ESCITALOPRAM OXALATE Drug Patent Profile
✉ Email this page to a colleague
When do Escitalopram Oxalate patents expire, and what generic alternatives are available?
Escitalopram Oxalate is a drug marketed by Rising, Amneal Pharms, Antrim Pharms Llc, Aurobindo Pharma Ltd, Chartwell Molecular, Hetero Labs Ltd Iii, Macleods Pharms Ltd, Sun Pharma Canada, Accord Hlthcare, Aiping Pharm Inc, Ascent Pharms Inc, Chartwell Rx, Graviti Pharms, Hikma Pharms, Invagen Pharms, Jubilant Cadista, Novitium Pharma, Prinston Inc, Teva Pharms Usa, Torrent Pharms Ltd, and Zydus Lifesciences. and is included in twenty-four NDAs.
The generic ingredient in ESCITALOPRAM OXALATE is escitalopram oxalate. There are twenty-six drug master file entries for this compound. Forty-six suppliers are listed for this compound. Additional details are available on the escitalopram oxalate profile page.
DrugPatentWatch® Litigation and Generic Entry Outlook for Escitalopram Oxalate
A generic version of ESCITALOPRAM OXALATE was approved as escitalopram oxalate by AMNEAL PHARMS on March 14th, 2012.
AI Deep Research
Questions you can ask:
- What is the 5 year forecast for ESCITALOPRAM OXALATE?
- What are the global sales for ESCITALOPRAM OXALATE?
- What is Average Wholesale Price for ESCITALOPRAM OXALATE?
Summary for ESCITALOPRAM OXALATE
| US Patents: | 0 |
| Applicants: | 21 |
| NDAs: | 24 |
| Finished Product Suppliers / Packagers: | 44 |
| Raw Ingredient (Bulk) Api Vendors: | 91 |
| Clinical Trials: | 25 |
| Drug Prices: | Drug price information for ESCITALOPRAM OXALATE |
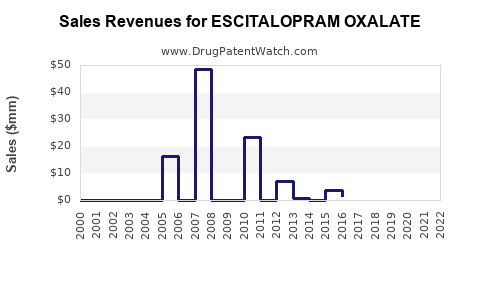
| Drug Sales Revenues: | Drug sales revenues for ESCITALOPRAM OXALATE |
| What excipients (inactive ingredients) are in ESCITALOPRAM OXALATE? | ESCITALOPRAM OXALATE excipients list |
| DailyMed Link: | ESCITALOPRAM OXALATE at DailyMed |
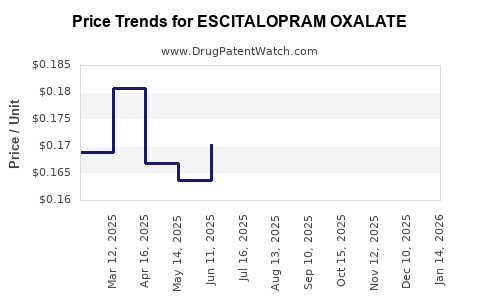
See drug prices for ESCITALOPRAM OXALATE
Recent Clinical Trials for ESCITALOPRAM OXALATE
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| Gang Wang | NA |
| Tianjin Anding Hospital | NA |
| Shandong Daizhuang Hospital | NA |
Pharmacology for ESCITALOPRAM OXALATE
| Drug Class | Serotonin Reuptake Inhibitor |
| Mechanism of Action | Serotonin Uptake Inhibitors |
Anatomical Therapeutic Chemical (ATC) Classes for ESCITALOPRAM OXALATE
Paragraph IV (Patent) Challenges for ESCITALOPRAM OXALATE
| Tradename | Dosage | Ingredient | Strength | NDA | ANDAs Submitted | Submissiondate |
|---|---|---|---|---|---|---|
| LEXAPRO | Capsules | escitalopram oxalate | 5 mg | 021323 | 1 | 2005-08-17 |
| LEXAPRO | Capsules | escitalopram oxalate | 10 mg and 20 mg | 021323 | 1 | 2005-03-30 |
US Patents and Regulatory Information for ESCITALOPRAM OXALATE
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Macleods Pharms Ltd | ESCITALOPRAM OXALATE | escitalopram oxalate | TABLET;ORAL | 202210-001 | Sep 11, 2012 | AB | RX | No | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | |||
| Hikma Pharms | ESCITALOPRAM OXALATE | escitalopram oxalate | TABLET;ORAL | 078766-001 | Sep 11, 2012 | DISCN | No | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | ||||
| Prinston Inc | ESCITALOPRAM OXALATE | escitalopram oxalate | TABLET;ORAL | 078032-003 | Aug 28, 2015 | AB | RX | No | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | |||
| Novitium Pharma | ESCITALOPRAM OXALATE | escitalopram oxalate | TABLET;ORAL | 077550-002 | May 14, 2015 | DISCN | No | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | ||||
| Teva Pharms Usa | ESCITALOPRAM OXALATE | escitalopram oxalate | TABLET;ORAL | 076765-001 | Mar 14, 2012 | DISCN | No | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | ||||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Market Dynamics and Financial Trajectory for Escitalopram Oxalate
More… ↓
