EVEKEO Drug Patent Profile
✉ Email this page to a colleague
When do Evekeo patents expire, and when can generic versions of Evekeo launch?
Evekeo is a drug marketed by Azurity and is included in two NDAs. There are three patents protecting this drug.
The generic ingredient in EVEKEO is amphetamine sulfate. There are fifty-five drug master file entries for this compound. Eleven suppliers are listed for this compound. Additional details are available on the amphetamine sulfate profile page.
DrugPatentWatch® Litigation and Generic Entry Outlook for Evekeo
A generic version of EVEKEO was approved as amphetamine sulfate by AMNEAL PHARMS on September 26th, 2018.
AI Deep Research
Questions you can ask:
- What is the 5 year forecast for EVEKEO?
- What are the global sales for EVEKEO?
- What is Average Wholesale Price for EVEKEO?
Summary for EVEKEO
| US Patents: | 0 |
| Applicants: | 1 |
| NDAs: | 2 |
| Finished Product Suppliers / Packagers: | 3 |
| Raw Ingredient (Bulk) Api Vendors: | 24 |
| Patent Applications: | 1,541 |
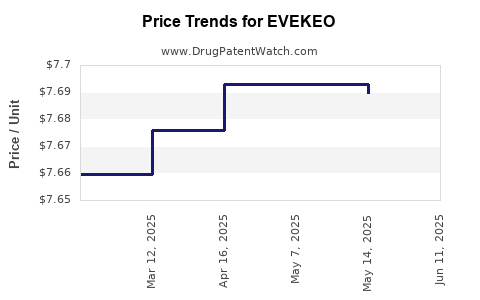
| Drug Prices: | Drug price information for EVEKEO |
| DailyMed Link: | EVEKEO at DailyMed |
Pharmacology for EVEKEO
| Drug Class | Central Nervous System Stimulant |
| Physiological Effect | Central Nervous System Stimulation |
US Patents and Regulatory Information for EVEKEO
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Azurity | EVEKEO | amphetamine sulfate | TABLET;ORAL | 200166-001 | Aug 9, 2012 | AA | RX | No | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | |||
| Azurity | EVEKEO ODT | amphetamine sulfate | TABLET, ORALLY DISINTEGRATING;ORAL | 209905-001 | Jan 30, 2019 | DISCN | Yes | No | 11,160,772 | ⤷ Start Trial | Y | ⤷ Start Trial | |||
| Azurity | EVEKEO ODT | amphetamine sulfate | TABLET, ORALLY DISINTEGRATING;ORAL | 209905-005 | Apr 16, 2021 | DISCN | Yes | No | 11,160,772 | ⤷ Start Trial | Y | ⤷ Start Trial | |||
| Azurity | EVEKEO ODT | amphetamine sulfate | TABLET, ORALLY DISINTEGRATING;ORAL | 209905-003 | Jan 30, 2019 | DISCN | Yes | No | 10,441,554 | ⤷ Start Trial | Y | ⤷ Start Trial | |||
| Azurity | EVEKEO ODT | amphetamine sulfate | TABLET, ORALLY DISINTEGRATING;ORAL | 209905-002 | Jan 30, 2019 | DISCN | Yes | No | 11,896,562 | ⤷ Start Trial | ⤷ Start Trial | ||||
| Azurity | EVEKEO ODT | amphetamine sulfate | TABLET, ORALLY DISINTEGRATING;ORAL | 209905-004 | Jan 30, 2019 | DISCN | Yes | No | 10,441,554 | ⤷ Start Trial | Y | ⤷ Start Trial | |||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Market Dynamics and Financial Trajectory for EVEKEO
More… ↓
