Last updated: February 16, 2026
Prednisone, a corticosteroid used for its anti-inflammatory and immunosuppressive properties, has sustained demand across multiple therapeutic areas, including autoimmune diseases, allergic conditions, and certain cancers. Its market environment is shaped by patent status, generic availability, evolving clinical guidelines, and regulatory policies.
Current Market Landscape
Global Market Size and Growth
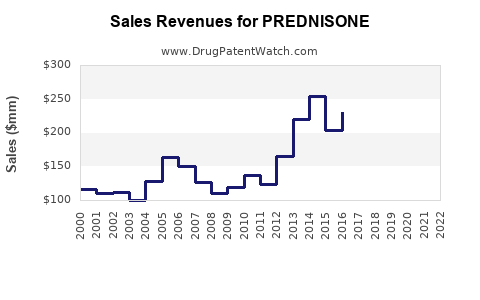
The global prednisone market was valued at approximately $1.2 billion in 2022 and is projected to grow at a compound annual growth rate (CAGR) of 3.2% through 2027, reaching nearly $1.5 billion. The growth is driven by expanded use in autoimmune conditions such as rheumatoid arthritis, asthma, and other inflammatory diseases.
Geographic Distribution
North America accounts for 45% of sales, owing to established healthcare infrastructure and the prevalence of autoimmune disorders. Europe holds around 30%, with growth influenced by aging demographics. Asia-Pacific, with a CAGR of 4.5%, exhibits increasing demand due to expanding healthcare access.
Market Segments
- Branded formulations: Comprise less than 10% of sales; primarily used where specific clinical preference persists.
- Generic versions: Constitute over 90%, dominant due to patent expiration (primarily post-2010).
Patent Status and Generics Influence
Patent Expiry Timeline
Prednisone’s primary patent expired by 2010 in the U.S. and EU, leading to widespread generic manufacturing. This transition has significantly depressed branded prices and concentrated market share among generics.
Impact of Generics
The post-patent environment has reduced manufacturing barriers, leading to increased competition and price erosion—downward pressure has reduced unit prices by approximately 50% between 2010 and 2022. Innovations, such as controlled-release formulations, remain patent-protected but account for a small market segment.
Pricing and Reimbursement Factors
Pricing Trends
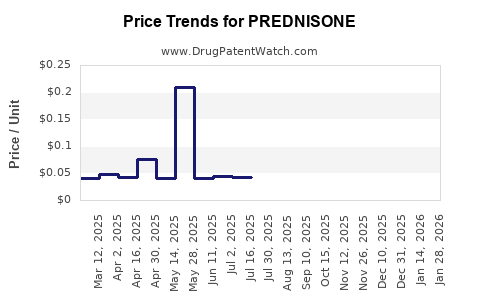
Average wholesale prices (AWP) for prednisone tablets decreased from ~$0.20 per 5mg dose in 2010 to ~$0.10 by 2022. Variability exists regionally, driven by reimbursement policies, pharmacy benefit management, and healthcare system structures.
Reimbursement Policies
In the U.S., Medicaid and Medicare Part D coverage favor generics, limiting revenue opportunities for branded versions. In Europe, national healthcare systems negotiate drug prices, which constrains revenue growth.
Regulatory Environment and Clinical Guidelines
Regulatory Approvals
Manufacturers must adhere to stringent quality standards mandated by FDA and EMA. Regulatory approval for new formulations is restricted due to the drug's age. However, orphan indication approvals can create new revenue streams.
Clinical Relevance and Usage Trends
Guidelines favor corticosteroids for specific indications but emphasize minimizing long-term use due to adverse effects. Use has plateaued in some therapeutic areas, limiting new sales opportunities but maintaining stable demand for existing formulations.
Competitive Landscape
Major Players
- Teva Pharmaceuticals
- Sandoz (Novartis)
- Mylan (now part of Viatris)
- Lupin Pharmaceuticals
Market Shares
Generics account for over 85% of sales. Teva and Sandoz lead with approximately 35% combined market share. Brand manufacturers largely exited or reduced presence post-patent expiry.
Market Entry Barriers
High manufacturing quality standards and regulatory approvals favor existing generic manufacturers. Strategic alliances with pharmacies and upstream suppliers influence market positioning.
Forecasted Financial Trajectory
Revenue Projections
Demand stability in chronic conditions and expanding use for short-term treatment sustain revenue levels. However, intensified price competition forecasts a decline in unit prices, constraining growth.
Profitability Outlook
Manufacturers with cost-efficient production and limited dependency on patent-exclusive formulations will experience margins ranging from 10-15%, with potential for margin compression owing to price competition.
Risks and Opportunities
- Risks: Price erosion, regulatory tightening, shifts toward alternative therapies.
- Opportunities: Development of unique formulations (e.g., liquid or injectable) for niche indications, potential orphan drug status, regional expansion, and biosimilars.
Key Takeaways
- Prednisone's market has transitioned primarily to generics, with limited opportunities for branded revenue growth.
- Market size is steady, around $1.2 billion globally, with slow CAGR of about 3.2%, driven by autoimmune and inflammatory disease prevalence.
- Price erosion remains significant, with unit prices cut by half post-expiry of patents.
- Regional variations influence revenue, with North America leading and Asia-Pacific showing higher growth potential.
- Future revenue streams lie in niche formulations, regional market expansion, and regulatory-driven indications.
FAQs
1. How does patent expiration affect prednisone's market?
Patent expiry in 2010 allowed multiple manufacturers to produce generics, increasing competition and reducing prices, which limits branded market share and revenue.
2. What factors influence prednisolone prices worldwide?
Regulatory policies, healthcare reimbursement systems, and manufacturing costs influence prices, with aggressive price negotiations in Europe and U.S. favoring low-cost generics.
3. Are there new formulations or indications for prednisone?
Limited new formulations are in development due to the drug's age. Some manufacturers explore controlled-release versions or injectable forms for specific conditions.
4. What is the outlook for branded prednisone sales?
Branded sales are declining; future growth hinges on orphan indications, specialty formulations, or regional niche markets.
5. How do regulatory changes impact the prednisone market?
Regulatory agencies focus on quality, safety, and patent protections. Stricter rules could delay or restrict new formulations, but they have minimal impact on existing generics.
References
- MarketWatch, "Prednisone Market Size, Share & Trends," 2022.
- IQVIA, "Global Pharmaceutical Market Report," 2022.
- U.S. Food and Drug Administration, "ANDA Approvals," 2022.
- European Medicines Agency, "Drug Marketing Authorizations," 2022.
- GlobalData, "Corticosteroids Market Analysis," 2022.