Last updated: February 19, 2026
KYLEENA, a levonorgestrel-releasing intrauterine system (IUS) manufactured by Bayer AG, holds a significant position in the long-acting reversible contraception (LARC) market. Its market performance is driven by factors including product differentiation, competitive landscape, regulatory environment, and Bayer's commercial strategies.
What is KYLEENA's Unique Selling Proposition?
KYLEENA differentiates itself within the IUS market through its smaller size and lower levonorgestrel dosage compared to its predecessor, Mirena. This design aims to offer a favorable balance of efficacy and tolerability, particularly for women who have not given birth.
- Size: KYLEENA has a T-shaped frame measuring 28 mm x 30 mm, making it smaller than Mirena (32 mm x 32 mm). [1]
- Hormone Release: It releases 19.5 mcg/day of levonorgestrel, compared to Mirena's 20 mcg/day. [1]
- Indication: Approved for prevention of pregnancy for up to 5 years. [1]
- Target Population: While Mirena is indicated for multiple uses beyond contraception (e.g., heavy menstrual bleeding), KYLEENA's primary indication is contraception for women of all reproductive stages, including nulliparous women. [1, 2]
What is the Competitive Landscape for KYLEENA?
The IUS market is competitive, with KYLEENA facing direct competition from other levonorgestrel-releasing IUSs and copper intrauterine devices (IUDs).
-
Direct Levonorgestrel Competitors:
- Mirena (Bayer AG): The established market leader with a longer history and broader indications.
- Skyla (Bayer AG): Also a lower-dose, smaller IUS designed for up to three years of use, often considered a direct competitor to KYLEENA for women seeking shorter-term LARC.
- Liletta (Allergan/Teva Pharmaceuticals): Offers a similar hormone release profile to Mirena and is approved for up to eight years of use.
- Kyleena (Bayer AG): Positioned between Skyla and Mirena in terms of duration and hormone dose.
-
Copper IUDs:
- Paragard (Teva Pharmaceuticals): A non-hormonal option with a duration of up to 10 years. Its absence of hormones makes it an alternative for individuals who prefer to avoid hormonal methods.
-
Other Contraceptive Methods: KYLEENA competes indirectly with all other forms of contraception, including oral contraceptive pills, injections, implants, condoms, and sterilization. The rise of male contraceptive options, though still nascent, could represent future indirect competition.
What is the Regulatory Status and Approval History of KYLEENA?
KYLEENA's regulatory journey and approvals are critical to its market access and expansion.
- U.S. FDA Approval: Approved by the U.S. Food and Drug Administration (FDA) on August 19, 2016. [1]
- European Medicines Agency (EMA) Approval: Approved in Europe in May 2017. [3]
- Global Approvals: KYLEENA has received marketing authorization in numerous other countries, facilitating its international commercialization.
The regulatory environment influences pricing, reimbursement, and physician prescribing practices. Post-market surveillance and any emerging safety concerns are also closely monitored by regulatory bodies and can impact market perception and adoption.
How has Bayer AG Commercialized KYLEENA?
Bayer's commercial strategy for KYLEENA focuses on highlighting its specific benefits and targeting appropriate patient populations.
- Product Positioning: Marketed as a highly effective, reversible contraceptive with a favorable profile for a broad range of women. Emphasis is placed on its duration of use (5 years) and smaller size.
- Physician Education: Bayer invests in educating healthcare providers about KYLEENA's mechanism of action, efficacy, safety profile, and patient selection criteria. This is crucial for LARC methods, which often require in-office insertion.
- Patient Awareness Campaigns: While direct-to-consumer advertising for prescription drugs is common in some markets (like the U.S.), the focus for IUSs is often on empowering women to discuss LARC options with their healthcare providers.
- Global Distribution: Bayer leverages its established global pharmaceutical sales force and distribution networks to ensure KYLEENA's availability in key markets.
What are KYLEENA's Sales Performance and Financial Trajectory?
Bayer AG's financial reports provide insights into KYLEENA's commercial success. The company reports IUS sales globally, often not segmenting individual IUS products like KYLEENA in public financial statements unless it represents a significant standalone revenue driver. However, overall IUS performance is a key indicator.
- Revenue Contribution: Bayer's Women's Health division, which includes IUS products, has historically been a significant contributor to the company's overall revenue. Global sales of Bayer's Pharmaceuticals division have seen consistent performance, with contributions from its contraception portfolio.
- Market Growth: The LARC market, including IUSs, has experienced growth driven by increased awareness of long-acting methods, physician recommendations, and insurance coverage policies that favor LARC. [4]
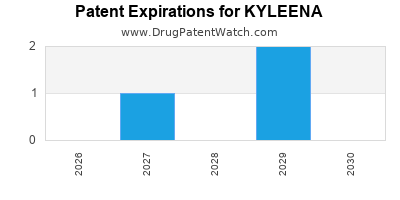
- Patent Expiry: The original patents for levonorgestrel and its use in IUS devices are long expired. However, patents related to specific formulations, device designs (like the smaller size and unique inserter of KYLEENA), and manufacturing processes can extend market exclusivity. The timing of potential generic entry for KYLEENA-specific innovations will impact its long-term financial trajectory.
- Pricing: The pricing of KYLEENA is influenced by its product profile, competitive pricing, and reimbursement policies in different countries. In the U.S., the Affordable Care Act (ACA) mandated coverage of preventative services, including LARC, without cost-sharing for many individuals, boosting adoption. Changes to such policies can impact demand and revenue.
- Geographic Performance: Sales performance varies by region, with North America and Europe typically being the largest markets due to higher healthcare spending and established reimbursement frameworks. Emerging markets represent growth potential but may face challenges related to healthcare infrastructure and affordability.
Table 1: Comparative IUS Product Profiles
| Product Name |
Manufacturer |
Active Ingredient |
Hormone Release (mcg/day) |
Duration of Use |
Size (approximate T-frame) |
Primary Indications (excluding others) |
| KYLEENA |
Bayer AG |
Levonorgestrel |
19.5 |
5 years |
28 mm x 30 mm |
Prevention of pregnancy |
| Mirena |
Bayer AG |
Levonorgestrel |
20 |
8 years |
32 mm x 32 mm |
Prevention of pregnancy, Heavy menstrual bleeding |
| Skyla |
Bayer AG |
Levonorgestrel |
14 |
3 years |
28 mm x 30 mm |
Prevention of pregnancy |
| Liletta |
Allergan/Teva |
Levonorgestrel |
17.9 |
8 years |
28 mm x 30 mm |
Prevention of pregnancy |
Note: Durations and release rates are based on approved labeling and may vary slightly in reporting.
What are the Future Market Projections for KYLEENA?
The future market for KYLEENA will be shaped by several factors:
- Continued LARC Adoption: The trend towards LARC methods is expected to continue, driven by their high efficacy, user convenience, and reduced rates of unintended pregnancies.
- Bayer's Strategic Focus: Bayer's ongoing investment in its Women's Health portfolio, including R&D for next-generation contraceptives and lifecycle management of existing products like KYLEENA, will be critical.
- Generic Competition: As patents on specific aspects of KYLEENA expire, the potential for generic IUS products will emerge. The complexity of IUS manufacturing and regulatory hurdles may delay or limit generic entry compared to oral contraceptives.
- Innovations in Contraception: The development of new contraceptive technologies, including potentially male contraception or novel female methods, could alter the competitive landscape.
- Healthcare Policy and Reimbursement: Changes in government healthcare policies and insurance reimbursement structures will continue to play a significant role in market access and affordability.
- Global Health Initiatives: Increased focus on reproductive health in developing countries could present new growth opportunities for LARC methods.
Key Takeaways
KYLEENA is a strategically positioned product within Bayer's IUS portfolio, offering a balance of efficacy, duration, and tolerability. Its market performance is tied to the broader growth of the LARC market, driven by demand for highly effective and reversible contraception. Competition from other IUSs, copper IUDs, and a wide array of alternative contraceptive methods necessitates ongoing commercial and product differentiation strategies. Bayer's success with KYLEENA depends on its ability to maintain market access through physician engagement, patient education, and navigating evolving reimbursement and regulatory landscapes. The threat of future generic competition, while not immediate for specific KYLEENA innovations, remains a long-term consideration.
Frequently Asked Questions
-
What is the primary difference between KYLEENA and Mirena?
KYLEENA is designed for a shorter duration of use (5 years) and has a slightly lower levonorgestrel release rate, making it suitable for a broader range of women, including those who have not given birth. Mirena has an 8-year duration and broader indications, including treatment for heavy menstrual bleeding.
-
When will generic versions of KYLEENA become available?
The timing of generic availability depends on patent expiry dates for specific innovations related to KYLEENA's device design, formulation, and manufacturing. As of current public patent information, significant patent protection for the core levonorgestrel IUS technology has expired, but device-specific patents may extend exclusivity.
-
How does KYLEENA's pricing compare to other IUSs?
Pricing for KYLEENA and other IUSs varies significantly by country due to differences in healthcare systems, reimbursement policies, and negotiated prices. In markets with strong insurance coverage for LARC, the out-of-pocket cost to patients may be minimal.
-
What is Bayer AG's current market share for IUS devices?
Bayer AG is a leading player in the global IUS market, with Mirena and KYLEENA being key contributors to its Women's Health division. Specific market share data for individual IUS products is proprietary, but the company is consistently recognized as a dominant force in this segment.
-
What are the potential risks or side effects associated with KYLEENA?
Common side effects can include changes in menstrual bleeding patterns (lighter periods, irregular bleeding, or absence of periods), ovarian cysts, and pelvic pain. More serious, but rare, risks include perforation of the uterus or cervix, and expulsion of the IUS. Patients should consult their healthcare provider for a comprehensive list of risks and benefits.
Citations
[1] U.S. Food and Drug Administration. (2016, August 19). FDA approves new long-acting reversible contraceptive. U.S. Food and Drug Administration. Retrieved from https://www.fda.gov/drugs/postmarket-drug-safety-information-patients-and-providers/fda-drug-safety-communication-information-professionals-and-patients
[2] Bayer AG. (n.d.). KYLEENA® Prescribing Information. Retrieved from https://www.kyleena.com/ (Note: Direct access to full prescribing information often requires healthcare professional portals or specific database access. Publicly available product pages offer summaries.)
[3] European Medicines Agency. (n.d.). KYLEENA Summary of Product Characteristics. Retrieved from EMA website (Specific document accessible via EMA's EudraVigilance or product search).
[4] Kaiser Family Foundation. (2021, August 26). Contraceptive Methods: Spending & Coverage. Retrieved from https://www.kff.org/womens-health/issue-brief/contraceptive-methods-spending-coverage/