ONIVYDE Drug Patent Profile
✉ Email this page to a colleague
Which patents cover Onivyde, and when can generic versions of Onivyde launch?
Onivyde is a drug marketed by Ipsen and is included in one NDA. There are seventeen patents protecting this drug.
This drug has one hundred and twenty-three patent family members in twenty-eight countries.
The generic ingredient in ONIVYDE is irinotecan hydrochloride. There are thirty-three drug master file entries for this compound. Sixteen suppliers are listed for this compound. Additional details are available on the irinotecan hydrochloride profile page.
DrugPatentWatch® Litigation and Generic Entry Outlook for Onivyde
A generic version of ONIVYDE was approved as irinotecan hydrochloride by ACTAVIS TOTOWA on February 27th, 2008.
Summary for ONIVYDE
| International Patents: | 123 |
| US Patents: | 17 |
| Applicants: | 1 |
| NDAs: | 1 |
| Finished Product Suppliers / Packagers: | 1 |
| Raw Ingredient (Bulk) Api Vendors: | 94 |
| Clinical Trials: | 35 |
| Patent Applications: | 37 |
| Formulation / Manufacturing: | see details |
| Drug Prices: | Drug price information for ONIVYDE |
| What excipients (inactive ingredients) are in ONIVYDE? | ONIVYDE excipients list |
| DailyMed Link: | ONIVYDE at DailyMed |

Recent Clinical Trials for ONIVYDE
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| National Cheng-Kung University Hospital | Phase 1/Phase 2 |
| Chang Gung Memorial Hospital | Phase 1/Phase 2 |
| National Health Research Institutes, Taiwan | Phase 1/Phase 2 |
Pharmacology for ONIVYDE
| Drug Class | Topoisomerase Inhibitor |
| Mechanism of Action | Topoisomerase Inhibitors |
Anatomical Therapeutic Chemical (ATC) Classes for ONIVYDE
US Patents and Regulatory Information for ONIVYDE
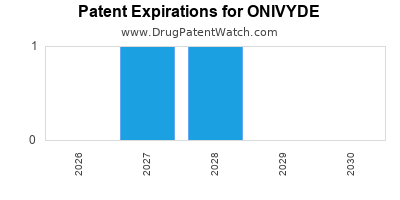
ONIVYDE is protected by twenty US patents and one FDA Regulatory Exclusivity.
Patents protecting ONIVYDE
Stabilizing camptothecin pharmaceutical compositions
Patent Number: ⤷ Try a Trial
Patent Expiration: ⤷ Try a Trial
Liposomes useful for drug delivery
Patent Number: ⤷ Try a Trial
Patent Expiration: ⤷ Try a Trial
Methods for treating pancreatic cancer using combination therapies comprising liposomal irinotecan
Patent Number: ⤷ Try a Trial
Patent Expiration: ⤷ Try a Trial
Patented Use: TREATMENT OF METASTATIC ADENOCARCINOMA OF THE PANCREAS THAT HAS PROGRESSED ON GEMCITABINE-BASED THERAPY, IN COMBINATION WITH 5-FLUOROURACIL AND LEUCOVORIN
Stabilizing camptothecin pharmaceutical compositions
Patent Number: ⤷ Try a Trial
Patent Expiration: ⤷ Try a Trial
Methods for treating pancreatic cancer using combination therapies
Patent Number: ⤷ Try a Trial
Patent Expiration: ⤷ Try a Trial
Patented Use: TREATMENT OF METASTATIC ADENOCARCINOMA OF THE PANCREAS THAT HAS PROGRESSED ON GEMCITABINE-BASED THERAPY, IN COMBINATION WITH 5-FLUOROURACIL AND LEUCOVORIN
Liposomes useful for drug delivery
Patent Number: ⤷ Try a Trial
Patent Expiration: ⤷ Try a Trial
Liposomes useful for drug delivery
Patent Number: ⤷ Try a Trial
Patent Expiration: ⤷ Try a Trial
Liposomes useful for drug delivery
Patent Number: ⤷ Try a Trial
Patent Expiration: ⤷ Try a Trial
Patented Use: TREATMENT OF PANCREATIC CANCER
Liposomes useful for drug delivery
Patent Number: ⤷ Try a Trial
Patent Expiration: ⤷ Try a Trial
Methods for treating pancreatic cancer using combination therapies comprising liposomal irinotecan
Patent Number: ⤷ Try a Trial
Patent Expiration: ⤷ Try a Trial
Patented Use: TREATMENT OF METASTATIC ADENOCARCINOMA OF THE PANCREAS THAT HAS PROGRESSED ON GEMCITABINE-BASED THERAPY, IN COMBINATION WITH 5-FLUOROURACIL AND LEUCOVORIN
Methods for treating pancreatic cancer using combination therapies comprising liposomal irinotecan
Patent Number: ⤷ Try a Trial
Patent Expiration: ⤷ Try a Trial
Patented Use: TREATMENT OF METASTATIC ADENOCARCINOMA OF THE PANCREAS THAT HAS PROGRESSED ON GEMCITABINE-BASED THERAPY, IN COMBINATION WITH 5-FLUOROURACIL AND LEUCOVORIN, IN A PATIENT HOMOZYGOUS FOR THE UGT1A1*28 ALLELE
Methods for treating pancreatic cancer using combination therapies comprising liposomal irinotecan
Patent Number: ⤷ Try a Trial
Patent Expiration: ⤷ Try a Trial
Patented Use: TREATMENT OF PANCREATIC CANCER THAT HAS PROGRESSED ON GEMCITABINE-BASED THERAPY, IN COMBINATION WITH 5-FLUOROURACIL AND LEUCOVORIN
Methods for treating pancreatic cancer using combination therapies comprising liposomal irinotecan
Patent Number: ⤷ Try a Trial
Patent Expiration: ⤷ Try a Trial
Patented Use: TREATMENT OF EXOCRINE PANCREATIC CANCER THAT HAS PROGRESSED ON GEMCITABINE-BASED THERAPY, IN COMBINATION WITH 5-FLUOROURACIL AND LEUCOVORIN
Methods for treating pancreatic cancer using combination therapies comprising liposomal irinotecan
Patent Number: ⤷ Try a Trial
Patent Expiration: ⤷ Try a Trial
Patented Use: TREATMENT OF PANCREATIC CANCER THAT HAS PROGRESSED ON GEMCITABINE-BASED THERAPY, IN COMBINATION WITH 5-FLUOROURACIL AND LEUCOVORIN
Methods for treating pancreatic cancer using combination therapies comprising liposomal irinotecan
Patent Number: ⤷ Try a Trial
Patent Expiration: ⤷ Try a Trial
Patented Use: TREATMENT OF METASTATIC ADENOCARCINOMA OF THE PANCREAS THAT HAS PROGRESSED ON GEMCITABINE-BASED THERAPY, IN COMBINATION WITH 5-FLUOROURACIL AND LEUCOVORIN
Methods for treating pancreatic cancer using combination therapies
Patent Number: ⤷ Try a Trial
Patent Expiration: ⤷ Try a Trial
Patented Use: TREATMENT OF METASTATIC ADENOCARCINOMA OF THE PANCREAS THAT HAS PROGRESSED ON GEMCITABINE-BASED THERAPY, IN COMBINATION WITH 5-FLUOROURACIL AND LEUCOVORIN, IN A PATIENT NOT HOMOZYGOUS FOR THE UGT1A1*28 ALLELE
Methods for treating pancreatic cancer using combination therapies
Patent Number: ⤷ Try a Trial
Patent Expiration: ⤷ Try a Trial
Patented Use: TREATMENT OF METASTATIC ADENOCARCINOMA OF THE PANCREAS THAT HAS PROGRESSED ON GEMCITABINE-BASED THERAPY, IN COMBINATION WITH 5-FLUOROURACIL AND LEUCOVORIN
Liposomes useful for drug delivery
Patent Number: ⤷ Try a Trial
Patent Expiration: ⤷ Try a Trial
Liposomes useful for drug delivery
Patent Number: ⤷ Try a Trial
Patent Expiration: ⤷ Try a Trial
Patented Use: TREATMENT OF METASTATIC ADENOCARCINOMA OF THE PANCREAS THAT HAS PROGRESSED ON GEMCITABINE-BASED THERAPY, IN COMBINATION WITH 5-FLUOROURACIL AND LEUCOVORIN
Liposomes useful for drug delivery
Patent Number: ⤷ Try a Trial
Patent Expiration: ⤷ Try a Trial
FDA Regulatory Exclusivity protecting ONIVYDE
FOR FIRST-LINE TREATMENT OF ADULT PATIENTS WITH METASTATIC PANCREATIC ADENOCARCINOMA
Exclusivity Expiration: ⤷ Try a Trial
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Ipsen | ONIVYDE | irinotecan hydrochloride | INJECTABLE, LIPOSOMAL;INTRAVENOUS | 207793-001 | Oct 22, 2015 | RX | Yes | Yes | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Ipsen | ONIVYDE | irinotecan hydrochloride | INJECTABLE, LIPOSOMAL;INTRAVENOUS | 207793-001 | Oct 22, 2015 | RX | Yes | Yes | ⤷ Try a Trial | ⤷ Try a Trial | Y | Y | ⤷ Try a Trial | ||
| Ipsen | ONIVYDE | irinotecan hydrochloride | INJECTABLE, LIPOSOMAL;INTRAVENOUS | 207793-001 | Oct 22, 2015 | RX | Yes | Yes | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Ipsen | ONIVYDE | irinotecan hydrochloride | INJECTABLE, LIPOSOMAL;INTRAVENOUS | 207793-001 | Oct 22, 2015 | RX | Yes | Yes | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Ipsen | ONIVYDE | irinotecan hydrochloride | INJECTABLE, LIPOSOMAL;INTRAVENOUS | 207793-001 | Oct 22, 2015 | RX | Yes | Yes | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Ipsen | ONIVYDE | irinotecan hydrochloride | INJECTABLE, LIPOSOMAL;INTRAVENOUS | 207793-001 | Oct 22, 2015 | RX | Yes | Yes | ⤷ Try a Trial | ⤷ Try a Trial | Y | Y | ⤷ Try a Trial | ||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
International Patents for ONIVYDE
See the table below for patents covering ONIVYDE around the world.
| Country | Patent Number | Title | Estimated Expiration |
|---|---|---|---|
| China | 107811971 | 用于药物输送的脂质体 (Liposomes useful for drug delivery) | ⤷ Try a Trial |
| Morocco | 42991 | STABILISATION DE COMPOSITIONS PHARMACEUTIQUES DE CAMPTOTHÉCINE | ⤷ Try a Trial |
| Japan | 2018530563 | 安定化カンプトテシン医薬組成物 | ⤷ Try a Trial |
| Russian Federation | 2424792 | ЛИПОСОМЫ, ИСПОЛЬЗУЕМЫЕ ДЛЯ ДОСТАВКИ ЛЕКАРСТВЕННЫХ СРЕДСТВ (LIPOSOMES USED FOR DELIVERY OF MEDICATIONS) | ⤷ Try a Trial |
| Hong Kong | 1258407 | 穩定喜樹碱藥物組合物 (STABILIZING CAMPTOTHECIN PHARMACEUTICAL COMPOSITIONS) | ⤷ Try a Trial |
| China | 104717961 | Methods for treating pancreatic cancer using combination therapies comprising liposomal irinotecan | ⤷ Try a Trial |
| >Country | >Patent Number | >Title | >Estimated Expiration |
Supplementary Protection Certificates for ONIVYDE
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 1746976 | 2017/029 | Ireland | ⤷ Try a Trial | PRODUCT NAME: IRINOTECAN SUCROSOFATE SALT; REGISTRATION NO/DATE: EU/1/16/1130 20161014 |
| 1746976 | LUC00026 | Luxembourg | ⤷ Try a Trial | PRODUCT NAME: IRINOTECAN SUCROSOFATE SALT, SUCH AS IRINOTECAN SUCROSOFATE SALT IN A PEGYLATED LIPOSOME, SUCH AS A LIPOSOME COMPRISING 1,2-DISTEAROYL-SN-PHOSPHATIDYLCHOLINE, CHOLESTEROL AND N-(OMEGA-METHOXY-POLY(ETHYLENE GLYCOL) (MOLECULAR WEIGHT 2000)-OXYCARBONYL)-1,2-DISTEAROYLPHOSPHATIDYL ETHANOLAMINE, E.G. IN THE MOLAR RATIO 3:2:0.015; AUTHORISATION NUMBER AND DATE: EU/1/16/1130 20161018 |
| 1746976 | CA 2017 00030 | Denmark | ⤷ Try a Trial | PRODUCT NAME: IRINOTECAN SUCROSOFATE SALT, SUCH AS IRINOTECAN SUCROSOFATE SALT IN A PEGYLATED LIPOSOME, SUCH AS A LIPOSOME COMPRISING 1,2-DISTEAROYL-SN-PHOSPHATIDYLCHOLINE, CHOLESTEROL AND N-(OMEGA-METHOXY-POLY(ETHYLENE GLYCOL)...; REG. NO/DATE: EU/1 /16/1130 20161018 |
| 1746976 | SPC/GB17/043 | United Kingdom | ⤷ Try a Trial | PRODUCT NAME: IRINOTECAN SUCROSOFATE SALT; REGISTERED: UK EU/1/16/1130 20161018 |
| 1746976 | 2017C/027 | Belgium | ⤷ Try a Trial | PRODUCT NAME: LE SEL DE SUCROSOFATE D'IRINOTECAN, PAR EXEMPLE LE SEL DE SUCROSOFATE D'IRINOTECAN INCORPORE DANS UN LIPOSOME MODIFIE PAR PEGYLATION, TEL QU'UN LIPOSOME COMPRENANT DE LA 1,2-DISTEAROYL-SN- PHOSPHATIDYLCHOLINE, DU CHOLESTEROL ET DU N- (OMEGA-METHOXY POLY (ETHYLENE GLYCOL) (POIDS MOLECULAIRE 2000) -OXYCARBONYL)-1,2-DISEEAROYLPHOSPHATIDYLETHANOLAMINE, PAR EXEMPLE DANS DES RAPPORTS MOLAIRES 3: 2: 0,015; OU COMME DEMANDE AUXILIAIRE, IRINOTECAN; AUTHORISATION NUMBER AND DATE: EU/1/16/1130 20161018 |
| 1746976 | CR 2017 00030 | Denmark | ⤷ Try a Trial | PRODUCT NAME: IRINOTECAN SUCROSOFATE SALT; REG. NO/DATE: EU/1/16/1130 20161018 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Make Better Decisions: Try a trial or see plans & pricing
Drugs may be covered by multiple patents or regulatory protections. All trademarks and applicant names are the property of their respective owners or licensors. Although great care is taken in the proper and correct provision of this service, thinkBiotech LLC does not accept any responsibility for possible consequences of errors or omissions in the provided data. The data presented herein is for information purposes only. There is no warranty that the data contained herein is error free. thinkBiotech performs no independent verification of facts as provided by public sources nor are attempts made to provide legal or investing advice. Any reliance on data provided herein is done solely at the discretion of the user. Users of this service are advised to seek professional advice and independent confirmation before considering acting on any of the provided information. thinkBiotech LLC reserves the right to amend, extend or withdraw any part or all of the offered service without notice.


