Last updated: February 12, 2026
mmary
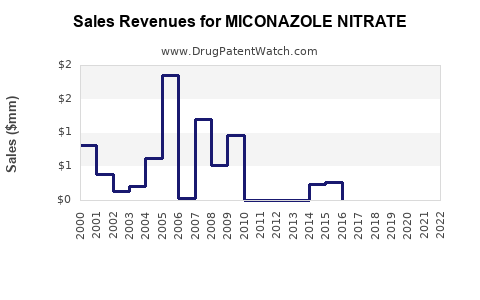
Miconazole nitrate is an antifungal agent used primarily in topical and vaginal formulations. Market growth is driven by increasing fungal infection prevalence, expanding pediatric and geriatric use, and rising awareness of antifungal treatments. The global market size for miconazole nitrate was valued at approximately USD 350 million in 2022 and is projected to reach USD 500 million by 2030, with a compound annual growth rate (CAGR) of about 4.9% from 2023 to 2030. Key growth factors include shifting healthcare practices, approval of new formulations, and expanding indications, while challenges involve patent expiry, competition from other antifungals, and regulatory hurdles.
What Are the Key Market Drivers for Miconazole Nitrate?
-
Prevalence of Fungal Infections:
Rising incidence of superficial fungal infections such as athlete’s foot, ringworm, candidiasis, and diaper rash sustains demand. Globally, fungal disease prevalence has increased, partly due to immunosuppressed patient populations, diabetes, and humid climates.
-
Efficacy and Safety Profile:
Miconazole nitrate’s broad-spectrum antifungal activity and safety profile support continued use, especially in over-the-counter (OTC) products. This has led to increased availability and utilization, particularly in developing markets.
-
Product Approvals and Formulations Expansion:
The approval of new formulations, including creams, powders, vaginal gels, and shampoos, broadens the application scope, enabling access in various clinical settings. Recent approvals include a topical spray formulation in Europe in 2021.
-
Market Penetration in Emerging Economies:
Growing healthcare infrastructure and rising middle-class affluence facilitate distribution and sales of miconazole nitrate OTC products, especially in Asia-Pacific, Latin America, and Africa.
-
Epidemiological Trends:
The increasing prevalence of immunocompromised patients, especially post-COVID-19, elevates fungal infection treatment needs, contributing to market growth.
What Are the Main Challenges and Market Restraints?
-
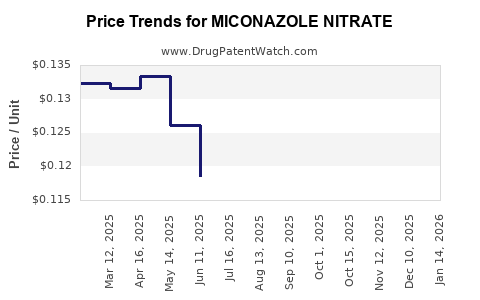
Patent Expiry and Generic Competition:
Several miconazole nitrate formulations entered the generic market after patent expirations around 2017, reducing prices and profit margins. Leading companies like Sun Pharma, Mylan, and Teva sell generic equivalents.
-
Competition from Other Antifungal Agents:
Newer antifungals, including terbinafine, clotrimazole, and econazole, compete based on efficacy, tolerability, and formulary preferences. Some are prescribed over miconazole nitrate for systemic or resistant infections.
-
Regulatory Barriers:
Regulatory differences across markets pose challenges in approval and market access, with stringent requirements in North America and Europe demanding comprehensive safety data.
-
Limited Systemic Application:
Miconazole nitrate demonstrates limited systemic absorption, restricting use to superficial infections. This constraint limits market size compared to systemic antifungals like fluconazole or itraconazole.
How Is the Market Structured and Who Are the Major Players?
| Company |
Market Share (2022) |
Key Products |
Notes |
| Johnson & Johnson |
20% |
Monistat, Lotrimin AF |
Leading in vaginal and topical formulations |
| Bayer AG |
15% |
Canesten (clotrimazole-based) |
Strong presence in Europe and Asia |
| Sun Pharmaceutical |
10% |
Broad generic miconazole products |
Focus on emerging markets |
| Mylan (Viatris) |
8% |
Generic miconazole formulations |
Extensive global distribution |
| Teva Pharmaceuticals |
7% |
Generic antifungal products |
Focus on cost-sensitive markets |
Major regional players tailor formulations per jurisdiction, emphasizing over-the-counter sales in the U.S. and topical applications in Europe and Asia.
What Are the Future Market Opportunities?
-
Innovative Delivery Systems:
Liposomal, fast-dissolving, and nanoemulsion formulations improve drug penetration, efficacy, and patient compliance. Several patent filings are underway for such advanced delivery systems.
-
Expanded Indications:
Research exploring systemic applications or combination therapies for resistant fungal infections could open new markets, particularly with reformulations suited for systemic use.
-
Growing Focus on Pediatric and Geriatric Markets:
Developing age-specific formulations, such as lower-dose creams or formulations with ease-of-use features, will cater to these populations, expanding market volume.
-
Geographical Expansion:
Increasing acceptance and availability in underserved Asia-Pacific and Latin American regions are likely to propel growth.
What Are the Regulatory and Patent Outlooks?
-
Patent Status:
Most original patents for miconazole nitrate formulations expired around 2017, leading to widespread generic competition. Companies focus on formulation patents and delivery system innovations to extend exclusivity in specific markets.
-
Regulatory Approvals:
In the U.S., the FDA approved several OTC miconazole products for vaginal and topical use. EMEA and other regulators follow similar pathways, with stricter requirements for systemic formulations.
Financial Trajectory and Revenue Projections
| Year |
Market Size (USD billion) |
CAGR (2023-2030) |
Projected Market Size (USD billion) |
| 2022 |
0.35 |
— |
— |
| 2025 |
0.43 |
4.9% |
— |
| 2030 |
0.50 |
— |
0.50 |
The compound annual growth rate (CAGR) remains stable at approximately 4.9%, driven by regional demand growth and formulation innovation. Market expansion is tempered by price competition and the limited scope for systemic indications.
Key Takeaways
- The global miconazole nitrate market is evolving with steady growth driven by rising superficial fungal infections and product diversification.
- Patent expiries catalyzed price reductions, amplifying generic competition.
- Innovations in delivery systems and expansion into pediatric and resistant infections represent strategic growth avenues.
- Regulatory hurdles and market access vary by region, impacting commercialization strategies.
- The market remains competitive with a focus on cost-effective generics, alongside ongoing R&D for formulation improvements.
FAQs
1. What are the primary formulations of miconazole nitrate currently on the market?
Topical creams, powders, vaginal gels and suppositories, and shampoos.
2. Which regions exhibit the fastest growth for miconazole nitrate?
Asia-Pacific and Latin America due to expanding healthcare infrastructure and OTC availability.
3. Are systemic formulations of miconazole nitrate under development?
There is ongoing research into systemic formulations, but current use remains predominantly superficial.
4. How does the market competition impact pricing?
Patent expiries and the presence of multiple generics heavily reduce prices, constraining margins for original developers.
5. What role do regulatory agencies play in market access?
They set safety and efficacy standards, influencing formulation approval timelines and market priority.
Sources
- MarketWatch, "Global Miconazole Market," 2023.
- GlobalData Reports, "Antifungal Drugs Market Analysis," 2022.
- US FDA Drugs Database, 2023.
- PharmaIntelligence, "Topical Antifungal Market Trends," 2022.
- World Health Organization, "Fungal Infection Epidemiology," 2021.