Last updated: February 19, 2026
FILSPARI (triptans) is a serotonin 5-HT1B/1D receptor agonist approved for the acute treatment of migraine with or without aura in adults. Its market performance is influenced by the competitive landscape of migraine therapies, prescription trends, and the patent protection timeline.
What is FILSPARI's Current Market Position?
FILSPARI was approved by the U.S. Food and Drug Administration (FDA) on January 15, 2020 [1]. It is marketed by AbbVie Inc. and is a prescription medication. The drug targets the CGRP pathway, a key mechanism in migraine pathophysiology, offering a novel approach compared to older treatments.
The migraine market is characterized by a significant unmet need, with millions of individuals experiencing debilitating headaches. While triptans and CGRP inhibitors (both monoclonal antibodies and small molecules) represent the primary treatment classes, the demand for effective and well-tolerated options continues to drive market growth. FILSPARI competes with established triptans and a growing number of CGRP-targeting drugs.
Key Market Competitors:
- Triptans: Sumatriptan (Imitrex), Rizatriptan (Maxalt), Eletriptan (Relpax), Zolmitriptan (Zomig). These are widely used and often available as generics.
- CGRP Inhibitors (Small Molecules): Ubrogepant (Ubrelvy), Rimegepant (Nurtec ODT). These orally administered drugs offer an alternative mechanism of action to triptans.
- CGRP Inhibitors (Monoclonal Antibodies): Erenumab (Aimovig), Fremanezumab (Ajovy), Galcanezumab (Emgality), Eptinezumab (Vyepti). These are primarily used for migraine prevention but represent a significant portion of the overall migraine therapeutic market.
FILSPARI's positioning as an acute treatment option in the crowded migraine space means its market share is contingent on its efficacy, safety profile, and patient/physician preference compared to existing and emerging alternatives. The drug's specific indication for acute treatment, rather than prevention, defines its role in patient care.
What are FILSPARI's Financial Performance Indicators?
Financial performance for FILSPARI is primarily reported through AbbVie's quarterly and annual earnings. As a relatively newer product, its revenue contribution is part of AbbVie's broader neuroscience portfolio. Direct, standalone revenue figures for FILSPARI may not always be publicly disclosed separately from other migraine therapies within AbbVie's reporting.
General Trends in Migraine Drug Revenue:
- Newer agents, particularly CGRP inhibitors, have seen rapid revenue growth due to their novel mechanism and perceived benefits for certain patient populations.
- Genericization of older triptans limits their revenue growth potential, making branded versions and novel formulations or mechanisms more attractive for market capture.
- Market access and reimbursement policies play a crucial role in determining a drug's commercial success. Physicians and payers evaluate efficacy, safety, and cost-effectiveness.
To assess FILSPARI's specific financial trajectory, detailed analysis of AbbVie's investor reports and pharmaceutical market research data is required. Key metrics to monitor would include:
- Net Sales: The total revenue generated from FILSPARI sales.
- Prescription Volume: The number of new and total prescriptions dispensed.
- Market Share: FILSPARI's percentage of the acute migraine treatment market.
- Reimbursement Status: Approval by major insurance providers and formulary placement.
As of the latest available data, specific quarterly or annual revenue figures solely attributed to FILSPARI are not consistently broken out in public financial statements. However, AbbVie's neuroscience segment, which includes FILSPARI, contributes significantly to the company's overall revenue. For instance, in Q1 2024, AbbVie reported net revenues of $14.4 billion, with its neuroscience portfolio a key driver [2].
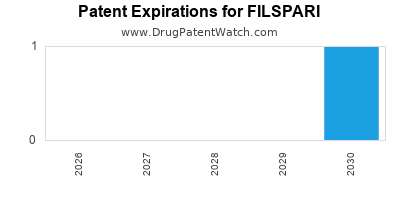
What is the Patent Landscape for FILSPARI?
The patent protection for FILSPARI is a critical determinant of its long-term market exclusivity and revenue potential. Pharmaceutical patents typically grant exclusivity for 20 years from the filing date, though extensions and other regulatory exclusivities can alter this timeline.
Key Patent Considerations for Pharmaceuticals:
- Composition of Matter Patents: These protect the drug molecule itself.
- Method of Use Patents: These protect specific indications or ways of administering the drug.
- Formulation Patents: These protect the specific delivery system or dosage form.
- Evergreening: Strategies employed by pharmaceutical companies to extend patent life through new patents on minor modifications, new formulations, or new uses.
Information on FILSPARI's specific patent portfolio is publicly available through patent databases. However, the complex nature of patent law, including potential litigation, inter partes reviews, and patent term extensions, means that the effective market exclusivity period can vary.
A preliminary search of U.S. Patent and Trademark Office (USPTO) records and other patent databases would reveal granted patents and pending applications related to FILSPARI. These would include patents covering the active pharmaceutical ingredient, its synthesis, and its use in treating migraine.
Example of Patent Timeline Impacts:
A drug with a strong composition of matter patent expiring in 2030 would face generic competition soon thereafter, significantly impacting its branded sales. Conversely, if key method of use patents for specific patient populations or a novel delivery system extend beyond the composition of matter patent, this can provide extended market protection.
For FILSPARI, understanding the expiry dates of its foundational patents is essential for forecasting future revenue streams and identifying potential generic entry points. Investors and R&D strategists must monitor patent litigation closely, as legal challenges can accelerate or delay generic competition.
How do Market Access and Reimbursement Influence FILSPARI's Adoption?
Market access and reimbursement are crucial gatekeepers for any new pharmaceutical product. For FILSPARI, its uptake by physicians and patients is directly tied to its inclusion on insurance formularies and the out-of-pocket costs for patients.
Factors Influencing Market Access:
- Clinical Efficacy and Safety Data: Robust data demonstrating superior or comparable efficacy to existing treatments, with an acceptable safety profile, is foundational.
- Health Technology Assessments (HTAs): Independent bodies in some countries assess the cost-effectiveness of new drugs.
- Payer Negotiations: Pharmaceutical companies negotiate pricing and reimbursement terms with health insurers and government payers.
- Patient Assistance Programs: Programs designed to reduce out-of-pocket costs for patients can increase affordability and access.
Reimbursement Landscape for Migraine Treatments:
- Triptans: Many triptans are available as generics, making them cost-effective options. Branded versions may face formulary restrictions or require prior authorization.
- CGRP Inhibitors: These newer agents, particularly monoclonal antibodies, are often associated with higher costs. Their reimbursement can be subject to strict criteria, such as failure of prior treatments or specific migraine frequency thresholds. Oral CGRP inhibitors generally have broader access but still involve co-pays.
FILSPARI's reimbursement status with major U.S. payers (e.g., Medicare, Medicaid, commercial insurers) is a key indicator of its market penetration. A favorable formulary position, with low co-pays for patients, would facilitate higher prescription volumes. Conversely, significant prior authorization requirements or high patient out-of-pocket costs can create barriers to access and limit the drug's commercial success.
Data to Monitor:
- Payer coverage announcements.
- Formulary tier placement (e.g., Tier 1, Tier 2).
- Prior authorization requirements.
- Average patient out-of-pocket cost.
This information is typically found in AbbVie's investor relations materials, commercial updates, and reports from market access analytics firms.
What are the Future Projections for FILSPARI's Revenue?
Projecting FILSPARI's future revenue involves assessing its competitive positioning, patent life, market growth potential, and evolving treatment paradigms.
Key Drivers for Future Revenue:
- Market Growth: The global migraine market is projected to grow, driven by increasing prevalence, improved diagnostics, and the development of novel therapies.
- Competitive Dynamics: The introduction of new migraine treatments, including those with different mechanisms of action or improved delivery methods, will influence FILSPARI's market share.
- Patent Expiry: The impending expiry of key patents will open the door for generic competition, leading to a significant decline in branded revenue.
- Label Expansion: If FILSPARI receives approval for additional indications or patient populations, its revenue potential could increase.
- Geographic Expansion: Launching FILSPARI in international markets would broaden its revenue base.
Forecasting Methodologies:
- Market Share Projections: Estimating FILSPARI's future market share based on its strengths, weaknesses, and competitive environment.
- Sales Volume Trends: Analyzing historical prescription data and projecting future trends.
- Pricing Power: Assessing the ability to maintain or increase pricing, factoring in competition and payer pressure.
- Patent Cliff Analysis: Quantifying the revenue loss expected upon patent expiry.
Without direct, publicly disclosed sales figures for FILSPARI, detailed revenue forecasts are speculative and rely on broader market data and assumptions about AbbVie's commercial strategy. However, as a branded pharmaceutical with a defined patent life, FILSPARI's revenue trajectory will likely follow a pattern of growth during its period of market exclusivity, followed by a sharp decline upon generic entry.
Companies specializing in pharmaceutical market intelligence provide proprietary forecasts. These typically incorporate detailed competitor analysis, physician prescribing habits, patient adherence rates, and patent expiry timelines. For R&D and investment decisions, such detailed projections are indispensable.
Key Takeaways
FILSPARI operates within the dynamic and growing migraine treatment market, facing competition from established triptans and newer CGRP inhibitors. Its market position is contingent on demonstrated clinical value, physician adoption, and favorable market access and reimbursement. The drug's financial trajectory is intrinsically linked to its patent exclusivity period; revenue is expected to grow during this period and decline significantly upon generic entry.
Frequently Asked Questions
-
What is the primary mechanism of action for FILSPARI?
FILSPARI is a serotonin 5-HT1B/1D receptor agonist.
-
When was FILSPARI approved by the FDA?
FILSPARI received FDA approval on January 15, 2020.
-
Which company markets FILSPARI?
AbbVie Inc. markets FILSPARI.
-
What is the typical duration of patent exclusivity for a new drug like FILSPARI?
Patent protection generally extends for 20 years from the filing date, though extensions and other exclusivities can modify this.
-
Does FILSPARI have any approved indications beyond acute migraine treatment?
As of current public information, FILSPARI is approved for the acute treatment of migraine with or without aura in adults.
Citations
[1] U.S. Food and Drug Administration. (2020, January 15). FDA approves FILSPARI (triptans) for the acute treatment of migraine. [Press Release].
[2] AbbVie Inc. (2024, April 26). AbbVie announces first quarter 2024 financial results. [Press Release].