Last updated: February 17, 2026
Pamelor (Nortriptyline) Overview and Current Market Position
Pamelor, the brand name for nortriptyline hydrochloride, is a tricyclic antidepressant (TCA) primarily used to treat depression. It functions by increasing the levels of certain natural chemicals (neurotransmitters) in the brain. While newer classes of antidepressants like selective serotonin reuptake inhibitors (SSRIs) and serotonin-norepinephrine reuptake inhibitors (SNRIs) have largely supplanted TCAs in first-line treatment due to their generally more favorable side-effect profiles, Pamelor retains a niche in specific patient populations and certain treatment-resistant cases. Its long history of use, established efficacy, and lower cost compared to newer generics contribute to its continued presence in the market.
The global market for antidepressants is substantial, driven by the increasing prevalence of mental health disorders, greater awareness, and reduced stigma. However, the market for older drugs like Pamelor is characterized by significant generic competition. The patent protection for the original Pamelor brand has long expired, leading to the availability of multiple generic versions. This genericization has dramatically reduced pricing power and sales revenue for the branded product, shifting the market dynamics towards volume-based sales of generic nortriptyline.
Key Market Segments for Nortriptyline:
- Depression Treatment: Remains the primary indication, particularly for patients who have not responded to other antidepressant classes or experience side effects from them.
- Neuropathic Pain Management: Off-label use for conditions like diabetic neuropathy and post-herpetic neuralgia.
- Other Psychiatric Disorders: Limited use in conditions like ADHD and panic disorder.
Patent Landscape and Generic Competition
The original patent for nortriptyline hydrochloride, which would have provided market exclusivity for the branded drug Pamelor, expired decades ago. Specifically, the foundational patents for nortriptyline were filed in the late 1950s and early 1960s. This extended period of patent expiry has paved the way for widespread generic manufacturing.
Key aspects of the Pamelor patent and generic landscape:
- Exclusivity Period: The period of market exclusivity granted by patent protection has long passed.
- Generic Entry: Multiple pharmaceutical companies manufacture and market generic nortriptyline hydrochloride.
- Formulations: Generic versions are available in various strengths and dosage forms (e.g., capsules).
- Regulatory Approvals: Generic versions have undergone Abbreviated New Drug Application (ANDA) processes with regulatory bodies like the U.S. Food and Drug Administration (FDA), demonstrating bioequivalence to the branded product.
The absence of patent protection means that Pamelor and its generic equivalents are subject to intense price competition. The primary differentiator for generic nortriptyline is often cost and availability, rather than novel therapeutic benefits or patent-protected innovation.
Financial Performance and Market Valuation
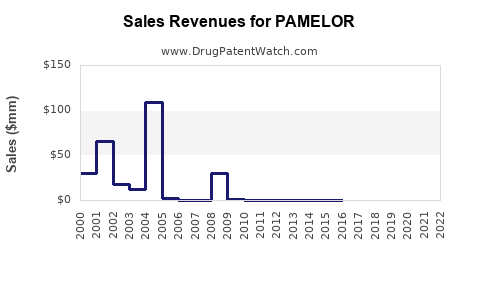
The financial performance of Pamelor as a branded product has been significantly impacted by genericization. Original sales figures for the branded drug are not publicly disaggregated in current financial reporting, as it is now part of a broader generic portfolio for the companies that manufacture it. However, the trajectory for branded TCAs like Pamelor is a well-established pattern: substantial revenue decline post-patent expiry.
Revenue Trends (General TCA Pattern Post-Patent Expiry):
- Peak Revenue: Branded drugs achieve peak sales during their patent-protected period.
- Post-Patent Decline: Upon patent expiry and the entry of generics, branded revenue experiences a sharp and sustained decrease, often exceeding 80-90% within a few years.
- Generic Market Dominance: The market share and revenue shift to generic manufacturers.
Market Valuation Considerations:
- Nortriptyline Market Size: The overall market for nortriptyline (branded and generic) is difficult to precisely quantify due to the fragmented nature of generic sales data. However, it is a mature and relatively stable market, characterized by high volume but low unit prices.
- Competitive Pricing: The average selling price (ASP) for generic nortriptyline is low, driven by intense competition among multiple suppliers.
- Manufacturing Efficiency: Companies with efficient manufacturing processes and strong distribution networks are best positioned to capture market share in the generic space.
While specific financial figures for the original Pamelor brand are historical, the current financial trajectory is dictated by the economics of generic pharmaceuticals. Companies that produce generic nortriptyline generate revenue through high-volume sales at low profit margins per unit.
Clinical Significance and Therapeutic Role
Despite the rise of newer antidepressant classes, nortriptyline retains a specific therapeutic role due to its pharmacological profile and historical efficacy data. Its mechanism of action, inhibiting the reuptake of norepinephrine and serotonin, differs from SSRIs and SNRIs, making it a viable option for patients who do not respond to or tolerate other treatments.
Key Clinical Considerations for Nortriptyline:
- Efficacy in Treatment-Resistant Depression: Studies and clinical experience indicate that nortriptyline can be effective in patients who have failed to respond to multiple other antidepressant trials [1].
- Side Effect Profile: TCAs, including nortriptyline, are associated with a distinct side-effect profile, which can include anticholinergic effects (dry mouth, constipation, urinary retention), sedation, orthostatic hypotension, and cardiac conduction abnormalities. These side effects can limit its use in certain patient populations, such as the elderly or those with cardiovascular disease.
- Therapeutic Drug Monitoring (TDM): Due to its narrow therapeutic index and potential for toxicity, TDM is often recommended to guide dosing and ensure optimal plasma concentrations, especially in older adults or those with comorbidities [2].
- Off-Label Uses: Nortriptyline is also prescribed off-label for chronic pain conditions, particularly neuropathic pain, where it can modulate descending pain pathways [3].
- Drug Interactions: TCAs can interact with a wide range of medications, including other CNS depressants, anticholinergics, and CYP enzyme inhibitors/inducers, requiring careful prescribing [4].
The clinical significance of nortriptyline is not in its novelty but in its established efficacy for specific patient profiles and its availability as a cost-effective treatment option, particularly within the generic market.
Regulatory Landscape and Market Access
The regulatory landscape for Pamelor, now largely represented by generic nortriptyline, is governed by the established approval processes for generic drugs. Regulatory bodies worldwide, such as the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA), have frameworks in place to ensure the safety, efficacy, and quality of generic pharmaceuticals.
Key Regulatory Aspects:
- Abbreviated New Drug Application (ANDA): Generic manufacturers submit ANDAs to regulatory agencies. These applications demonstrate that the generic drug is bioequivalent to the reference listed drug (the branded Pamelor in this context) and meets all quality standards [5].
- Bioequivalence: This is a critical component of generic drug approval, meaning the generic drug delivers the same amount of active ingredient into the bloodstream over the same time period as the branded drug.
- Labeling: Generic drug labeling must be the same as the reference listed drug, except for differences in inactive ingredients.
- Post-Market Surveillance: Like all approved drugs, generic nortriptyline is subject to post-market surveillance to monitor for adverse events and ensure ongoing safety.
- Quality Standards: Manufacturers must adhere to Good Manufacturing Practices (GMPs) to ensure consistent product quality.
Market Access:
- Formulary Inclusion: Generic nortriptyline is widely available on insurance formularies due to its low cost and established clinical utility.
- Prescribing Patterns: Prescribing patterns are influenced by physician familiarity with TCAs, patient response to other agents, and cost considerations.
- International Markets: Nortriptyline is available in many countries, with regulatory approvals and market access varying by jurisdiction. The cost-effectiveness of generic nortriptyline makes it a valuable treatment option in resource-limited settings.
The regulatory pathway for generic nortriptyline is mature. Market access is generally broad, contingent on demonstrating bioequivalence and adherence to manufacturing standards. The financial viability hinges on efficient production and distribution rather than novel regulatory hurdles or market exclusivity.
Competitive Landscape Analysis
The competitive landscape for nortriptyline is characterized by a large number of generic manufacturers. The market is highly fragmented, with many players competing primarily on price and supply chain reliability. The absence of patent protection means that any company with the capability to manufacture generic nortriptyline according to regulatory standards can enter the market.
Key Competitors (Generic Manufacturers):
- Teva Pharmaceutical Industries
- Mylan N.V. (now Viatris)
- Sun Pharmaceutical Industries
- Dr. Reddy's Laboratories
- Apotex Inc.
- Actavis plc (now part of Teva)
This list is not exhaustive, as numerous smaller and regional generic manufacturers also produce nortriptyline.
Competitive Dynamics:
- Price Erosion: The intense competition leads to significant price erosion, making profit margins per unit very low.
- Supply Chain and Distribution: Reliability of supply and efficient distribution networks are critical for success. Shortages of any essential medication can create opportunities for competitors.
- Cost of Goods Sold (COGS): Companies with lower manufacturing costs have a competitive advantage. This includes efficient sourcing of raw materials and optimized production processes.
- Market Share: Market share is typically driven by the ability to offer competitive pricing and maintain consistent supply across a broad customer base, including retail pharmacies, hospitals, and wholesalers.
- Limited Innovation: There is little incentive for significant product innovation (e.g., new formulations, delivery systems) in a market dominated by low-cost generics. The focus remains on cost-efficient production and distribution of existing formulations.
The competitive environment for nortriptyline is that of a mature generic drug market, where price, availability, and manufacturing efficiency are the primary drivers of success, rather than product differentiation or intellectual property.
Future Market Projections and Trends
The future market for nortriptyline is expected to remain stable, characterized by continued generic competition and consistent, albeit low-volume, demand. Significant growth or decline is unlikely unless there are major shifts in clinical practice guidelines or unforeseen regulatory changes.
Projected Market Trends:
- Continued Generic Dominance: Generic nortriptyline will continue to dominate the market. The branded Pamelor product will likely represent a negligible share, if it is still actively marketed as a distinct brand.
- Stable Demand: Demand is projected to be stable, driven by its role as a cost-effective option for certain patient populations with depression and neuropathic pain. There is no expectation of a resurgence in demand comparable to newer drug classes.
- Price Stability (Low): Prices for generic nortriptyline are expected to remain low and relatively stable due to persistent competition. Minor price fluctuations may occur due to supply chain issues or changes in raw material costs, but significant price increases are improbable.
- Focus on Manufacturing Efficiency: Companies will continue to focus on optimizing manufacturing processes and supply chains to maintain profitability in a low-margin environment.
- Potential for Supply Chain Disruptions: As with many generic drugs, the market remains susceptible to supply chain disruptions caused by manufacturing issues, raw material shortages, or geopolitical events. Such disruptions can temporarily impact availability and pricing.
- Niche Therapeutic Role: Nortriptyline will maintain its niche therapeutic role. It is unlikely to be displaced entirely from clinical practice due to its unique pharmacological properties and established efficacy in treatment-resistant cases, despite its side effect profile.
- No Significant R&D Investment: Given the mature generic status, substantial research and development investment in new nortriptyline-based therapies or advanced formulations is not anticipated.
The market trajectory for nortriptyline is one of a mature, essential generic medication. Its future is defined by its established role, cost-effectiveness, and the competitive dynamics inherent in the generic pharmaceutical sector.
Key Takeaways
- Pamelor (nortriptyline hydrochloride) is a genericized tricyclic antidepressant with a long-expired patent.
- The market is dominated by generic manufacturers, leading to intense price competition and low profit margins.
- Nortriptyline retains a niche therapeutic role in treatment-resistant depression and neuropathic pain.
- Regulatory approval for generic versions relies on demonstrating bioequivalence and adherence to GMPs.
- The competitive landscape is fragmented, with success driven by manufacturing efficiency and supply chain reliability.
- Future market projections indicate stable demand and low, consistent pricing, with no significant growth anticipated.
Frequently Asked Questions
-
What is the current patent status of Pamelor (nortriptyline)?
The original patents for nortriptyline hydrochloride, the active ingredient in Pamelor, expired decades ago, allowing for widespread generic manufacturing.
-
How does the generic competition affect the pricing of nortriptyline?
Intense competition among multiple generic manufacturers drives prices down significantly, making nortriptyline a cost-effective treatment option.
-
What are the primary therapeutic uses for nortriptyline today?
Nortriptyline is primarily used for the treatment of depression, particularly in cases of treatment resistance, and off-label for neuropathic pain.
-
Are there any new formulations or delivery methods for nortriptyline being developed?
Due to its generic status and mature market, there is limited ongoing research and development into novel formulations or delivery methods for nortriptyline.
-
What are the main challenges for companies manufacturing generic nortriptyline?
The primary challenges include maintaining profitability amidst intense price competition, ensuring supply chain reliability, and optimizing manufacturing costs to remain competitive.
Citations
[1] Greden, J. F. (2001). The evolution of the treatment of depression. Journal of Clinical Psychiatry, 62(Suppl. 19), 4-10.
[2] Preskorn, S. H. (1997). Use of plasma levels of antidepressants. Journal of Clinical Psychiatry, 58(Suppl. 2), 14-21.
[3] Ong, G. Y., & Krishnan, K. R. (2007). Tricyclic antidepressants in chronic pain management. Pain Management, 3(1), 1-10.
[4] Spina, E., & Pollicino, A. M. (2003). Tricyclic antidepressants: drug interactions. Clinical Pharmacokinetics, 42(11), 907-944.
[5] U.S. Food & Drug Administration. (2020, April 23). Generic Drugs Program. Retrieved from https://www.fda.gov/drugs/generic-drugs/generic-drugs-program