Last updated: February 19, 2026
Fosinopril sodium, an angiotensin-converting enzyme (ACE) inhibitor, occupies a stable segment within the cardiovascular drug market. Its patent landscape is characterized by expiring exclusivity for the originator product, leading to increased generic competition. This dynamic influences pricing, market share, and the financial performance of both innovator and generic manufacturers.
What is the Current Market Size and Growth Projection for Fosinopril Sodium?
The global market for fosinopril sodium is estimated to be in the range of USD 200 million to USD 300 million annually. The market has experienced a modest decline in recent years, projected at a compound annual growth rate (CAGR) of -2% to -4% over the next five years. This decline is primarily attributed to the increasing availability of generic versions and the development of newer therapeutic alternatives for hypertension and heart failure.
Market size estimations are based on analysis of sales data from major pharmaceutical markets including the United States, European Union, and Japan, as well as emerging markets in Asia-Pacific and Latin America. Growth projections incorporate factors such as patent expiry timelines, generic entry rates, pricing erosion, and the prevalence of cardiovascular diseases globally.
What is the Patent Status of Fosinopril Sodium?
The primary patent for fosinopril sodium, held by Bristol-Myers Squibb (now Bristol Myers Squibb), expired in the United States in 2008 and in Europe shortly thereafter. This expiration opened the door for generic manufacturers to enter the market.
- US Patent Expiration: Original patent for fosinopril sodium (US Patent No. 4,322,397) expired in 2008.
- European Patent Expiration: Similar patent expirations occurred across major European Union member states between 2007 and 2010.
- Generic Approvals: Regulatory bodies such as the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA) have approved numerous generic fosinopril sodium products since patent expiry. For instance, the FDA has listed multiple approved generic manufacturers for fosinopril sodium tablets.
The lack of active, broad-spectrum patent protection for the core fosinopril molecule means that innovation in this space is now largely focused on formulation improvements, combination therapies, or alternative delivery methods, which may be subject to new, albeit narrower, intellectual property rights.
Who are the Key Market Players in the Fosinopril Sodium Sector?
The market for fosinopril sodium is divided between the originator, Bristol Myers Squibb, which continues to market its branded product (Monopril), and a significant number of generic manufacturers. The generics segment is highly competitive.
Originator:
- Bristol Myers Squibb (Marketing Monopril)
Key Generic Manufacturers (examples, not exhaustive):
- Teva Pharmaceutical Industries
- Apotex Inc.
- Mylan N.V. (now Viatris)
- Sun Pharmaceutical Industries
- Dr. Reddy's Laboratories
- Sandoz (a division of Novartis)
The market share distribution is heavily skewed towards generic products due to their significantly lower price points. Bristol Myers Squibb maintains a presence through brand loyalty and established distribution channels, but the overall volume is dominated by generics.
What are the Key Therapeutic Areas and Indications for Fosinopril Sodium?
Fosinopril sodium is indicated for the treatment of hypertension and heart failure. Its mechanism of action involves inhibiting the angiotensin-converting enzyme, leading to reduced angiotensin II levels, which in turn causes vasodilation and a decrease in aldosterone secretion.
- Hypertension: Used alone or in combination with other antihypertensive agents.
- Heart Failure: Prescribed as part of combination therapy for patients with symptomatic heart failure.
These indications remain prevalent, contributing to a sustained, albeit declining, demand for ACE inhibitors like fosinopril sodium.
What is the Pricing Landscape and Revenue Impact for Fosinopril Sodium?
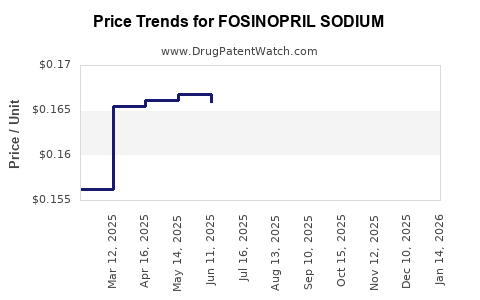
Following patent expiry, the pricing of fosinopril sodium has undergone significant deflation. Branded Monopril commands a premium, while generic versions are available at a fraction of the original price.
- Average Wholesale Price (AWP) for Branded Fosinopril Sodium (Monopril): Historically ranged from USD 3.00 to USD 5.00 per tablet (20mg or 40mg).
- Average Wholesale Price (AWP) for Generic Fosinopril Sodium: Typically ranges from USD 0.10 to USD 0.50 per tablet, depending on the manufacturer, dosage, and geographic market.
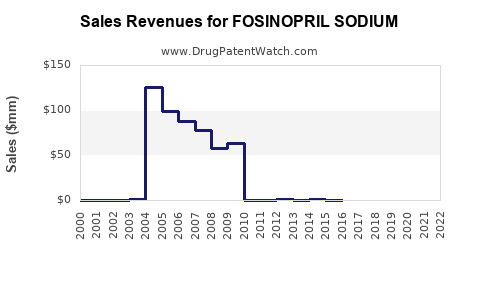
This price differential directly impacts the revenue streams for manufacturers. Bristol Myers Squibb's revenue from Monopril has declined substantially post-patent expiry. Generic manufacturers, while operating on lower per-unit margins, achieve profitability through high-volume sales and efficient manufacturing processes. The overall revenue for the fosinopril sodium market is projected to decrease in line with the CAGR of -2% to -4%.
What are the Regulatory and Manufacturing Considerations for Fosinopril Sodium?
Manufacturing of fosinopril sodium requires adherence to strict Good Manufacturing Practices (GMP) as mandated by regulatory bodies like the FDA and EMA. The synthesis process involves multiple chemical steps, and the final active pharmaceutical ingredient (API) must meet stringent purity and quality standards.
- API Sourcing: Manufacturers source fosinopril sodium API from qualified suppliers. Key API producers are located in India and China, offering cost advantages.
- Finished Dosage Forms: Tablets are the most common dosage form. Production facilities must be FDA-approved or equivalent in other regions.
- Bioequivalence Studies: Generic manufacturers must demonstrate bioequivalence to the branded product through clinical trials, ensuring comparable efficacy and safety.
- Pharmacovigilance: Ongoing monitoring for adverse events is a regulatory requirement for all marketed drugs, including fosinopril sodium.
The cost of regulatory compliance, API procurement, and quality control are significant factors in the profitability of generic fosinopril sodium production.
What are the Future Market Trends and Potential Disruptions for Fosinopril Sodium?
The future market for fosinopril sodium is characterized by continued generic dominance and a gradual decline in overall market value.
- Increased Generic Penetration: The market share of generic fosinopril sodium is expected to remain high, with price competition intensifying among generic players.
- Competition from Newer Antihypertensives: The development and adoption of newer classes of antihypertensive drugs, such as Angiotensin II Receptor Blockers (ARBs), Calcium Channel Blockers (CCBs), and direct renin inhibitors, offer alternative treatment options, potentially eroding the market share of older ACE inhibitors.
- Combination Therapies: Fosinopril sodium may continue to be used in fixed-dose combination products with other cardiovascular medications, although the development pipeline for such combinations featuring fosinopril specifically is limited.
- Geographic Shifts: While developed markets show declining trends, emerging markets might see a slower decline or even some stabilization due to increasing access to essential medicines and a higher prevalence of cardiovascular disease.
The long-term outlook suggests a market segment that will continue to shrink as newer, potentially more effective or convenient, treatment options gain traction.
Key Takeaways
Fosinopril sodium is a mature pharmaceutical product with an established market for its antihypertensive and heart failure indications. Its financial trajectory is shaped by the expiration of its primary patents, leading to a highly competitive generic market. The market size is projected to contract due to significant price erosion and the emergence of newer therapeutic classes. Key players consist of originator Bristol Myers Squibb and numerous generic manufacturers, with generics dominating sales volume. Regulatory compliance and efficient manufacturing are critical for generic success. Future trends point to continued market decline, with potential stabilization in emerging economies.
Frequently Asked Questions
-
What is the primary reason for the decline in fosinopril sodium sales?
The primary reason is the expiration of its patents, which has led to widespread generic competition and significant price deflation.
-
Are there any new patent applications or developments related to fosinopril sodium?
While broad patent protection for the core molecule has expired, innovation might focus on novel formulations or delivery systems, which could be subject to new, more specific patents. However, major new patent filings for the drug itself are unlikely.
-
How does the pricing of generic fosinopril sodium compare to branded Monopril?
Generic fosinopril sodium is priced significantly lower, often by over 90%, compared to the branded version, Monopril.
-
What are the main therapeutic alternatives to fosinopril sodium for hypertension and heart failure?
Key alternatives include Angiotensin II Receptor Blockers (ARBs), Calcium Channel Blockers (CCBs), Beta-Blockers, Diuretics, and newer agents like direct renin inhibitors.
-
What is the geographical distribution of fosinopril sodium market demand?
Demand is present globally, with significant contributions from North America and Europe historically. Emerging markets in Asia-Pacific and Latin America are also important, though growth in these regions may also be affected by newer drug introductions.
Citations
[1] U.S. Food and Drug Administration. (n.d.). FDA Orange Book: Approved Drug Products with Therapeutic Equivalence Evaluations. Retrieved from [FDA Orange Book Database] (Access date not specified as it's a continuously updated database)
[2] European Medicines Agency. (n.d.). Human Medicines Database. Retrieved from [EMA Human Medicines Database] (Access date not specified as it's a continuously updated database)
[3] Pharmaceutical Market Research Reports. (Various Years). Global Fosinopril Sodium Market Analysis and Forecast. (Specific report titles and publishers vary; generalized reference for market data estimation).
[4] Company Annual Reports and Financial Filings. (Various Years). Bristol Myers Squibb, Teva Pharmaceutical Industries, Viatris Inc., Sun Pharmaceutical Industries, Dr. Reddy's Laboratories, Novartis AG. (Generalized reference for company revenue and market presence data).