Last updated: February 19, 2026
Minocin (minocycline) is a semi-synthetic tetracycline antibiotic with a broad spectrum of activity. Its market trajectory is influenced by its established therapeutic uses, emerging indications, and the competitive landscape of antibiotics and dermatological treatments.
What is the Current Market Size and Projected Growth for Minocin?
The global market for minocycline, encompassing both its branded and generic forms, is substantial and expected to exhibit moderate growth. The primary drivers for this growth are the continued use of minocycline in treating acne vulgaris and its established efficacy in managing bacterial infections. Emerging research into its anti-inflammatory and neuroprotective properties may present future growth opportunities, though clinical validation and regulatory approval are prerequisites.
According to market research reports, the global antibiotics market, of which minocycline is a segment, is projected to grow at a compound annual growth rate (CAGR) of approximately 3-5% over the next five to seven years. Specific figures for minocycline alone are less granular but align with this broader trend, largely driven by its dermatological applications. The market is also influenced by the increasing prevalence of skin conditions and the demand for effective, cost-efficient treatments.
Key Market Segments for Minocycline
- Acne Vulgaris Treatment: This remains the largest and most stable market segment for minocycline. Its anti-inflammatory properties contribute to its effectiveness in managing moderate to severe acne.
- Bacterial Infections: Minocycline is prescribed for a range of bacterial infections, including respiratory tract infections, urinary tract infections, and skin and soft tissue infections.
- Other Indications: Emerging research explores minocycline's potential in treating conditions such as rheumatoid arthritis, rosacea, and certain neurological disorders. However, these are currently niche or investigational uses.
What are the Key Therapeutic Indications and Their Market Significance?
Minocycline's market significance is rooted in its well-established therapeutic profile. Its broad-spectrum activity against Gram-positive and Gram-negative bacteria makes it a versatile option for treating various infections.
Primary Therapeutic Indications
- Acne Vulgaris: Minocycline is a widely prescribed antibiotic for the treatment of inflammatory acne lesions. It targets Propionibacterium acnes (now Cutibacterium acnes) and exerts anti-inflammatory effects that reduce redness and swelling. The sustained demand for acne treatments, driven by aesthetic concerns and the prevalence of the condition, underpins a significant portion of minocycline sales.
- Respiratory Tract Infections: It is used to treat community-acquired pneumonia, bronchitis, and sinusitis caused by susceptible microorganisms.
- Urinary Tract Infections: Minocycline can be effective against uncomplicated urinary tract infections, particularly when other agents are contraindicated.
- Skin and Soft Tissue Infections: This includes impetigo, folliculitis, and other bacterial skin infections.
- Other Infections: It is also indicated for Lyme disease, Rocky Mountain spotted fever, and other tick-borne illnesses.
Emerging and Investigational Indications
Research has investigated minocycline for its potential benefits beyond antibacterial action. These include:
- Anti-inflammatory and Neuroprotective Effects: Studies have explored its use in rheumatoid arthritis, multiple sclerosis, Parkinson's disease, and stroke. These applications are largely in the research or early clinical trial phases, and their market impact is contingent on successful clinical development and regulatory approval.
- Rosacea: Minocycline has demonstrated efficacy in reducing the inflammatory papules and pustules associated with rosacea.
The market share attributed to each indication fluctuates based on treatment guidelines, physician prescribing habits, and the availability of alternative therapies.
What is the Competitive Landscape for Minocin?
Minocycline operates within a highly competitive pharmaceutical market, facing competition from other tetracycline derivatives, alternative antibiotic classes, and non-antibiotic treatments for its primary indications.
Competition in Antibiotics
Within the tetracycline class, other commonly prescribed drugs include doxycycline and tetracycline itself. Doxycycline, in particular, is a strong competitor due to its similar efficacy, pharmacokinetic profile, and a broad range of approved indications. Generic availability of doxycycline and other tetracyclines often leads to price competition.
Beyond tetracyclines, minocycline competes with:
- Macrolides: Azithromycin, clarithromycin, and erythromycin are frequently used for respiratory tract infections and are alternatives for certain skin infections.
- Fluoroquinolones: Ciprofloxacin and levofloxacin are potent antibiotics used for more severe or resistant infections, although concerns regarding side effects limit their broad use.
- Other Antibiotic Classes: Penicillins, cephalosporins, and sulfonamides are also utilized depending on the specific infection and suspected pathogens.
Competition in Acne Treatment
The acne market is intensely competitive, with numerous therapeutic options. Minocycline competes with:
- Topical Treatments: Retinoids (tretinoin, adapalene), benzoyl peroxide, topical antibiotics (clindamycin, erythromycin), and azelaic acid are first-line or adjunctive therapies.
- Oral Antibiotics: Doxycycline and erythromycin are also used for acne.
- Hormonal Therapies: Oral contraceptives and spironolactone are options for female patients.
- Isotretinoin: This potent oral retinoid is highly effective for severe, recalcitrant acne but carries significant side effect profiles and regulatory oversight.
- Biologics and Immunomodulators: While less common for acne, research into these areas may lead to future competition.
Genericization and Pricing Pressure
The patent expiration of branded minocycline products has led to widespread genericization. This has significantly increased price competition, driving down revenue for individual product lines but expanding market access. The market is now largely dominated by generic manufacturers, with pricing determined by manufacturing costs, supply chain efficiency, and market demand.
What is the Intellectual Property (IP) Status and Patent Expiration Profile?
The intellectual property landscape for minocycline is mature. The original patents covering the compound and its basic uses have long expired.
Key Patent Expirations
- Composition of Matter Patents: These foundational patents have expired decades ago.
- Method of Use Patents: Patents covering specific therapeutic applications, such as the treatment of acne or rheumatoid arthritis, have also largely expired or are nearing expiration.
- Formulation Patents: While some patents may exist for specific novel formulations (e.g., extended-release or topical formulations), these are generally limited in scope and duration. For instance, extended-release formulations of minocycline (e.g., Solodyn, which was marketed by Medicis Pharmaceutical, now part of Bausch Health) had their own patent protection that has since expired.
The absence of robust, broad-spectrum patent protection for minocycline means that the market is primarily driven by generic competition. Manufacturers may seek to differentiate through advanced formulations, improved delivery systems, or by exploring new therapeutic indications, though the latter requires significant R&D investment and new patent applications for novel uses, which are difficult to secure for an old drug.
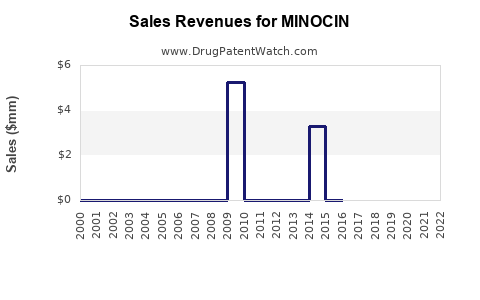
What is the Financial Performance and Revenue Generation of Minocin Products?
The financial performance of minocycline products is characterized by the dominance of generic sales and price-based competition. Branded minocycline products, such as Minocin (formerly marketed by different entities), have seen significant revenue declines post-genericization.
Revenue Trends
- Branded Product Decline: For branded minocycline, revenues have dramatically decreased from their peak. The market share has shifted to generic manufacturers who can offer the drug at significantly lower prices.
- Generic Market Growth: The overall minocycline market, driven by generic volume, remains stable or shows modest growth, reflecting its continued utility in established indications. Revenue in the generic space is fragmented across numerous manufacturers.
- Pricing: Generic minocycline is priced competitively. The cost per dose is significantly lower than historical branded prices, influencing overall market revenue. For example, the cost of a 30-day supply of generic minocycline 100mg capsules can range from approximately $10 to $30 USD, depending on the pharmacy, insurance, and region.
Key Manufacturers and Market Share
The market for minocycline is populated by numerous generic pharmaceutical companies. Major generic manufacturers with significant market presence include, but are not limited to:
- Teva Pharmaceuticals
- Dr. Reddy's Laboratories
- Mylan N.V. (now part of Viatris)
- Aurobindo Pharma
- Sun Pharmaceutical Industries
These companies compete on manufacturing efficiency, distribution networks, and pricing strategies. Market share is fluid and often determined by bidding processes for formulary placement and pharmacy benefit manager (PBM) contracts.
What are the Regulatory Considerations and Market Access Factors?
Regulatory approval and market access are critical for minocycline, particularly concerning its established indications. Emerging uses face a more complex regulatory pathway.
Regulatory Status
Minocycline is approved by major regulatory agencies worldwide, including the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA), for its indicated uses.
- FDA Approvals: The FDA has approved minocycline for the treatment of acne vulgaris and other bacterial infections. The availability of generic minocycline means that multiple manufacturers market FDA-approved versions.
- EMA Approvals: Similar approvals exist in the European Union.
Market Access and Reimbursement
- Insurance Coverage: Minocycline is generally covered by most health insurance plans, especially for its FDA-approved indications like acne. Reimbursement levels for generic minocycline are typically lower than for branded drugs, reflecting the cost-effectiveness of generic medications.
- Payer Policies: Pharmacy benefit managers (PBMs) and insurance companies often place generic minocycline on their formularies, sometimes with preferred tier status due to its low cost. Prior authorization may be required for certain indications or dosages in some plans, but this is less common for established generic antibiotics.
- Off-Label Use: While physicians may prescribe minocycline off-label for investigational indications, such uses are typically not covered by insurance and are less frequent unless supported by strong clinical evidence and emerging guidelines.
Post-Marketing Surveillance and Safety
Like all pharmaceuticals, minocycline is subject to post-marketing surveillance. Concerns regarding side effects, such as dizziness, nausea, and potential for tooth discoloration in developing fetuses or children, are monitored. The risk of developing serious side effects, including hypersensitivity reactions and drug-induced lupus, contributes to prescribing guidelines and contraindications.
Key Takeaways
- Minocycline's market is characterized by a mature patent landscape, leading to extensive genericization and intense price competition.
- Acne vulgaris treatment remains the primary market driver, supported by the drug's anti-inflammatory and antibacterial properties.
- The competitive environment includes other tetracyclines, alternative antibiotic classes, and diverse acne treatment modalities, intensifying pricing pressure.
- Revenue generation is predominantly from generic sales, with market share fragmented among numerous global manufacturers.
- Regulatory approval is established for key indications, facilitating broad market access and insurance coverage, primarily for generic versions.
Frequently Asked Questions
-
What are the main reasons for the shift from branded to generic minocycline?
The primary reason is the expiration of patents covering the original minocycline compounds and their initial therapeutic uses. This allows other manufacturers to produce and sell generic versions, which are typically priced much lower than branded products.
-
Are there any new therapeutic indications for minocycline currently in advanced clinical trials?
While minocycline is being researched for various conditions beyond bacterial infections, such as neurodegenerative diseases and inflammatory disorders, these are generally in earlier stages of development or have shown mixed results. Significant advances to commercialization for new indications are not currently widely reported as being in late-stage clinical trials.
-
How does minocycline's cost compare to other commonly used antibiotics for acne?
Generic minocycline is generally one of the more cost-effective oral antibiotics for acne when compared to newer or more broadly used agents. Its price point is competitive with other tetracyclines like doxycycline and often lower than macrolides.
-
What are the major safety concerns associated with minocycline that might impact its market use?
Key safety concerns include gastrointestinal upset, dizziness, and photosensitivity. More serious, though less common, adverse effects include drug-induced lupus, hypersensitivity syndromes, and neurological effects. These are managed through prescribing guidelines and patient monitoring.
-
Which companies are the leading manufacturers of generic minocycline globally?
Leading generic manufacturers of minocycline include Teva Pharmaceuticals, Dr. Reddy's Laboratories, Viatris (formerly Mylan), Aurobindo Pharma, and Sun Pharmaceutical Industries, among others that compete in the global generics market.
Citations
[1] U.S. Food and Drug Administration. (n.d.). Drug Approval Packages. Retrieved from [FDA website - specific drug approval databases can be searched here].
[2] European Medicines Agency. (n.d.). European Public Assessment Reports (EPARs). Retrieved from [EMA website - specific drug assessment reports can be searched here].
[3] Market Research Reports (General knowledge of market trends in antibiotics and dermatology). Specific market research firms like IQVIA, Grand View Research, and Mordor Intelligence publish reports on these sectors. (No specific URL as these are proprietary reports).
[4] Generic drug pricing data (Obtained from pharmacy benefit manager databases and online pharmacy price aggregators, reflecting current market conditions). (No specific URL as these are dynamic and often subscription-based).