ISENTRESS HD Drug Patent Profile
✉ Email this page to a colleague
Which patents cover Isentress Hd, and what generic alternatives are available?
Isentress Hd is a drug marketed by Msd Sub Merck and is included in one NDA. There are five patents protecting this drug and two Paragraph IV challenges.
This drug has one hundred and thirty-nine patent family members in forty-six countries.
The generic ingredient in ISENTRESS HD is raltegravir potassium. There are five drug master file entries for this compound. Nine suppliers are listed for this compound. Additional details are available on the raltegravir potassium profile page.
DrugPatentWatch® Generic Entry Outlook for Isentress Hd
Isentress Hd was eligible for patent challenges on October 12, 2011.
There are two Paragraph IV patent challenges for this drug. This may lead to patent invalidation or a license for generic production.
Indicators of Generic Entry
Summary for ISENTRESS HD
| International Patents: | 139 |
| US Patents: | 5 |
| Applicants: | 1 |
| NDAs: | 1 |
| Finished Product Suppliers / Packagers: | 9 |
| Raw Ingredient (Bulk) Api Vendors: | 90 |
| Clinical Trials: | 88 |
| Patent Applications: | 126 |
| Formulation / Manufacturing: | see details |
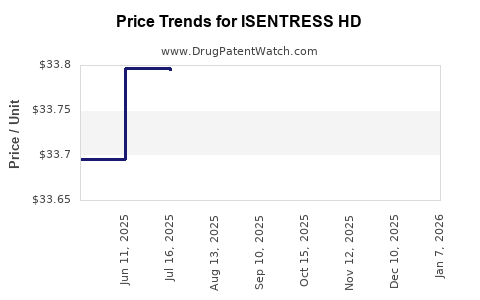
| Drug Prices: | Drug price information for ISENTRESS HD |
| DailyMed Link: | ISENTRESS HD at DailyMed |
Recent Clinical Trials for ISENTRESS HD
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| Unity Health Toronto | Phase 2 |
| St. Michael's Hospital, Toronto | Phase 2 |
| Canadian Institutes of Health Research (CIHR) | Phase 2 |
Pharmacology for ISENTRESS HD
| Drug Class | Human Immunodeficiency Virus Integrase Strand Transfer Inhibitor |
| Mechanism of Action | HIV Integrase Inhibitors |
Anatomical Therapeutic Chemical (ATC) Classes for ISENTRESS HD
Paragraph IV (Patent) Challenges for ISENTRESS HD
| Tradename | Dosage | Ingredient | Strength | NDA | ANDAs Submitted | Submissiondate |
|---|---|---|---|---|---|---|
| ISENTRESS HD | Tablets | raltegravir potassium | 600 mg | 022145 | 1 | 2022-10-21 |
| ISENTRESS HD | Tablets | raltegravir potassium | 400 mg | 022145 | 1 | 2011-10-12 |
US Patents and Regulatory Information for ISENTRESS HD
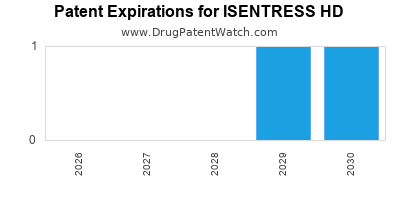
ISENTRESS HD is protected by five US patents.
Patents protecting ISENTRESS HD
Solid pharmaceutical compositions containing an integrase inhibitor
Patent Number: ⤷ Try a Trial
Patent Expiration: ⤷ Try a Trial
Patented Use: TREATMENT OF HIV-1 INFECTION
N-substituted hydroxypyrimidinone carboxamide inhibitors of HIV integrase
Patent Number: ⤷ Try a Trial
Patent Expiration: ⤷ Try a Trial
Potassium salt of an HIV integrase inhibitor
Patent Number: ⤷ Try a Trial
Patent Expiration: ⤷ Try a Trial
Pharmaceutical composition containing an anti-nucleating agent
Patent Number: ⤷ Try a Trial
Patent Expiration: ⤷ Try a Trial
Patented Use: TREATMENT OF HIV INFECTION
Solid pharmaceutical compositions containing an integrase inhibitor
Patent Number: ⤷ Try a Trial
Patent Expiration: ⤷ Try a Trial
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Msd Sub Merck | ISENTRESS HD | raltegravir potassium | TABLET;ORAL | 022145-002 | May 26, 2017 | RX | Yes | Yes | ⤷ Try a Trial | ⤷ Try a Trial | Y | Y | ⤷ Try a Trial | ||
| Msd Sub Merck | ISENTRESS HD | raltegravir potassium | TABLET;ORAL | 022145-002 | May 26, 2017 | RX | Yes | Yes | ⤷ Try a Trial | ⤷ Try a Trial | Y | ⤷ Try a Trial | |||
| Msd Sub Merck | ISENTRESS HD | raltegravir potassium | TABLET;ORAL | 022145-002 | May 26, 2017 | RX | Yes | Yes | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Msd Sub Merck | ISENTRESS HD | raltegravir potassium | TABLET;ORAL | 022145-002 | May 26, 2017 | RX | Yes | Yes | ⤷ Try a Trial | ⤷ Try a Trial | Y | ⤷ Try a Trial | |||
| Msd Sub Merck | ISENTRESS HD | raltegravir potassium | TABLET;ORAL | 022145-002 | May 26, 2017 | RX | Yes | Yes | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for ISENTRESS HD
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Msd Sub Merck | ISENTRESS HD | raltegravir potassium | TABLET;ORAL | 022145-002 | May 26, 2017 | ⤷ Try a Trial | ⤷ Try a Trial |
| Msd Sub Merck | ISENTRESS HD | raltegravir potassium | TABLET;ORAL | 022145-002 | May 26, 2017 | ⤷ Try a Trial | ⤷ Try a Trial |
| Msd Sub Merck | ISENTRESS HD | raltegravir potassium | TABLET;ORAL | 022145-002 | May 26, 2017 | ⤷ Try a Trial | ⤷ Try a Trial |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
International Patents for ISENTRESS HD
See the table below for patents covering ISENTRESS HD around the world.
| Country | Patent Number | Title | Estimated Expiration |
|---|---|---|---|
| Luxembourg | 91428 | ⤷ Try a Trial | |
| Argentina | 101429 | SAL POTÁSICA DE UN INHIBIDOR DE LA INTEGRASA DE VIH | ⤷ Try a Trial |
| Slovenia | 1819700 | ⤷ Try a Trial | |
| China | 101068793 | Potassium salt of an HIV integrase inhibitor | ⤷ Try a Trial |
| Eurasian Patent Organization | 007060 | N-ЗАМЕЩЁННЫЕ ГИДРОКСИПИРИМИДИНОН-КАРБОКСАМИДНЫЕ ИНГИБИТОРЫ ВИЧ ИНТЕГРАЗЫ (N-SUBSTITUTED HYDROXYPYRIMIDINONE CARBOXAMIDE INHIBITORS OF HIV INTEGRASE) | ⤷ Try a Trial |
| Brazil | PI0518760 | sal de potÁssio do composto, e, composiÇço farmacÊutica | ⤷ Try a Trial |
| >Country | >Patent Number | >Title | >Estimated Expiration |
Supplementary Protection Certificates for ISENTRESS HD
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 1441735 | C20080001 | Estonia | ⤷ Try a Trial | PRODUCT NAME: ISENTRESS; AUTHORISATION NO.: EMA/106827/2018; AUTHORISATION DATE: 20180222 |
| 1441735 | C 2008 008 | Romania | ⤷ Try a Trial | PRODUCT NAME: N-(2-(4-(4-FLUOROBENZILCARBAMOIL)-5-HIDROXI-1-METIL-6-OXO-1,6-DIHIDROPIRIMIDIN-2-IL)PROPAN-2+IL)-5-METIL-1,3,4-OXADIAZOL-2-CARBOXAMIDA - RALTEGRAVIR SAU O SARE ACCEPTABILAFARMACEUTIC A ACESTUIA, INSPECIAL SAREA DE POTASIU; NATIONAL AUTHORISATION NUMBER: EMEA EU/1/07/436/001, EMEA EU/1/07/436/002; DATE OF NATIONAL AUTHORISATION: 20071220; NUMBER OF FIRST AUTHORISATION IN EUROPEAN ECONOMIC AREA (EEA): EMEA EU/1/07/436/001, EMEA EU/1/07/436/002; DATE OF FIRST AUTHORISATION IN EEA: 20071220 |
| 1441735 | DO 67; 3-2008 | Slovakia | ⤷ Try a Trial | PRODUCT NAME: RALTEGRAVIR; REGISTRATION NO/DATE: Registration in Community EU/1/07/436/001-002 20071220; FORMER OWNER: ISTITUTO DI RICERCHE DI BIOLOGIA MOLECOLARE P. ANGELETTI SPA. IT |
| 1441735 | 20221021 | Netherlands | ⤷ Try a Trial | 20221021, EXPIRES: 20221219 |
| 1441735 | PA2008007 | Lithuania | ⤷ Try a Trial | PRODUCT NAME: RALTEGRAVIRUM; REG. NO/DATE: EU/1/07/436/001-002 20071220 |
| 1441735 | SPC/GB08/020 | United Kingdom | ⤷ Try a Trial | PRODUCT NAME: RALTEGRAVIR OR A PHARMACEUTICALLY ACCEPTABLE SALT THEREOF, ESPECIALLY THE POTASSIUM SALT; REGISTERED: UK EU/1/07/436/001 20080102 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Make Better Decisions: Try a trial or see plans & pricing
Drugs may be covered by multiple patents or regulatory protections. All trademarks and applicant names are the property of their respective owners or licensors. Although great care is taken in the proper and correct provision of this service, thinkBiotech LLC does not accept any responsibility for possible consequences of errors or omissions in the provided data. The data presented herein is for information purposes only. There is no warranty that the data contained herein is error free. thinkBiotech performs no independent verification of facts as provided by public sources nor are attempts made to provide legal or investing advice. Any reliance on data provided herein is done solely at the discretion of the user. Users of this service are advised to seek professional advice and independent confirmation before considering acting on any of the provided information. thinkBiotech LLC reserves the right to amend, extend or withdraw any part or all of the offered service without notice.


