EVOMELA Drug Patent Profile
✉ Email this page to a colleague
Which patents cover Evomela, and what generic alternatives are available?
Evomela is a drug marketed by Acrotech Biopharma and is included in one NDA. There are seven patents protecting this drug and one Paragraph IV challenge.
This drug has one hundred and five patent family members in twenty-two countries.
The generic ingredient in EVOMELA is melphalan hydrochloride. There are twelve drug master file entries for this compound. Sixteen suppliers are listed for this compound. Additional details are available on the melphalan hydrochloride profile page.
DrugPatentWatch® Litigation and Generic Entry Outlook for Evomela
A generic version of EVOMELA was approved as melphalan hydrochloride by MYLAN INSTITUTIONAL on June 9th, 2009.
AI Deep Research
Questions you can ask:
- What is the 5 year forecast for EVOMELA?
- What are the global sales for EVOMELA?
- What is Average Wholesale Price for EVOMELA?
Summary for EVOMELA
| International Patents: | 105 |
| US Patents: | 7 |
| Applicants: | 1 |
| NDAs: | 1 |
| Finished Product Suppliers / Packagers: | 1 |
| Raw Ingredient (Bulk) Api Vendors: | 32 |
| Clinical Trials: | 21 |
| Patent Applications: | 1,752 |
| Drug Prices: | Drug price information for EVOMELA |
| Patent Litigation and PTAB cases: | See patent lawsuits and PTAB cases for EVOMELA |
| What excipients (inactive ingredients) are in EVOMELA? | EVOMELA excipients list |
| DailyMed Link: | EVOMELA at DailyMed |
Recent Clinical Trials for EVOMELA
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| Ehsan Malek | Early Phase 1 |
| Washington University School of Medicine | Phase 1 |
| CASI Pharmaceuticals (China) Co., Ltd. | PHASE4 |
Pharmacology for EVOMELA
| Drug Class | Alkylating Drug |
| Mechanism of Action | Alkylating Activity |
Paragraph IV (Patent) Challenges for EVOMELA
| Tradename | Dosage | Ingredient | Strength | NDA | ANDAs Submitted | Submissiondate |
|---|---|---|---|---|---|---|
| EVOMELA | Powder for Injection | melphalan hydrochloride | 50 mg/vial | 207155 | 1 | 2017-09-08 |
US Patents and Regulatory Information for EVOMELA
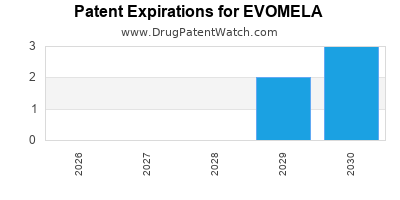
EVOMELA is protected by seven US patents.
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Acrotech Biopharma | EVOMELA | melphalan hydrochloride | POWDER;INTRAVENOUS | 207155-001 | Mar 10, 2016 | RX | Yes | Yes | 10,940,128 | ⤷ Start Trial | Y | ⤷ Start Trial | |||
| Acrotech Biopharma | EVOMELA | melphalan hydrochloride | POWDER;INTRAVENOUS | 207155-001 | Mar 10, 2016 | RX | Yes | Yes | 8,410,077 | ⤷ Start Trial | Y | ⤷ Start Trial | |||
| Acrotech Biopharma | EVOMELA | melphalan hydrochloride | POWDER;INTRAVENOUS | 207155-001 | Mar 10, 2016 | RX | Yes | Yes | 10,040,872 | ⤷ Start Trial | Y | ⤷ Start Trial | |||
| Acrotech Biopharma | EVOMELA | melphalan hydrochloride | POWDER;INTRAVENOUS | 207155-001 | Mar 10, 2016 | RX | Yes | Yes | 11,020,363 | ⤷ Start Trial | Y | ⤷ Start Trial | |||
| Acrotech Biopharma | EVOMELA | melphalan hydrochloride | POWDER;INTRAVENOUS | 207155-001 | Mar 10, 2016 | RX | Yes | Yes | 10,864,183 | ⤷ Start Trial | Y | ⤷ Start Trial | |||
| Acrotech Biopharma | EVOMELA | melphalan hydrochloride | POWDER;INTRAVENOUS | 207155-001 | Mar 10, 2016 | RX | Yes | Yes | 9,493,582 | ⤷ Start Trial | Y | ⤷ Start Trial | |||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
International Patents for EVOMELA
See the table below for patents covering EVOMELA around the world.
| Country | Patent Number | Title | Estimated Expiration |
|---|---|---|---|
| European Patent Office | 2434886 | COMPOSITIONS INJECTABLES À BASE DE MELPHALAN COMPRENANT UN DÉRIVÉ DE CYCLODEXTRINE ET LEURS PROCÉDÉS DE FABRICATION ET D'UTILISATION (INJECTABLE MELPHALAN COMPOSITIONS COMPRISING A CYCLODEXTRIN DERIVATIVE AND METHODS OF MAKING AND USING THE SAME) | ⤷ Start Trial |
| World Intellectual Property Organization (WIPO) | 2009134347 | ⤷ Start Trial | |
| South Korea | 20200106100 | 알킬화된 시클로덱스트린 조성물 및 이의 제조 및 사용 방법 (Alkylated Cyclodextrin Compositions and Processes for Preparing and using the same) | ⤷ Start Trial |
| Israel | 243654 | תכשירי סולפואלקיל אתר ציקלודקסטרין (Sulfoalkyl ether cyclodextrin compositions) | ⤷ Start Trial |
| China | 110066351 | ⤷ Start Trial | |
| Eurasian Patent Organization | 201590617 | ⤷ Start Trial | |
| >Country | >Patent Number | >Title | >Estimated Expiration |
Supplementary Protection Certificates for EVOMELA
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 2701720 | 23C1000 | France | ⤷ Start Trial | PRODUCT NAME: MELPHALAN FLUFENAMIDE ET SES SELS PHARMACEUTIQUEMENT ACCEPTABLES, TELS QUE LE CHLORHYDRATE; REGISTRATION NO/DATE: EU/1/22/1669 20220818 |
| 2701720 | CA 2022 00054 | Denmark | ⤷ Start Trial | PRODUCT NAME: MELPHALAN FLUFENAMIDE HYDROCHLORIDE; REG. NO/DATE: EU/1/22/1669 20220818 |
| 2701720 | 202240050 | Slovenia | ⤷ Start Trial | PRODUCT NAME: MELPHALAN FLUFENAMIDE HYDROCHLORIDE; NATIONAL AUTHORISATION NUMBER: EU/1/22/1669/001; DATE OF NATIONAL AUTHORISATION: 20220817; AUTHORITY FOR NATIONAL AUTHORISATION: EU |
| 2701720 | 122023000007 | Germany | ⤷ Start Trial | PRODUCT NAME: MELPHALANFLUFENAMID HYDROCHLORID; REGISTRATION NO/DATE: EU/1/22/1669 20220817 |
| 2701720 | 2290047-6 | Sweden | ⤷ Start Trial | PRODUCT NAME: MELPHALAN FLUFENAMIDE HYDROCHLORIDE; REG. NO/DATE: EU/1/22/1669 20220818 |
| 2701720 | CR 2022 00054 | Denmark | ⤷ Start Trial | PRODUCT NAME: MELPHALAN FLUFENAMIDE HYDROCHLORIDE; REG. NO/DATE: EU/1/22/1669 20220818 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
EVOMELA (Melphalan Flufenamide) Market Dynamics and Financial Trajectory
More… ↓
Make Better Decisions: Try a trial or see plans & pricing
Drugs may be covered by multiple patents or regulatory protections. All trademarks and applicant names are the property of their respective owners or licensors. Although great care is taken in the proper and correct provision of this service, thinkBiotech LLC does not accept any responsibility for possible consequences of errors or omissions in the provided data. The data presented herein is for information purposes only. There is no warranty that the data contained herein is error free. We do not provide individual investment advice. This service is not registered with any financial regulatory agency. The information we publish is educational only and based on our opinions plus our models. By using DrugPatentWatch you acknowledge that we do not provide personalized recommendations or advice. thinkBiotech performs no independent verification of facts as provided by public sources nor are attempts made to provide legal or investing advice. Any reliance on data provided herein is done solely at the discretion of the user. Users of this service are advised to seek professional advice and independent confirmation before considering acting on any of the provided information. thinkBiotech LLC reserves the right to amend, extend or withdraw any part or all of the offered service without notice.