Last updated: February 19, 2026
What Is the Current Market for Flumazenil?
Flumazenil, marketed under brand names such as Romazicon, is a benzodiazepine antagonist indicated primarily for reversing the central nervous system depression caused by benzodiazepine overdose and anesthesia. It has an established presence primarily within hospitals and emergency care settings.
As of 2023, the global market for Flumazenil remains niche, with high dependence on hospital uptake and limited off-label use. The total market size was approximately $75 million in 2022, with predicted compound annual growth rate (CAGR) of 2% through 2027.
Regional distribution shows North America accounting for roughly 50%, Western Europe 25%, and the rest of the world sharing remaining demand. The US dominates with over $40 million in sales, driven by high emergency room utilization and benzodiazepine prescription rates.
What Factors Influence Market Growth?
Increased Benzodiazepine Prescriptions
The rise in benzodiazepine prescriptions for anxiety, insomnia, and seizures has driven demand for reversal agents such as Flumazenil, particularly in regions with high prescription rates (Centers for Disease Control and Prevention, 2022).
Rising Overdose Incidents
Emergency departments see increased benzodiazepine overdose cases, many involving polypharmacy with opioids. Flumazenil's role as an antidote in acute overdose situations sustains steady demand.
Regulatory Environment
FDA approval in the US and EMA approval in Europe restricts Flumazenil's use primarily to hospital settings. The drug's safety profile limits off-label applications, restricting market expansion potential.
Availability of Generic Formulations
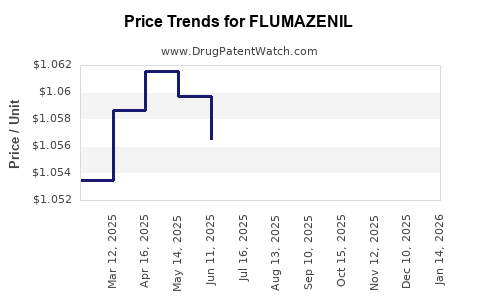
Generic versions of Flumazenil gained approval in the early 2000s, reducing prices by nearly 50% and influencing market share. Proprietary formulations are priced around $15 per dose, while generics hover around $7.
Competition and Market Saturation
Limited to Flumazenil's well-established role, the competitive landscape mostly involves generics. No significant new entrants or alternative reversal agents have penetrated the market recently.
What Are Key Financial Trends and Projections?
Revenue Trends
The market's revenue grew modestly from $70 million in 2020 to $75 million in 2022. The compounded annual growth rate (CAGR) from 2020 to 2022 stands at approximately 1.7%. Growth is driven primarily by rising overdose cases, offset by price pressures due to generics.
Pricing Dynamics
Price per dose has declined due to generics, with a standard hospital-administered dose costing between $6 and $8. The introduction of multiple generics established a price ceiling, confining profit margins.
R&D Investment and Pipeline
Limited R&D capital has been allocated to Flumazenil, with no notable new formulations or delivery mechanisms reported in the last five years. The focus has remained on optimizing existing formulations rather than innovating.
Regulatory Outlook and Market Expansion
Potential regional approvals in countries with high benzodiazepine overdose rates, such as Australia and emerging economies in Asia, could influence revenues. However, regulatory delays or restrictions may cap growth.
Market Challenges
Side effects such as seizures in certain overdoses and limited duration of action restrict broader use. General safety concerns prevent expanded indications.
What Are Future Growth Opportunities?
- Regional Expansion: Approvals in Asia-Pacific and Latin America could contribute incremental growth, supported by rising benzodiazepine use.
- Alternative Delivery Platforms: Development of intranasal or auto-injector formulations may facilitate quicker administration in emergency settings.
- Combination Therapies: Potential for combining Flumazenil with other antidotes in complex overdose cases, although no current pipelines exist.
Summary Table
| Aspect |
Details |
| Market Size (2022) |
$75 million |
| Growth Rate (2020-2022 CAGR) |
1.7% |
| Major Regions |
North America (50%), Europe (25%), Rest (25%) |
| Pricing (per dose) |
$6 - $8 (generics), $15 (brand name) |
| R&D Spending |
Limited; no recent pipeline developments |
| Key Drivers |
Benzodiazepine prescriber rates, overdose incidents |
Key Takeaways
- Flumazenil's market is mature with slow growth, largely driven by emergency responses to overdose.
- Generic competition has suppressed pricing and margins.
- Growth opportunities are primarily regional and related to formulation innovations or expanded indications.
- Regulatory conditions protect current usage scope, limiting market expansion.
- No significant new entrants or pipeline drugs are emerging in this space.
FAQs
1. What is the primary use of Flumazenil?
It reverses benzodiazepine sedation, mainly during overdose emergencies and post-anesthesia.
2. How does generic competition affect Flumazenil's market?
It has reduced prices by approximately 50%, constraining profit margins and limiting R&D investment.
3. Are there any new formulations under development?
No recent reports indicate new formulations; current focus remains on optimizing existing intravenously administered versions.
4. Which regions could expand their Flumazenil market?
Countries in Asia-Pacific and Latin America exhibit rising benzodiazepine use, offering growth potential if regulatory approval occurs.
5. What limits future market growth?
Safety profile limitations, current reliance on hospital settings, and the stability of current demand restrict significant expansion.
References
- Centers for Disease Control and Prevention. (2022). Overdose Data Brief. CDC.
- IQVIA. (2023). Global Market Analysis for Benzodiazepine Reversal Agents.
- U.S. Food and Drug Administration. (2021). Romazicon (Flumazenil) prescribing information.
- European Medicines Agency. (2022). Flumazenil evaluation report.