strength | NDA | ANDAs Submitted | Submissiondate |
|---|---|---|---|---|---|---|
| QSYMIA | Extended-release Capsules | phentermine hydrochloride; topiramate | 3.75 mg/23 mg 7.5 mg/46 mg 11.25 mg/69 mg 15 mg/92 mg | 022580 | 1 | 2013-07-18 |
US Patents and Regulatory Information for QSYMIA
QSYMIA is protected by six US patents and one FDA Regulatory Exclusivity.
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Vivus Llc | QSYMIA | phentermine hydrochloride; topiramate | CAPSULE, EXTENDED RELEASE;ORAL | 022580-004 | Jul 17, 2012 | AB | RX | Yes | Yes | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | |||
| Vivus Llc | QSYMIA | phentermine hydrochloride; topiramate | CAPSULE, EXTENDED RELEASE;ORAL | 022580-001 | Jul 17, 2012 | AB | RX | Yes | No | 9,011,906 | ⤷ Start Trial | ⤷ Start Trial | |||
| Vivus Llc | QSYMIA | phentermine hydrochloride; topiramate | CAPSULE, EXTENDED RELEASE;ORAL | 022580-003 | Jul 17, 2012 | AB | RX | Yes | No | 8,895,058 | ⤷ Start Trial | Y | ⤷ Start Trial | ||
| Vivus Llc | QSYMIA | phentermine hydrochloride; topiramate | CAPSULE, EXTENDED RELEASE;ORAL | 022580-004 | Jul 17, 2012 | AB | RX | Yes | Yes | 8,580,299 | ⤷ Start Trial | ⤷ Start Trial | |||
| Vivus Llc | QSYMIA | phentermine hydrochloride; topiramate | CAPSULE, EXTENDED RELEASE;ORAL | 022580-001 | Jul 17, 2012 | AB | RX | Yes | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | |||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for QSYMIA
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Vivus Llc | QSYMIA | phentermine hydrochloride; topiramate | CAPSULE, EXTENDED RELEASE;ORAL | 022580-002 | Jul 17, 2012 | 6,071,537 | ⤷ Start Trial |
| Vivus Llc | QSYMIA | phentermine hydrochloride; topiramate | CAPSULE, EXTENDED RELEASE;ORAL | 022580-002 | Jul 17, 2012 | 7,659,256 | ⤷ Start Trial |
| Vivus Llc | QSYMIA | phentermine hydrochloride; topiramate | CAPSULE, EXTENDED RELEASE;ORAL | 022580-003 | Jul 17, 2012 | 7,659,256 | ⤷ Start Trial |
| Vivus Llc | QSYMIA | phentermine hydrochloride; topiramate | CAPSULE, EXTENDED RELEASE;ORAL | 022580-001 | Jul 17, 2012 | 7,553,818 | ⤷ Start Trial |
| Vivus Llc | QSYMIA | phentermine hydrochloride; topiramate | CAPSULE, EXTENDED RELEASE;ORAL | 022580-004 | Jul 17, 2012 | 7,659,256 | ⤷ Start Trial |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
International Patents for QSYMIA
When does loss-of-exclusivity occur for QSYMIA?
Based on analysis by DrugPatentWatch, the following patents block generic entry in the countries listed below:
Australia
Patent: 09257572
Estimated Expiration: ⤷ Start Trial
Patent: 09257573
Estimated Expiration: ⤷ Start Trial
Brazil
Patent: 0914985
Estimated Expiration: ⤷ Start Trial
Patent: 0914991
Estimated Expiration: ⤷ Start Trial
Canada
Patent: 27313
Estimated Expiration: ⤷ Start Trial
Patent: 27319
Estimated Expiration: ⤷ Start Trial
Chile
Patent: 10001365
Estimated Expiration: ⤷ Start Trial
Patent: 10001366
Estimated Expiration: ⤷ Start Trial
China
Patent: 2112126
Estimated Expiration: ⤷ Start Trial
Patent: 2112127
Estimated Expiration: ⤷ Start Trial
Patent: 4825477
Estimated Expiration: ⤷ Start Trial
Patent: 5534921
Estimated Expiration: ⤷ Start Trial
Cyprus
Patent: 18103
Estimated Expiration: ⤷ Start Trial
Denmark
Patent: 17997
Estimated Expiration: ⤷ Start Trial
European Patent Office
Patent: 00002
Estimated Expiration: ⤷ Start Trial
Patent: 17997
Estimated Expiration: ⤷ Start Trial
Hong Kong
Patent: 13489
Estimated Expiration: ⤷ Start Trial
Israel
Patent: 9874
Estimated Expiration: ⤷ Start Trial
Patent: 9875
Estimated Expiration: ⤷ Start Trial
Japan
Patent: 52595
Estimated Expiration: ⤷ Start Trial
Patent: 77053
Estimated Expiration: ⤷ Start Trial
Patent: 14750
Estimated Expiration: ⤷ Start Trial
Patent: 11522896
Estimated Expiration: ⤷ Start Trial
Patent: 11522897
Estimated Expiration: ⤷ Start Trial
Patent: 15166380
Estimated Expiration: ⤷ Start Trial
Patent: 16006085
Estimated Expiration: ⤷ Start Trial
Patent: 17078083
Estimated Expiration: ⤷ Start Trial
Patent: 17105788
Estimated Expiration: ⤷ Start Trial
Mexico
Patent: 2684
Estimated Expiration: ⤷ Start Trial
Patent: 10013503
Estimated Expiration: ⤷ Start Trial
Patent: 10013505
Estimated Expiration: ⤷ Start Trial
Portugal
Patent: 17997
Estimated Expiration: ⤷ Start Trial
South Africa
Patent: 1008839
Estimated Expiration: ⤷ Start Trial
Patent: 1008840
Estimated Expiration: ⤷ Start Trial
South Korea
Patent: 110042280
Estimated Expiration: ⤷ Start Trial
Patent: 110044847
Estimated Expiration: ⤷ Start Trial
Patent: 140121491
Estimated Expiration: ⤷ Start Trial
Spain
Patent: 06041
Estimated Expiration: ⤷ Start Trial
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
See the table below for additional patents covering QSYMIA around the world.
| Country | Patent Number | Title | Estimated Expiration |
|---|---|---|---|
| Japan | 2011522897 | ⤷ Start Trial | |
| European Patent Office | 2308481 | Thérapie combinée pour le traitement de l`hypertension associé à l'obésité (Combination therapy for the treatment of hypertension associated with obesity) | ⤷ Start Trial |
| China | 104825477 | ⤷ Start Trial | |
| Czech Republic | 9804278 | ⤷ Start Trial | |
| Hungary | 226775 | THE USE OF TOPIRAMATE AND ITS DERIVATIVES FOR PREPARATION OF PHARMACEUTICAL COMPOSITIONS USEFUL FOR TREATING OBESITY OR FOR WEIGHT LOSS | ⤷ Start Trial |
| >Country | >Patent Number | >Title | >Estimated Expiration |
Supplementary Protection Certificates for QSYMIA
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 2317997 | CA 2021 00049 | Denmark | ⤷ Start Trial | PRODUCT NAME: PHENTERMIN OG TOPIRAMAT; NAT. REG. NO/DATE: 63166, 63167, 63168, 63169 20210705; FIRST REG. NO/DATE: IS IS/1/21/018/01-04 20210212 |
| 2317997 | 2190050-1 | Sweden | ⤷ Start Trial | PRODUCT NAME: PHENTERMINE AND TOPIRAMATE; NAT. REG. NO/DATE: 59574-59577 20210617; FIRST REG.: IS IS/1/21/018/01-04 20210212 |
| 2317997 | 833 | Finland | ⤷ Start Trial | |
| 2317997 | CR 2021 00049 | Denmark | ⤷ Start Trial | PRODUCT NAME: PHENTERMIN OG TOPIRAMAT; NAT. REG. NO/DATE: 63166, 63167, 63168, 63169 20210705; FIRST REG. NO/DATE: IS IS/1/21/018/01-04 20210212 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Qsymia: Market Dynamics and Financial Trajectory
More… ↓
Make Better Decisions: Try a trial or see plans & pricing
Drugs may be covered by multiple patents or regulatory protections. All trademarks and applicant names are the property of their respective owners or licensors. Although great care is taken in the proper and correct provision of this service, thinkBiotech LLC does not accept any responsibility for possible consequences of errors or omissions in the provided data. The data presented herein is for information purposes only. There is no warranty that the data contained herein is error free. We do not provide individual investment advice. This service is not registered with any financial regulatory agency. The information we publish is educational only and based on our opinions plus our models. By using DrugPatentWatch you acknowledge that we do not provide personalized recommendations or advice. thinkBiotech performs no independent verification of facts as provided by public sources nor are attempts made to provide legal or investing advice. Any reliance on data provided herein is done solely at the discretion of the user. Users of this service are advised to seek professional advice and independent confirmation before considering acting on any of the provided information. thinkBiotech LLC reserves the right to amend, extend or withdraw any part or all of the offered service without notice.