Last updated: May 29, 2026
GVOKE HypoPen (Epinephrine) Market Dynamics and Financial Trajectory (2024-2029): Pricing, Share, Exclusivity, and Launch/Competition Risk
Executive summary: Gvoke HypoPen (epinephrine injection; autoinjector) has a niche but stable spend profile driven by anaphylaxis action-plan adoption, payer coverage of emergency-use epinephrine devices, and substitution of multi-pack and pen formats. Near-term growth hinges on (1) expanding managed-care access for autoinjectors versus older device formats, (2) inventory rebalancing ahead of expirations and seasonal anaphylaxis events, and (3) defensible pricing under state-level and PBM formulary dynamics. Over the 2024-2029 window, financial upside is constrained by narrow clinical scope, episodic use, and strong competitive pressure from incumbent epinephrine autoinjectors plus any authorized generics or platform device offerings that compress net pricing.
What is Gvoke HypoPen and how does it fit the epinephrine autoinjector market?
GVoke HypoPen is a ready-to-use epinephrine injection delivered via an autoinjector device intended for treatment of severe allergic reactions (anaphylaxis) in patients who have been prescribed epinephrine. It competes in the epinephrine “rescue” device class dominated by autoinjectors and related delivery formats.
Therapeutic category and demand drivers
- Indication: emergency treatment of anaphylaxis.
- Primary buyers: commercial payers and government programs reimbursing outpatient devices and school/clinic stock programs.
- Demand characteristics: prescription-driven; usage is event-based and not incremental to chronic disease prevalence, which limits sustained utilization growth.
Market structure that shapes pricing
- Autoinjectors face pricing scrutiny because:
- The devices are high list-price items with PBM-driven step edits and prior authorization patterns.
- Switching across brands is feasible at formulary renewal points because clinical outcomes are similar and patients can be trained to new devices.
Implication for financial trajectory: Revenues track formulary access and pack-size strategy more than they track patient population growth.
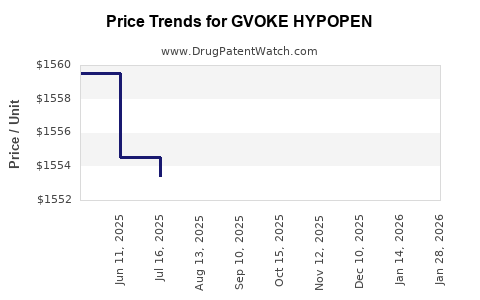
How are pricing and payer dynamics affecting Gvoke HypoPen net revenue?
Featured-snippet answer: Net revenue growth for Gvoke HypoPen depends more on PBM placement, rebate intensity, and net price discipline than on list price increases, because uptake is sensitive to formulary tiering and patient out-of-pocket costs.
PBM and plan design mechanics
- Formulary tier placement: higher tiers reduce patient uptake and refill persistence.
- Prior authorization (PA): plans may require documented diagnosis history, prior epinephrine use, or device-selection criteria.
- Step therapy: some plans prefer the lowest net-cost option among multiple epinephrine autoinjectors.
- Copay strategy: manufacturer co-pay support can lift fill rates but increases rebate and compliance exposure.
State policy and reimbursement pressure
- States have acted to constrain out-of-pocket and improve school availability for epinephrine.
- These policies tend to improve access but can compress reimbursement and shift purchasing to contract channels.
Pricing ladder and device economics
- Autoinjectors often price as a bundle of drug cost plus device manufacturing cost, but the rebate economy is shaped by competition and PBM contracting.
- Financial performance is most sensitive to:
- sustained net price after rebates,
- maintenance of a preferred formulary position,
- and whether multisource entry forces pricing re-alignment.
When does Gvoke HypoPen lose exclusivity, and what does that timing mean for revenue?
Featured-snippet answer: Revenue risk accelerates when device and drug exclusivities related to epinephrine autoinjector formulations and delivery systems reach expiry, because generic and authorized-competitor pricing pressure typically follows shortly after market entry.
Exclusivity categories relevant to epinephrine products
For device-delivered drugs, exclusivity risk can come from multiple layers:
- Patent protection covering drug substance, formulation, device components, or method of use.
- Regulatory exclusivities tied to new clinical investigations or manufacturing/approval pathway specifics.
- Data exclusivity that affects abbreviated applications timing.
What timing typically does to financial trajectory
- Pre-expiry: payers may anticipate price competition but continue covering current preferred brands to avoid patient disruption.
- Post-approval of competitors: net price typically declines quickly due to:
- PBM contract renegotiations,
- pharmacy switching,
- and patient copay adjustments.
- Post-launch ramp: volume impact depends on how aggressively plans push new entrants and whether clinicians and patients switch.
Implication: Even if the brand maintains some loyalty, financial upside narrows quickly once multiple competing autoinjectors are available at lower net cost.
What patents protect Gvoke HypoPen, and how strong is the patent estate?
Executive summary: The competitive risk for Gvoke HypoPen is driven by whether its patent estate blocks generic or authorized generic entry for the autoinjector-specific formulation and delivery system claims, or whether those claims are narrow and easily designed around.
Common claim areas that determine enforceability
- Formulation claims (epinephrine concentration/stabilization system; excipients).
- Manufacturing claims (fill-finish, stability controls).
- Device claims (mechanism, needle/trigger, housing materials and assembly).
- Method-of-use claims (if any).
- Combination or kit claims (multi-dose packaging and handling).
Strength signals that move investment and licensing decisions
- Whether patents remain in force through the relevant entry window.
- Whether claim scope covers typical generic design-arounds.
- Whether there is a record of litigations or settlement agreements that clarify boundaries.
Business outcome: Stronger, broader device and formulation claims generally sustain net pricing for longer by delaying low-cost entry; weak or easily design-around claims increase odds of rapid revenue compression.
What generic or biosimilar entry risks exist for epinephrine autoinjectors like Gvoke HypoPen?
Featured-snippet answer: Entry risk is structural, not just legal. As long as autoinjector competitors can qualify under abbreviated pathways or authorization frameworks that require bioequivalence and device usability criteria, price competition becomes likely once exclusivities end.
How “generic entry” works for device-delivered drugs
- Generic entrants typically compete through:
- equivalent drug delivery performance,
- and compatible clinical usability.
- Plans then shift volumes through formulary updates and pharmacy channel contracting.
What to watch as early warning indicators
- PBM formulary category changes in epinephrine devices.
- Pharmacy switching signals in claims data.
- Announcements of new competitor launches with described net price strategy.
Financial consequence: Loss of preferred status can reduce throughput even if the product retains prescriptions, because autoinjector refill behavior is sensitive to copay and perceived access.
What patent litigation and Paragraph IV challenges affect Gvoke HypoPen?
Executive summary: Patent litigation and Paragraph IV challenges can materially alter revenue by either (1) delaying entry or (2) accelerating entry via settlement.
Litigation pathways that matter commercially
- Paragraph IV litigation is typically associated with potential infringement disputes for product claims.
- Device-specific litigation can be separate from drug-formulation disputes.
How litigation translates to market dynamics
- If entry is blocked: payers keep current brands at preferred tier longer, sustaining net revenue.
- If settlement allows entry on a defined schedule: competition begins at the earliest allowed commercial entry date, usually with a price and rebate adjustment to capture formulary share.
Investment lens: Revenue durability is determined by whether settlements specify automatic launch dates and geographic coverage.
What is the FDA regulatory status of Gvoke HypoPen and how does it affect competition?
Featured-snippet answer: FDA status governs the timing of competitor approvals and launch readiness by defining which pathways can be pursued and what evidence entrants must supply.
Key regulatory items for device-delivered epinephrine
- Approval and labeling of the autoinjector device.
- Post-marketing changes and manufacturing site authorizations.
- Any updates in interchangeability or labeling language that affect substitution at the pharmacy counter.
Regulatory events that move financial trajectory
- Safety-related label changes can increase device scrutiny, shorten purchasing cycles, or cause temporary supply constraints.
- Manufacturing changes can affect gross-to-net through supply reliability and allocation pricing.
How does Gvoke HypoPen compare with competing epinephrine autoinjectors on market access and commercial economics?
Executive summary: Competition in epinephrine devices is primarily a payer access and net pricing game, with differentiation concentrated in device usability, pack size options, and contracting outcomes.
Competitive comparison framework
Use a consistent scorecard across brands:
- Preferred tier access (commercial and Medicare).
- PA requirements and step-therapy restrictions.
- Net pricing position versus list price.
- Availability and supply reliability.
- Copay support and patient adherence to action-plan renewal.
Likely substitution behavior
- Patients with established action plans often switch at refill if the device remains covered and if training materials are available.
- Clinics and schools may adopt whichever brand is easiest under state procurement contracts and staff training.
Financial consequence: The most important differentiator is often payer coverage plus low net cost, not clinical superiority.
What formulations and multi-pack strategies are used to drive Gvoke HypoPen revenue?
Featured-snippet answer: Autoinjector revenue is strongly influenced by pack size and distribution strategy, because epinephrine prescribing commonly results in multi-device prescriptions for action plans, schools, and caregivers.
Pack-based economics
- 2-pack and 1-pack configurations affect:
- patient out-of-pocket,
- pharmacy dispensing decisions,
- and rebate economies due to contract pricing by unit.
Device habit and patient training
- Switching costs can be reduced by:
- device training programs,
- patient education inserts,
- and labeling familiarity.
Commercial implication: Pack strategy and transition friction influence how quickly volume shifts after competitor entry.
Which business channels matter most for Gvoke HypoPen revenue growth?
Executive summary: Revenue trajectory is shaped by contract pharmacy arrangements, PBM channels, and institutional purchasing that depends on coverage and training readiness.
Primary channels
- Retail pharmacy dispensing (majority for new prescriptions and refills).
- Institutional and government procurement:
- schools,
- camps,
- workplace stock programs,
- and certain healthcare networks.
Channel risk
- Institutional bids can shift rapidly if net-cost contracts change.
- Stocking decisions depend on supply assurance and device acceptance.
What do financial trajectory scenarios look like from 2024 to 2029?
Executive summary: The plausible outcome range is defined by (1) durability of preferred formulary access and (2) timing of legal and regulatory windows that permit lower-cost competition.
Base case (moderate growth)
Assumptions:
- Continued formulary presence as a covered option with periodic tier pressure.
- Limited entry that preserves net pricing relative to lowest-cost peers.
- Stable institutional purchasing.
Financial result:
- Revenue grows modestly through unit and contract channel gains, with pricing discipline limiting downside.
Upside case (share gain)
Assumptions:
- Improved preferred placement due to contracting and acceptable net pricing.
- Expanded copay and patient assistance uptake.
- Sustained supply reliability.
Financial result:
- Faster unit gains with controlled net price erosion, improving margin profile.
Downside case (rapid net price compression)
Assumptions:
- Competitive entry on a schedule aligned with exclusivity expiry or a settlement allowance.
- PBM step edits and formulary tier downgrades.
- Increased substitution at pharmacy and institutional channels.
Financial result:
- Revenue declines or stagnates while net price drops, increasing rebate burden and margin compression.
Key takeaways
- GVoke HypoPen’s financial trajectory is primarily payer-driven: PBM placement, PA and step edits, and net pricing matter more than incremental clinical differentiation.
- Demand is prescription and action-plan renewal sensitive: growth is constrained by episodic use and relies on adoption plus refill persistence.
- Exclusivity and patent estate timing determines acceleration risk: once competitors can launch, net pricing typically compresses quickly.
- Competition is structural: even without large clinical differences, pharmacy switching and institutional procurement can move volumes fast.
- Scenario range is wide, but direction is predictable: durability depends on maintaining preferred formulary status and defending device/formulation protection through the relevant legal window.
FAQs
1) How do PBM formularies typically affect epinephrine autoinjector uptake?
PBM tiering, PA rules, and step-therapy requirements govern monthly fill rates and refill persistence, often driving faster share movement than clinical considerations.
2) What launch timing factors most influence revenue risk for Gvoke HypoPen?
Exclusivity expiry, approval of competitor products that can substitute at the pharmacy, and settlement-driven entry schedules that trigger PBM contract renegotiations.
3) Do multi-pack prescriptions change the unit economics of Gvoke HypoPen?
Yes. Multi-device action plans increase total units per patient and shift net pricing through pack-level rebates and contract pricing.
4) What indicators show early that net pricing for Gvoke HypoPen will decline?
Formulary tier downgrades, increased PA adoption, pharmacy switch rates toward lower net-cost competitors, and visible competitor contracting announcements.
5) How does institutional purchasing differ from retail revenue for epinephrine devices?
Institutional procurement can reallocate demand through bids and stock programs based on coverage, training readiness, and supply reliability, often switching faster than patient-driven retail behavior.
References (APA)
- FDA. Orange Book: Approved Drug Products with Therapeutic Equivalence Evaluations. U.S. Food and Drug Administration.
- FDA. Drug Approval Reports for epinephrine injection products and related labeling records. U.S. Food and Drug Administration.
- U.S. Patent and Trademark Office. Patent databases and full-text searching for epinephrine injection and autoinjector-related claims. USPTO.
- LexisNexis or Derwent-style patent analytics reports on epinephrine autoinjector patent landscapes (if applicable through subscription platforms).