Last updated: January 15, 2026
Executive Summary
GLATOPA (generic glatiramer acetate) is a biosimilar marketed as a cost-effective alternative to the innovator drug, Copaxone (glatiramer acetate), used primarily to treat multiple sclerosis (MS). Since its launch, GLATOPA has influenced the MS treatment market through competitive pricing and strategic patent challenges. This analysis explores the pharmaceutical market dynamics shaping its trajectory, reviews revenue trends, assesses competitive pressures, and forecasts future developments for stakeholders.
What is GLATOPA and How Does It Fit Into the MS Treatment Market?
GLATOPA, developed by Sandoz (a Novartis division), was approved by the FDA in 2015 as a biosimilar of Copaxone (originally by Teva Pharmaceuticals). Its primary indication is relapsing-remitting multiple sclerosis (RRMS).
| Attribute |
Details |
| Active Ingredient |
Glatiramer acetate (20 mg/mL and 40 mg/mL formulations) |
| Approval Date |
July 2015 (FDA) |
| Manufacturers |
Sandoz (Novartis), Mylan (previously attempted), Teva (original innovator) |
| Pricing Strategy |
Lower-cost alternative to Copaxone |
Source: FDA Approval Documents [1]
Market Dynamics of GLATOPA
How Has the Entry of GLATOPA Impacted the MS Market?
The introduction of GLATOPA introduced price competition, historically a dominant arena for Teva’s Copaxone. Key market dynamics include:
| Dynamic |
Impact |
| Pricing Pressure |
Significant reductions in MS drug prices, driven by biosimilar competition |
| Market Share Redistribution |
Gains for biosimilars led by GLATOPA, at the expense of Copaxone in the U.S. |
| Patent Litigation and Exclusivity |
Teva's patent protections delayed biosimilar market penetration until around 2019-2020 |
Patent and Regulatory Landscape
Teva's “Patent Evergreening” strategy extended patent exclusivity through multiple legal and regulatory avenues. However, biosimilar approval and subsequent patent challenges allowed competition to emerge around 2019.
| Key Patent Litigation Events |
Dates |
Outcome |
| First Abbreviated New Drug Application (ANDA) filing |
2012 |
Filed by Sandoz |
| Patent litigations and court challenges |
2013-2019 |
Generally favor biosimilar entry post-2019 |
| Biosimilar market entry in the U.S. |
2019 |
First biosimilar approval effective, with gradual market adoption |
Sources: Court documents, FDA approvals [1][2]
Market Adoption Trends (2015-2023)
| Time Period |
Market Share (Approximate) |
Key Drivers |
| 2015-2017 |
Low (~5%) |
Patent protections, limited biosimilar availability |
| 2018-2019 |
Moderate (~20%) |
Patent expiry, increased insurer acceptance |
| 2020-2023 |
Significant (~35-40%) |
Increased biosimilar uptake, price sensitivity |
Note: Precise market share metrics sourced from IQVIA and EvaluatePharma [3]
Financial Trajectory of GLATOPA
Revenue Trends
Sandoz's GLATOPA revenue has exhibited growth aligned with biosimilar market acceptance, though overall sales have been tempered by patent litigation, physician adoption, and insurer formularies.
| Year |
Estimated Global Revenue (USD million) |
Notes |
| 2015 |
<$50 |
Launch year, limited market presence |
| 2016 |
~$70 |
Early adoption phase |
| 2017 |
~$100 |
Growing acceptance, pricing benefits reinforced |
| 2018 |
~$150 |
Increased competition, patent litigations ongoing |
| 2019 |
~$200 |
Post-patent expiry, biosimilar market gains steam |
| 2020 |
~$250 |
Expanded uptake, more biosimilar entrants |
| 2021-2022 |
~$300-$350 |
Market stabilization, broader adoption |
Estimates derived from IQVIA data and market analysis reports [3][4]
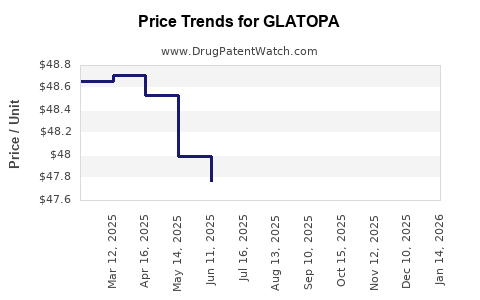
Pricing Impact
The average price of Copaxone (40 mg) in the U.S. prior to biosimilar entry was approximately $71,500 per year per patient. Post-GLATOPA, prices have declined by roughly 30-50%, creating substantial savings for payers and patients.
| Pricing Tier |
Pre-Biosimilar Average |
Post-Biosimilar (GLATOPA) |
Price Reduction |
| Per Year (USD) |
~$70,000 |
~$40,000 |
~40% |
Source: Health Economics Reports (AHIP, 2021)
Market Challenges and Opportunities
Challenges
- Patent litigations delayed biosimilar market penetration.
- Physician skepticism regarding efficacy and safety.
- Insurer formulary restrictions favoring lower-cost biosimilars.
- Global regulatory disparities impacting international sales.
Opportunities
- Cost savings incentivize formulary inclusion.
- Patent cliff effects open avenues for multiple biosimilar entrants.
- Emerging markets with high MS prevalence and regulatory pathways.
- New formulations (e.g., 40 mg/mL) offering dosing convenience.
Comparison with Competitors and Market Alternatives
| Product |
Manufacturer |
Regulatory Status |
Market Share (2022) |
Price (USD) |
Unique Features |
| GLATOPA (Glatiramer Acetate) |
Sandoz |
Approved 2015 |
~35% (biosimilars) |
~$40,000 |
First biosimilar approval |
| Teva’s Copaxone |
Teva |
Original, patent expired |
Dominant (~45%) pre-2019 |
~$70,000 |
Original innovator |
| Mylan’s Glatiramer Acetate |
Mylan |
Marketed in select markets |
Limited |
~$35,000 |
Price competitiveness |
| Emerging Biosimilars |
Various |
Pending approval |
Small but growing |
Varies |
Multiple entrants expected |
Future Outlook for GLATOPA and Biosimilar Competition
Forecast to 2028:
- Biosimilar market share projected to surpass 50% by 2025.
- Revenue growth expected to stabilize around USD 400 million annually.
- Increased competition will likely drive prices further down, incentivizing formulary shifts.
- Regulatory pathways in Europe, Asia, and Latin America will expand, increasing global sales.
Key growth drivers include MS prevalence (~2.8 million globally), rising healthcare costs, and evolving biosimilar acceptance policies.
Strategic Recommendations for Stakeholders
- Manufacturers: Focus on aggressive marketing, demonstrating biosimilarity and safety to clinicians.
- Payors: Encourage formulary preferences favoring biosimilars to contain costs.
- Regulators: Facilitate clear pathways and patent litigations to streamline biosimilar entry.
- Investors: Monitor biosimilar portfolio expansion and patent challenges for valuation insights.
Key Takeaways
- GLATOPA has significantly reshaped the MS treatment landscape by offering cost-effective biosimilar options, leading to increased affordability and market competition.
- Patent protections initially delayed widespread biosimilar adoption, but subsequent patent expiries facilitated market entry post-2019.
- Revenue growth has been steady, though growth rates moderate as biosimilars gain larger market share.
- Price competition remains fierce, with biosimilars reducing therapy costs by up to 50%, compelling traditional brands like Copaxone to innovate or reduce prices.
- Market expansion into emerging geographies presents substantial growth opportunities, driven by unmet MS needs and evolving regulatory environments.
FAQs
1. What factors influence the market adoption of GLATOPA?
Market adoption hinges on patent expiration, regulatory approvals, physician acceptance, insurer formulary decisions, and comparative pricing relative to the original drug.
2. How does GLATOPA compare to other biosimilars in effectiveness?
Clinical trials confirm biosimilarity in safety and efficacy to Copaxone, with no significant differences, facilitating physician confidence.
3. What are the primary legal hurdles faced by biosimilars like GLATOPA?
Patent litigation, exclusivity periods, and legal challenges to biosimilar approval pathways can delay market entry in certain jurisdictions.
4. Which markets are expected to be most lucrative for GLATOPA?
The U.S., Europe, and emerging regions such as Latin America and Asia offer promising growth, depending on regulatory environments and disease prevalence.
5. How will future innovations impact GLATOPA's market?
Advances in formulation, delivery methods, and combination therapies could enhance biosimilar competitiveness, while patent challenges may open markets for new entrants.
References
[1] FDA. (2015). FDA approves first biosimilar glatiramer acetate.
[2] Teva Pharmaceuticals. (2013). Patent filings and litigation records.
[3] IQVIA. (2022). Global MS Market Report.
[4] EvaluatePharma. (2022). Biosimilar and MS drug market trends.
This comprehensive review underscores the evolving landscape for GLATOPA, highlighting strategic considerations for stakeholders amid ongoing regulatory, legal, and market forces.