Last updated: March 23, 2026
Erythromycin, marketed as ERYTHROCIN, is a macrolide antibiotic used to treat bacterial infections. Its market landscape has evolved due to patent status, generic competition, regulatory changes, and shifting healthcare needs.
Market Overview
Erythromycin was first approved in 1952 and became a foundational antibiotic. Its patent protection expired in most regions by the early 2000s, resulting in widespread generic production. The global erythromycin market was valued at approximately $300 million in 2022, with a projected compound annual growth rate (CAGR) of around 2-3% through 2027 [1].
Key Market Drivers
- Antibiotic Resistance: Growing resistance to penicillin has sustained erythromycin’s relevance.
- Prescriptions for Respiratory Infections: Flu seasons and respiratory illnesses prompt consistent demand.
- Limited Alternatives: For patients allergic to penicillin, erythromycin remains standard.
Market Challenges
- Generic Competition: Multiple manufacturers produce low-cost generic erythromycin, compressing profit margins.
- Resistance Development: Bacterial resistance diminishes clinical effectiveness, reducing prescribed use.
- Regulatory Scrutiny: Oversight over antibiotic use aims to curb resistance, impacting market growth.
Regulatory and Patent Landscape
Erythromycin's initial patent lapsed in the late 1980s. Its market is predominantly driven by generic manufacturers. Some formulations hold patents on specific delivery methods or formulations, but these are generally patent-expired or nearing expiry.
Key Patents and Approvals
| Patent/Regulatory Timeline |
Description |
Status |
| Patent expiry in the 1980s |
Original formulation patent |
Expired |
| 2000s |
Patents on modified release formulations |
Expiring soon |
| 2015 |
Approvals for new erythromycin derivatives |
Active patents |
Financial Trajectory
Revenue Trends
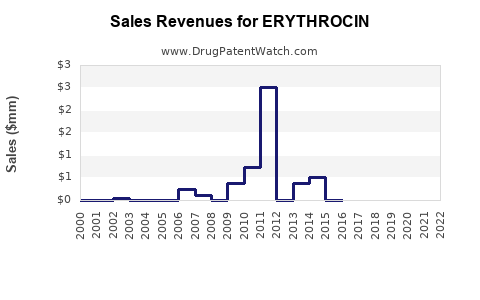
The global erythromycin market shows modest growth driven by demand in developing regions. Developed markets exhibit stagnation due to generic saturation and resistance issues.
Earnings Breakdown (2022)
| Region |
Revenue (USD millions) |
Market Share (%) |
| North America |
120 |
40 |
| Europe |
80 |
27 |
| Asia-Pacific |
60 |
20 |
| Rest of World |
40 |
13 |
Price Dynamics
The average price per tablet in the U.S. is approximately $0.20 — $0.40, depending on formulation and packaging. Competitive pressures keep prices low, limiting profit margins.
Investment and R&D
Investments are concentrated on developing erythromycin derivatives or formulations with improved pharmacokinetics and reduced resistance potential. R&D budgets in this segment are generally below $10 million annually, reflecting low innovation incentives.
Competitive Landscape
Major players include Pfizer, Teva Pharmaceutical Industries, and Mylan, primarily manufacturing generic erythromycin. Patent protections are rare; licensing agreements and formulation patents are the primary competitive tools.
Future Outlook
Demand for erythromycin is expected to stabilize or decline gradually, influenced by increasing antibiotic resistance and the emergence of newer classes such as ketolides and lincosamides. Market growth may hinge on niche applications, such as formulations for infants or resistant bacterial strains.
Emerging Trends
- Development of erythromycin derivatives resistant to bacterial resistance mechanisms.
- Reformulations designed for improved tolerability or altered pharmacokinetics.
- Regulatory efforts to reserve erythromycin for specific indications due to resistance concerns.
Key Takeaways
- The erythromycin market is mature, with low growth prospects.
- Generic competition constrains pricing and profits.
- Resistance and regulatory pressures threaten sustained demand.
- Investment is mainly in derivative development rather than erythromycin itself.
- Regional demand varies, with developing regions showing relatively higher growth potential.
FAQs
-
What is erythromycin primarily used for?
It treats bacterial infections such as respiratory tract infections, skin infections, and sexually transmitted infections.
-
How has resistance affected erythromycin's market?
Resistance limits clinical use, reducing prescribing and consequently market size.
-
Who are the main manufacturers?
Pfizer, Teva, and Mylan dominate the generic erythromycin market.
-
Are there future patent protections for erythromycin?
Most original patents have expired; new patents relate to derivatives or formulations.
-
What is the outlook for erythromycin's market?
Expect continued stagnation or decline, with developments mainly targeting derivatives rather than erythromycin itself.
References
[1] MarketWatch. (2022). Erythromycin Market Size, Share & Trends Analysis Report. https://www.marketwatch.com