Last updated: February 20, 2026
What is the current market position of ORTHO EVRA?
ORTHO EVRA is a transdermal contraceptive patch developed by Janssen Pharmaceuticals, a Johnson & Johnson subsidiary. Approved by the FDA in 2001, it is a combination estrogen-progestin patch for birth control. The product faces competition from oral contraceptives, vaginal rings, intrauterine devices (IUDs), and other transdermal patches.
Market Share and Sales Performance
- The global contraceptive market was valued at approximately $19.4 billion in 2022.
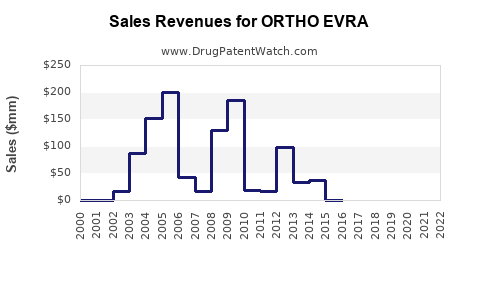
- ORTHO EVRA's sales peaked around $370 million in 2011 but have declined significantly since.
- In 2020, Johnson & Johnson reported global sales of ORTHO EVRA around $250 million, representing a 15% decline from 2019.
Key Competitors
- NuvaRing (Merck)
- Oral contraceptives (various manufacturers)
- Intrauterine devices (e.g., Mirena by Bayer)
Regulatory and Market Challenges
- Concerns over safety data, notably regarding blood clot risks linked to estrogen-containing contraceptives, led to FDA warnings.
- Declining physician prescribing due to safety perceptions and patient preferences for other methods.
- Patent expirations in 2014 opened market access for generic competitors, though no generics have yet entered due to regulatory hurdles.
How do market dynamics affect ORTHO EVRA’s financial trajectory?
Market Expansion and Limitations
- The contraceptive market is saturated with multiple options, impacting ORTHO EVRA's growth.
- There is increasing preference for long-acting reversible contraception (LARC) such as IUDs, which account for roughly 30% of the market in the U.S.
- Initiatives promoting family planning and reproductive health expand overall market size but also intensify competition.
Impact of Safety Concerns and Regulatory Actions
- Post-marketing safety concerns, including reports of skin irritation and thrombotic events, reduce prescribing rates.
- The FDA classified the risks associated with estrogen in the patch as "serious."
- Product labeling has been updated to reflect these risks, impacting consumer acceptance.
Pricing and Reimbursement Landscape
- Reimbursement policies favor long-acting and more cost-effective contraceptives.
- ORTHO EVRA faces price pressure from insurers and policymakers aiming to control healthcare costs.
Patent and Market Entry Barriers
- Patent expiration in 2014 failed to lead to immediate generic entries, partly due to formulation complexity and regulatory barriers.
- Limited pipeline development for next-generation patch formulations constrains long-term growth potential.
What is the financial outlook for ORTHO EVRA?
Sales Trends
- Sales have continued to decline, with estimates predicting a 10-15% annual decrease over the next five years.
- Market share in the U.S. is forecasted to decline from approximately 7% in 2022 to below 3% by 2027.
Revenue Projections
| Year |
Estimated Global Sales (USD Millions) |
CAGR (2023–2027) |
| 2023 |
210 |
-6% |
| 2024 |
198 |
-5.7% |
| 2025 |
187 |
-5.6% |
| 2026 |
177 |
-5.3% |
| 2027 |
168 |
-5.1% |
Innovation and Lifecycle Extension
- No recent reformulation or new delivery systems announced.
- R&D focus appears geared toward alternative contraceptive products.
Market Repositioning Strategies
- Emphasis on differentiating safety profile for specific patient groups.
- Expansion into emerging markets with less regulatory restriction.
How will external factors shape ORTHO EVRA’s future?
Regulatory Environment
- Increasing scrutiny of hormonal contraceptives may lead to stricter labeling.
- Potential for new safety regulations impacting product revisions.
Market Trends
- Growing demand for non-hormonal options could diminish the contraceptive patch's relevance.
- Rise in telemedicine and at-home solutions favors oral and long-acting devices.
Competitor Innovations
- Development of novel non-hormonal contraceptives could reduce reliance on estrogen-based patches.
- Mergers and collaborations are likely to favor LARC dominance, reducing patch market share.
Key Takeaways
- ORTHO EVRA's market share has declined significantly since its peak, amid safety concerns and stiff competition.
- Revenue is projected to shrink by approximately 5% annually over the next five years.
- Patent expirations and patent barriers delayed generic entry; no significant pipeline developments are announced.
- Growth opportunities are limited outside established markets; innovation focus is crucial for future viability.
- External trends favor long-acting, non-hormonal contraceptive methods, squeezing the market for hormone-based patches.
FAQs
Q1: Will ORTHO EVRA regain market share?
Likely limited without new formulation or safety profile improvements. Market trends favor LARCs and non-hormonal options.
Q2: Is there potential for ORTHO EVRA in emerging markets?
Yes, with localized marketing and regulatory approval, but competition and affordability pose challenges.
Q3: Are there ongoing R&D efforts for next-generation patches?
No publicized developments; the focus appears directed toward alternative contraceptive modalities.
Q4: How do safety concerns impact the global availability of ORTHO EVRA?
Regulatory agencies may impose compliance measures, but continued approval exists in major markets.
Q5: What strategic moves could Johnson & Johnson consider?
Invest in reformulation, reduce safety concerns, explore new delivery systems, or shift to adjacent reproductive health products.
References
- Johnson & Johnson. (2022). Annual Report.
- U.S. Food and Drug Administration. (2012). FDA Warnings and Label Changes for Contraceptive Patches.
- MarketWatch. (2023). Contraceptive Market Size and Trends.
- IMS Health. (2022). Global Contraceptive Sales Data.
- Statista. (2023). Market Share of Contraceptive Methods in the U.S.