Last updated: February 19, 2026
Progesterone, a key endogenous steroid hormone, exhibits significant market presence and financial potential within the pharmaceutical sector. Its therapeutic applications span reproductive health, oncology, and neurology, driving sustained demand and diverse revenue streams. Key market segments include hormone replacement therapy (HRT), contraception, gynecological disorders, and emerging indications.
What are the Primary Therapeutic Areas for Progesterone?
Progesterone's therapeutic utility is multifaceted, addressing a range of physiological and pathological conditions.
- Reproductive Health and Obstetrics: This is the largest segment, encompassing treatments for infertility, recurrent pregnancy loss, premenstrual syndrome (PMS), and symptoms associated with menopause. Progesterone is essential for preparing the uterus for implantation and maintaining pregnancy.
- Gynecological Disorders: Progesterone is a cornerstone treatment for conditions such as endometriosis, abnormal uterine bleeding, and uterine fibroids. It helps to regulate the menstrual cycle and counteract the proliferative effects of estrogen.
- Contraception: Combined oral contraceptives and progestin-only pills frequently incorporate synthetic progestins, mimicking progesterone's action to prevent ovulation.
- Oncology: Progesterone and its analogues are used in the treatment of certain hormone-sensitive cancers, particularly breast cancer and endometrial cancer. They can inhibit tumor growth by modulating hormone receptor signaling.
- Neurology: Emerging research suggests potential neuroprotective roles for progesterone, with investigations into its efficacy for traumatic brain injury, stroke, and neurodegenerative diseases.
What are the Major Market Segments and Their Growth Drivers?
The global progesterone market is segmented by application, formulation, and distribution channel.
-
Application Segments:
- Hormone Replacement Therapy (HRT): Driven by an aging global population and increasing awareness of menopause management, this segment shows consistent growth. The demand for bioidentical progesterone in HRT is a notable trend.
- Contraception: The high prevalence of unintended pregnancies globally fuels demand for hormonal contraceptives. Market growth is influenced by product innovation, accessibility, and healthcare policies.
- Gynecological Disorders Treatment: This segment benefits from an increasing diagnosis rate of conditions like endometriosis and abnormal uterine bleeding, alongside the development of more targeted progesterone-based therapies.
- Fertility Treatment: Growing rates of infertility, delayed childbearing, and advancements in assisted reproductive technologies (ART) are significant growth catalysts. Progesterone is critical in ovulation induction and luteal phase support during IVF cycles.
- Oncology: While a smaller segment, its growth is linked to advancements in cancer diagnostics and the development of novel progesterone receptor modulators.
-
Formulation Segments:
- Oral: The most common and convenient formulation, including tablets and capsules.
- Injectable: Used for longer-acting effects and in specific clinical scenarios, such as preventing preterm birth.
- Vaginal: Employed for direct delivery to the reproductive tract, reducing systemic side effects, particularly in HRT and fertility treatments. This includes gels, suppositories, and inserts.
- Transdermal: Creams and patches offering steady absorption, gaining traction for HRT.
-
Distribution Channels:
- Hospital Pharmacies: Significant channel for acute care and specialized treatments.
- Retail Pharmacies: Primary channel for chronic condition management and contraceptives.
- Online Pharmacies: Growing channel, offering convenience and accessibility.
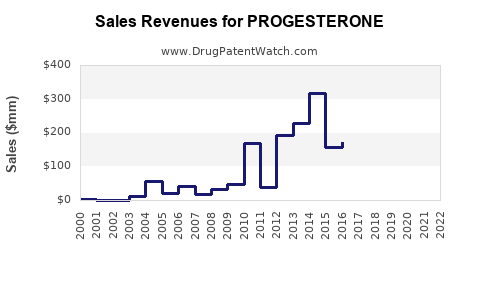
What is the Global Market Size and Projected Financial Trajectory?
The global progesterone market has demonstrated robust financial performance.
| Metric |
Value (USD Billion) |
Projected Compound Annual Growth Rate (CAGR) |
Year |
Source Reference |
| Market Size |
2.5 |
4.8% |
2023 (Estimated) |
[1] |
| Projected Size |
3.5 |
|
2028 (Projected) |
[1] |
| Market Size |
2.2 |
4.5% |
2022 (Estimated) |
[2] |
| Projected Size |
3.3 |
|
2029 (Projected) |
[2] |
The market's growth is driven by an increasing prevalence of hormonal imbalances, a rising aging population seeking HRT, and a growing demand for fertility treatments. Technological advancements in drug delivery systems and the development of new therapeutic applications also contribute to the positive financial outlook.
What are the Key Market Trends and Innovations?
Several trends are shaping the progesterone market landscape.
- Shift Towards Bioidentical Hormones: There is a growing patient and physician preference for bioidentical progesterone (chemically identical to endogenous progesterone) over synthetic progestins in HRT and fertility treatments. This is due to perceived improved safety profiles and reduced side effects.
- Advancements in Drug Delivery Systems: Innovations in vaginal delivery systems (e.g., extended-release inserts, targeted gel formulations) are enhancing efficacy and patient compliance for gynecological and fertility applications. Development of long-acting injectable formulations is also a key area of research.
- Exploration of Novel Therapeutic Indications: Research into progesterone's neuroprotective properties is a significant area of emerging opportunity. Clinical trials investigating its use in stroke recovery and traumatic brain injury could open new, substantial market segments if successful.
- Increased Focus on Personalized Medicine: As understanding of progesterone receptor signaling deepens, there is a move towards tailoring progesterone therapies based on individual patient profiles, including genetic predispositions and specific disease biomarkers.
- Regulatory Landscape Impact: Evolving regulatory guidelines regarding hormone therapy safety and efficacy, particularly for HRT, influence product development and market access. The FDA's ongoing review of hormone therapy risks and benefits is a critical factor.
What is the Competitive Landscape and Key Players?
The progesterone market is characterized by a mix of large pharmaceutical companies and specialized biopharmaceutical firms. Competition centers on product efficacy, safety, formulation innovation, and market access.
-
Key Manufacturers and Product Developers:
- AbbVie Inc.: Offers products for endometriosis and uterine bleeding, including those utilizing progestins.
- Pfizer Inc.: A major player in contraception and HRT with a broad portfolio of hormonal products.
- Bayer AG: Significant presence in contraception and gynecological therapies.
- Theramex: Focuses on women's health, including HRT and contraception.
- Exeltis USA: Offers progesterone products for fertility and gynecological indications.
- Mayne Pharma Group Limited: Produces progesterone formulations for various therapeutic uses.
- Sun Pharmaceutical Industries Ltd.: A diversified pharmaceutical company with progesterone-based products.
- Merck & Co., Inc. (MSD): Involved in hormonal therapies, including those related to reproductive health.
-
Market Entry Strategies: Companies employ strategies such as mergers and acquisitions, strategic partnerships for research and development, and expansion into emerging markets to strengthen their competitive positions. Patent protection for novel formulations and therapeutic uses is a critical aspect of market strategy.
What are the Regulatory and Patent Landscape Considerations?
The regulatory and patent landscape significantly influences market dynamics and investment decisions.
- Regulatory Approvals: Progesterone products require stringent regulatory approval from bodies like the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA). Approval pathways vary based on the indication and formulation. Specific guidelines exist for HRT, contraception, and fertility treatments.
- Patent Exclusivity: Patents provide market exclusivity for a defined period, protecting intellectual property for novel progesterone compounds, formulations, manufacturing processes, and therapeutic uses. The expiration of key patents can lead to increased generic competition and price erosion.
- Generic Competition: Once patents expire, generic manufacturers can enter the market, often at lower price points. This intensifies competition and can impact revenue for originator products.
- Post-Marketing Surveillance: Pharmaceutical companies are required to conduct post-marketing surveillance to monitor the safety and efficacy of their products. Adverse event reporting and pharmacovigilance are critical.
- Intellectual Property Litigation: Disputes over patent infringement are common in the pharmaceutical industry and can impact market exclusivity and financial outcomes for companies.
What are the Key Challenges and Opportunities?
The progesterone market presents both challenges and opportunities for stakeholders.
Challenges:
- Safety Concerns and Side Effect Profiles: Although generally well-tolerated, progesterone therapies can be associated with side effects such as mood changes, weight fluctuations, and gastrointestinal issues. Concerns regarding the long-term safety of HRT have also impacted market perception and prescribing habits.
- Stringent Regulatory Requirements: Navigating the complex and evolving regulatory landscape for drug approval and marketing is resource-intensive and time-consuming.
- Intense Competition from Generics: Patent expirations lead to increased competition from generic products, which can significantly reduce profit margins for branded therapies.
- Reimbursement Policies: Variable reimbursement policies from public and private payers can affect product affordability and market access, particularly for newer or more expensive formulations.
- Public Perception and Misinformation: Public perception of hormone therapy, influenced by historical studies and media coverage, can create barriers to adoption for certain treatments.
Opportunities:
- Growing Demand in Emerging Markets: Increasing healthcare infrastructure, rising disposable incomes, and growing awareness of reproductive health issues in emerging economies present significant growth opportunities.
- Development of Novel Formulations: Innovations in drug delivery, such as sustained-release formulations and targeted delivery systems, can improve patient compliance, reduce side effects, and create differentiated products.
- Expansion into New Therapeutic Areas: Continued research into progesterone's potential applications in neurology, immunology, and other areas could lead to the development of entirely new market segments.
- Personalized and Precision Medicine: Leveraging biomarkers and genetic profiling to tailor progesterone therapies can improve treatment outcomes and create value-added services.
- Addressing Unmet Needs in Women's Health: A significant unmet need exists for effective and well-tolerated treatments for various women's health conditions, providing a fertile ground for innovation in progesterone-based therapies.
Key Takeaways
The global progesterone market is projected for steady growth, driven by its essential role in reproductive health, obstetrics, and gynecological treatments. Key growth drivers include an aging population, increasing infertility rates, and advancements in therapeutic applications. The market is characterized by a trend towards bioidentical hormones and innovation in drug delivery systems. While facing challenges from regulatory hurdles and generic competition, opportunities exist in emerging markets, novel therapeutic indications, and personalized medicine approaches.
Frequently Asked Questions
-
What is the primary difference between bioidentical progesterone and synthetic progestins?
Bioidentical progesterone is chemically identical to the progesterone produced by the human body, whereas synthetic progestins are chemically altered versions. This difference can influence their binding affinity to receptors and their metabolic pathways, potentially leading to varied efficacy and side effect profiles.
-
How does patent expiration impact the financial outlook for progesterone-based drugs?
Patent expiration typically leads to increased competition from generic manufacturers, often resulting in a significant decrease in the price of the drug and a corresponding reduction in revenue for the originator company. This necessitates a focus on pipeline development and lifecycle management for pharmaceutical firms.
-
What are the key regulatory considerations for a new progesterone formulation seeking FDA approval?
A new progesterone formulation would undergo rigorous review by the FDA, requiring extensive preclinical and clinical data demonstrating its safety and efficacy for the intended indication. This includes studies on pharmacokinetics, pharmacodynamics, toxicology, and well-controlled clinical trials.
-
Which emerging therapeutic area for progesterone shows the most significant future market potential?
The potential neuroprotective role of progesterone, particularly in conditions like stroke and traumatic brain injury, is considered a significant emerging therapeutic area with substantial future market potential, provided clinical trials demonstrate clear efficacy and safety.
-
How does reimbursement policy affect the adoption of advanced progesterone formulations?
Reimbursement policies from insurance providers and government health programs can significantly impact the affordability and accessibility of advanced progesterone formulations. If not adequately reimbursed, the higher cost of these formulations may limit their uptake by patients and healthcare providers.
Citations
[1] Grand View Research. (2024). Progesterone Market Size, Share & Trends Analysis Report by Application (HRT, Contraception, Fertility Treatment, Gynecology), By Formulation (Oral, Injectable, Vaginal), By Region, And Segment Forecasts, 2024 - 2030. Retrieved from https://www.grandviewresearch.com/industry-analysis/progesterone-market
[2] Allied Market Research. (2023). Progesterone Market: Global Opportunity Analysis and Industry Forecast, 2023-2032. Retrieved from https://www.alliedmarketresearch.com/progesterone-market-A15760