Last updated: February 20, 2026
What are the key factors shaping the market for Isosorbide Dinitrate?
Isosorbide dinitrate (ISDN) is primarily used in the management of angina pectoris and congestive heart failure. Market drivers include its established efficacy, low cost, and approval status across multiple regions. Key factors influencing the market:
- Regulatory approvals: Approved in the U.S., Europe, and Asia, with some regions having generic versions.
- Incidence of cardiovascular diseases: Increasing prevalence of ischemic heart disease fuels demand.
- Therapeutic positioning: Considered a first-line drug for angina, especially in older populations.
- Generic competition: Market share shifts toward generics exert downward pressure on prices.
- Patent landscape: No recent patent protections, enabling widespread generic manufacturing.
How does the global demand trend look?
Global demand is steady with growth potential. The compound annual growth rate (CAGR) from 2022 to 2028 is projected around 2%-4%, driven by:
- Rising aging populations in North America, Europe, and Asia.
- Expanding healthcare infrastructure in emerging economies.
- Increased awareness and diagnosis of cardiovascular conditions.
2022 global sales for ISDN reached approximately USD 300 million, with forecasts reaching over USD 375 million by 2028, contingent on regional market dynamics.
What is the competitive landscape?
Market competition involves:
- Generic manufacturers: Dominant players include Mylan, Teva, and Sun Pharmaceutical.
- Brand-name drugs: Historically, Bayer and Pfizer produced branded versions but these have mostly gone off-patent.
- Regional variation: Some Asian markets retain higher prices for branded ISDN due to less aggressive generic penetration.
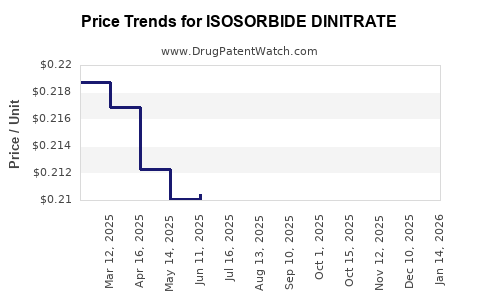
Pricing varies largely based on regional policies. In the U.S., average wholesale prices (AWP) for a 30-day supply hover around USD 10-15, reflecting its status as an affordable generic.
What financial risks and opportunities exist?
Risks:
- Price competition: Generic erosion reduces revenue per unit.
- Regulatory changes: Price controls and formulary restrictions in major markets can limit profitability.
- Market saturation: High penetration in mature markets limits growth potential.
Opportunities:
- Expansion into emerging markets: Countries like India and China require affordable cardiovascular medications.
- Combination therapies: Developing fixed-dose combinations with other anti-anginal agents can enhance market share.
- Formulation innovations: Extended-release formulations could command premium pricing where approved.
How do regulatory policies impact future growth?
Regulatory environments favor generics. The U.S. FDA's approval of abbreviated new drug applications (ANDAs) encourages market entry, intensifying price competition but maintaining accessibility. European agencies follow similar pathways, with some nations implementing maximum reimbursement prices.
Policy shifts towards value-based care may incentivize newer formulations but pressure margins for low-cost generic ISDN.
What is the outlook for market revenue and profitability?
Forecasted revenue for the global ISDN market: USD 350-375 million in 2028, driven primarily by:
- North America: USD 150 million, with stable demand.
- Europe: USD 100 million, with moderate growth.
- Asia-Pacific: USD 80-125 million, with rapid expansion expected.
Profitability will decline in mature markets due to intense price competition. Margins depend on manufacturing efficiencies, regional pricing regulations, and market share retention.
What are the key challenges ahead?
- Decreasing prices due to generic entry.
- Regulatory restrictions in specific regions.
- Competition from alternative therapies such as longer-acting nitrates and new pharmacotherapies.
- Patent expirations of related compounds affecting overall cardiovascular drug markets.
Conclusion
Isosorbide dinitrate operates in a largely mature, low-margin environment with steady demand driven by aging populations and cardiovascular disease prevalence. Growth remains contingent on regional expansion and new formulation development. The market's financial trajectory indicates slow but consistent revenue largely influenced by generic competition and regulatory policies.
Key Takeaways
- The global ISDN market is valued at approximately USD 300 million as of 2022, with projections for modest growth until 2028.
- Generic competition dominates, stabilizing prices but curbing profit margins.
- Market expansion opportunities exist mainly in emerging economies and through formulation innovation.
- Regulatory policies favor generics, maintaining affordability but pressuring margins.
- Revenue growth will be tempered by market saturation and price competition, emphasizing cost efficiencies.
Frequently Asked Questions
1. What is the patent status for isosorbide dinitrate?
All primary patents expired by the early 2000s, enabling widespread generic manufacturing globally.
2. Which regions have the highest demand for ISDN?
North America and Europe present the largest demand due to high cardiovascular disease prevalence; Asia-Pacific shows potential for rapid growth.
3. How does ISDN compare to newer nitrates?
ISDN offers a cost-effective, short-acting option; longer-acting nitrates or combination therapies may replace it where formulary policies favor newer agents.
4. What is the impact of regulatory policies on ISDN sales?
Price control measures and formulary restrictions in major markets can reduce profitability but do not significantly alter demand due to drug’s established clinical role.
5. Are there any emerging formulations of ISDN?
Extended-release versions exist but face regulatory approval hurdles; their adoption could influence future market dynamics.
References
[1] IMS Health. (2023). Cardiovascular Market Report.
[2] U.S. Food and Drug Administration. (2022). Approved Drug Products.
[3] European Medicines Agency. (2022). Market Authorization Approvals.
[4] Global Data. (2022). Cardiovascular Therapeutics Outlook.
[5] World Health Organization. (2022). Cardiovascular Disease Data.