Last updated: February 19, 2026
Keppra, the brand name for levetiracetam, is an antiepileptic drug (AED) developed by UCB Pharma. It is primarily used to treat partial-onset seizures, myoclonic seizures, and generalized tonic-clonic seizures in patients with epilepsy. The drug's market performance has been shaped by its patent exclusivity, the emergence of generic competition, and its established clinical utility.
What is the Current Market Status of Keppra?
The global market for levetiracetam is characterized by a mature product lifecycle, driven by its long-standing presence and the availability of affordable generic alternatives. The original patent protection for Keppra has expired in major markets, leading to significant price erosion due to generic competition.
- Market Size: While specific real-time global market size figures for levetiracetam as a branded product are increasingly difficult to isolate due to generic fragmentation, the overall antiepileptic drug market, which levetiracetam contributes to, remains substantial. In 2022, the global epilepsy drugs market was valued at approximately $18.2 billion, with projections to reach $22.3 billion by 2030, growing at a CAGR of 2.6% from 2023 to 2030 [1]. Keppra, in its branded form, would have captured a significant portion of this market during its patent-protected period.
- Key Geographies: North America and Europe historically represented the largest markets for Keppra due to high epilepsy prevalence and established healthcare systems. The Asia-Pacific region is also a significant market, with growing demand for AEDs.
- Competitive Landscape: The market is now dominated by generic manufacturers offering levetiracetam at substantially lower prices. Key generic players include Teva Pharmaceuticals, Mylan (now Viatris), Sun Pharmaceutical Industries, and Cipla. Branded Keppra's market share has consequently declined significantly.
What Was Keppra's Patent and Exclusivity Timeline?
The patent history of Keppra is critical to understanding its market trajectory. UCB Pharma secured patent protection that allowed for a period of market exclusivity, during which it was the sole provider of levetiracetam.
- Original Patent Filing: The foundational patents for levetiracetam were filed in the early 1990s.
- US Patent Expiration: The primary US patent for levetiracetam expired in 2008. Following this, generic manufacturers began launching their versions of the drug.
- EU Patent Expiration: In Europe, patent expiries varied by country, but generally occurred in the late 2000s to early 2010s.
- Supplementary Protection Certificates (SPCs): UCB Pharma utilized SPCs in Europe to extend patent-like protection for a period of up to five years beyond the original patent expiry, aiming to compensate for patent term lost during the regulatory approval process. These extensions also expired, allowing for broader generic entry.
- Pediatric Exclusivity: In the US, UCB Pharma received six months of pediatric exclusivity in 2004 for conducting studies in the pediatric population. This extended its market exclusivity until early 2005.
- Patent Litigation: UCB Pharma engaged in numerous patent litigation cases to defend its exclusivity against generic challenges. These litigations often focused on secondary patents covering manufacturing processes, specific formulations, or new uses, but ultimately, the core patents expired, paving the way for generics.
How Did Generic Entry Impact Keppra's Revenue?
The introduction of generic levetiracetam had a profound and immediate impact on UCB Pharma's revenue derived from Keppra. This is a predictable pattern for blockbuster drugs once patent protection lapses.
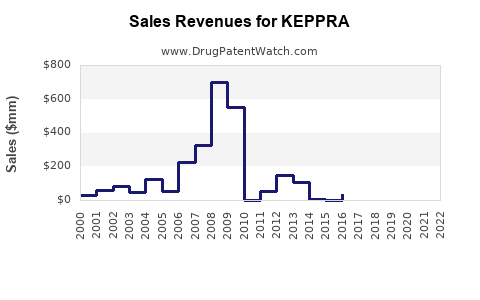
- Revenue Peak: Keppra (levetiracetam) was a significant revenue driver for UCB Pharma. In 2008, the year of its US patent expiry, Keppra sales reached approximately €1.1 billion [2]. By 2010, global sales of Keppra peaked at over €1.7 billion [3].
- Post-Genericization Decline: Following the widespread availability of generics in major markets from 2008 onwards, UCB Pharma experienced a sharp decline in Keppra sales.
- By 2012, Keppra sales had fallen to approximately €1.0 billion [3].
- By 2015, sales had further decreased to around €730 million [3].
- In 2022, UCB reported that its epilepsy franchise, which was heavily reliant on Keppra historically, generated €1.4 billion in revenue, with newer products like Briviact playing a more significant role [4]. Branded Keppra's direct contribution to this figure is now a fraction of its former self.
- Price Erosion: The entry of multiple generic manufacturers led to intense price competition. Generic levetiracetam is typically available at a fraction of the branded Keppra price, often 80-90% lower, depending on the market and volume.
What are the Current Clinical and Commercial Aspects of Levetiracetam?
Despite the revenue shift from branded to generic, levetiracetam remains a widely prescribed medication due to its efficacy, tolerability, and established safety profile.
- Therapeutic Profile: Levetiracetam is recognized for its broad-spectrum antiepileptic activity. Its mechanism of action is not fully understood but is believed to involve binding to the synaptic vesicle protein 2A (SV2A), which may modulate neurotransmitter release.
- Indications:
- Adjunctive therapy for partial-onset seizures in patients 1 month of age and older.
- Adjunctive therapy for myoclonic seizures in patients 12 years of age and older with juvenile myoclonic epilepsy.
- Adjunctive therapy for primary generalized tonic-clonic seizures in patients 6 years of age and older with idiopathic generalized epilepsy.
- Dosage Forms: Available in immediate-release (IR) tablets, oral solutions, and intravenous (IV) formulations. Extended-release (ER) formulations also exist.
- Generics Dominance: The vast majority of levetiracetam prescriptions are now filled with generic products. This has made it a first-line or second-line treatment option for many patients due to its affordability.
- UCB Pharma's Strategy: UCB Pharma has transitioned its focus to newer, more innovative epilepsy treatments with potentially higher margins and longer exclusivity periods, such as Brivaracetam (Briviact) and Cenobamate (Xcopri). However, Keppra remains a foundational product within UCB's portfolio, contributing to cash flow through its established brand recognition and preferred formulary status in some instances.
What are the Future Market Projections for Levetiracetam?
The future market trajectory for levetiracetam is largely dictated by its status as a genericized commodity.
- Continued Generic Sales: The demand for levetiracetam is expected to remain stable or grow modestly, driven by the ongoing prevalence of epilepsy and the cost-effectiveness of generic formulations. The overall antiepileptic drug market growth will continue to absorb levetiracetam volume.
- Price Stability (Low): Pricing for generic levetiracetam is expected to remain low and potentially decline further with continued market competition, although significant price drops are unlikely given the already low base.
- Impact of New Therapies: The development and adoption of newer AEDs with novel mechanisms of action and potentially improved efficacy or tolerability profiles for specific patient subgroups could gradually impact levetiracetam's market share, but its broad utility and low cost will ensure its continued relevance.
- UCB Pharma's Portfolio Shift: UCB Pharma will continue to prioritize its newer pipeline and marketed products, with levetiracetam primarily serving as a legacy product that contributes to revenue and market presence in epilepsy.
Key Takeaways
- Keppra's (levetiracetam) market trajectory shifted from significant revenue generation during patent exclusivity to a predominantly generic market following patent expiries in the late 2000s and early 2010s.
- Global sales of branded Keppra peaked at over €1.7 billion in 2010 before experiencing a substantial decline due to generic competition, with prices eroding by 80-90%.
- Levetiracetam remains a widely prescribed and clinically important antiepileptic drug due to its efficacy, tolerability, and the affordability of generic versions, contributing to the overall growth of the epilepsy drugs market.
- UCB Pharma has pivoted its strategic focus to newer epilepsy therapies, although levetiracetam continues to be a foundational product within its portfolio.
- The future market for levetiracetam will be characterized by stable, low-cost generic sales, with demand supported by its established role in epilepsy management.
Frequently Asked Questions
-
When did Keppra lose its US market exclusivity?
Keppra lost its primary US market exclusivity in 2008.
-
What is the primary mechanism of action for levetiracetam?
Levetiracetam's mechanism of action is not fully understood but is believed to involve binding to the synaptic vesicle protein 2A (SV2A).
-
Which are the main indications for levetiracetam?
Levetiracetam is indicated for partial-onset seizures, myoclonic seizures, and generalized tonic-clonic seizures.
-
Has UCB Pharma discontinued the branded Keppra product?
UCB Pharma continues to market branded Keppra, but its market share is significantly diminished due to generic availability.
-
What is the projected market growth for the overall antiepileptic drug market?
The global epilepsy drugs market is projected to grow at a compound annual growth rate of 2.6% from 2023 to 2030.
Citations
[1] Grand View Research. (2023). Epilepsy Drugs Market Size, Share & Trends Analysis Report By Drug Class, By Distribution Channel, By Region, And Segment Forecasts, 2023 - 2030. Retrieved from https://www.grandviewresearch.com/industry-analysis/epilepsy-drugs-market
[2] UCB Pharma. (2009). UCB Annual Report 2008. Retrieved from UCB Investor Relations archives.
[3] UCB Pharma. (Various Years). UCB Annual Reports and Financial Statements. Retrieved from UCB Investor Relations archives.
[4] UCB Pharma. (2023). UCB Annual Report 2022. Retrieved from https://www.ucb.com/investors/UCB-Annual-Reports