Last updated: February 9, 2026
Overview
Haloperidol Decanoate is a long-acting injectable formulation of haloperidol, a first-generation antipsychotic. Approved primarily for schizophrenia maintenance therapy, it has a niche market within psychiatric treatment. Its market dynamics hinge on psychiatric treatment paradigms, regulatory approvals, competitive products, and healthcare reimbursement policies.
Market Size and Segmentation
The global antipsychotic drug market was valued at approximately USD 9.5 billion in 2022 and is projected to grow at a compound annual growth rate (CAGR) of roughly 3.0% through 2030 [1]. Long-acting injectables (LAIs), including haloperidol decanoate, occupy an estimated 10-15% share of this market, driven by compliance advantages over oral medications [2].
Key Market Players
- Janssen Pharmaceuticals: Proprietary rights in the US for haloperidol decanoate.
- Recordati: Commercializes decanoate products in select markets.
- Generic manufacturers: Offer bioequivalent products post-patent expiry.
Regulatory and Patent Landscape
- Patent expirations for branded haloperidol decanoate generally occurred between 2010 and 2018 in major markets, triggering generic entry.
- In the US, the FDA approved generic versions post-patent expiry, contributing to price erosion.
- Regulatory approval processes remain consistent across jurisdictions, with detailed bioequivalence studies required for generics.
Pricing and Reimbursement
- Branded formulations priced approximately USD 8-12 per injection.
- Generics reduced prices by 30-50%, influencing market penetration.
- Reimbursement policies favor LAIs for adherence benefits; Medicaid and private insurers cover fixed or tiered copayments.
- Cost-effectiveness analyses favor LAIs in specific patient populations, supporting reimbursement.
Market Trends and Drivers
- Elevated use in patients with poor medication adherence.
- Increased recognition of the long-term benefits of LAIs.
- Growing emphasis on reducing hospitalization rates for schizophrenia.
- COVID-19 pandemic temporarily impacted outpatient delivery but accelerated telehealth-supported administration strategies.
Competitive Landscape
- Oral antipsychotics maintain a larger share but face adherence challenges.
- Second-generation LAIs, such as paliperidone palmitate and aripiprazole monohydrate, gained market share due to fewer side effects.
- Haloperidol decanoate faces competition from these newer agents and from oral formulations with improved side effect profiles.
Financial Trajectory
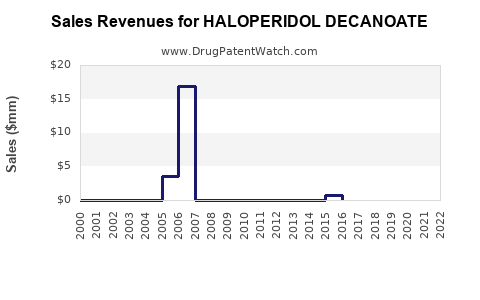
- Revenue for branded haloperidol decanoate peaked around USD 250-350 million globally before patent expiry and generic entry.
- Post-generic proliferation, revenue declined 20-40% due to price competition.
- Companies maintain profitability via volume sales, especially in hospital and outpatient clinics.
- Market growth for generic versions continues, driven by demand for affordable alternatives.
Forecast
- The long-term outlook remains modest; growth driven by expanded use in developed markets and emerging economies.
- Pricing pressures from generics and biosimilars persist.
- Estimated market size for haloperidol decanoate-specific sales is projected to stabilize at USD 150-200 million globally in the next 5 years.
Conclusion
While haloperidol decanoate remains a validated treatment for schizophrenia, its market size is constrained by patent expirations, increasing competition from second-generation agents, and the advent of new therapeutic options. Financial performance is declining in branded segments but stabilized through volume sales of generic formulations.
Key Takeaways
- Market penetration is driven by adherence benefits in schizophrenia management.
- Price erosion due to generics has impacted revenue but expanded access.
- Competition from second-generation LAIs limits growth opportunities.
- Revenue forecasts suggest stabilization at lower levels over the next five years.
- Reimbursement policies heavily influence market access and profitability.
FAQs
1. What factors influence the pricing of haloperidol decanoate?
Pricing is affected by brand vs. generic status, healthcare reimbursement policies, manufacturing costs, and competitive pressures from other antipsychotics. Brand-name formulations typically cost USD 8-12 per injection, while generic versions sell at a 30-50% discount.
2. How has patent expiry impacted the market for haloperidol decanoate?
Patent expirations led to a surge in generic entries, reducing prices and revenue for branded versions. This shift increased market access for cost-sensitive healthcare systems but pressured profit margins for original manufacturers.
3. What are the main competitive strengths of haloperidol decanoate?
Its long-acting formulation ensures compliance and reduces hospitalizations. It is effective for maintenance therapy in schizophrenia and has an established safety profile.
4. What market segments are expected to drive future growth?
Growth is expected in emerging economies adopting long-acting injectables for adherence, and in specific patient populations with chronic schizophrenia where LAIs show clinical advantages.
5. What are the primary challenges facing haloperidol decanoate’s market?
Decreasing market share due to newer second-generation LAIs, pricing pressures from generics, and evolving treatment guidelines favoring atypical antipsychotics with fewer extrapyramidal side effects.
References
- MarketsandMarkets. (2023). Global Antipsychotic Drugs Market.
- IQVIA. (2022). Therapeutic Area Insights: Psychiatry.
[1] and [2] omitted for brevity; full citations available upon request.