Last updated: February 19, 2026
Entacapone, a catechol-O-methyltransferase (COMT) inhibitor, is a critical component in the treatment of Parkinson's disease. Its market trajectory is shaped by patent expirations, generic competition, and evolving therapeutic guidelines. This analysis outlines the current patent status, historical sales performance, and projected market evolution for entacapone.
What is Entacapone and Its Mechanism of Action?
Entacapone is a selective, reversible inhibitor of COMT. This enzyme is responsible for the peripheral metabolism of levodopa, the primary precursor to dopamine. By inhibiting COMT, entacapone prolongs the plasma half-life of levodopa, increasing its availability to the brain. This leads to improved motor function in patients with Parkinson's disease, particularly those experiencing motor fluctuations.
Entacapone is typically administered orally in combination with levodopa and carbidopa. The standard dosage is 200 mg per dose, taken at the same time as levodopa/carbidopa, up to five times daily. The pharmacological action of entacapone is crucial for managing "off" episodes, which are periods of worsening Parkinson's symptoms.
The therapeutic benefit of entacapone is directly linked to its pharmacokinetic and pharmacodynamic properties. By blocking COMT, it prevents the breakdown of levodopa in the periphery, ensuring a more consistent and sustained delivery of levodopa to the central nervous system. This mechanism underpins its efficacy in reducing motor fluctuations and improving the quality of life for Parkinson's patients.
What is the Patent Status of Entacapone?
The original patent for entacapone, held by Orion Corporation, has expired. The key compound patent, EP0371750B1, expired in Europe in 2008 and in the United States in 2007. This expiration paved the way for generic manufacturers to enter the market.
Several secondary patents related to formulation, manufacturing processes, and specific uses may still be in effect or have recently expired, but these generally offer less robust market exclusivity compared to compound patents. For instance, patents pertaining to fixed-dose combinations or specific polymorphic forms can influence manufacturing and market entry strategies for generic producers.
The expiration of the core compound patent has led to the availability of multiple generic versions of entacapone. This competition has significantly impacted pricing and market share dynamics. The primary patent landscape is characterized by the absence of strong, compound-level protection, allowing for widespread generic penetration.
A review of patent databases reveals ongoing activity around entacapone, primarily related to process improvements and new formulations. However, these are unlikely to restore the level of market exclusivity previously afforded by the expired compound patent. The focus for brand holders and new entrants is now on lifecycle management through differentiated product offerings or strategic market positioning rather than on blocking generic entry through core patent protection.
How Has Entacapone Performed Financially?
Entacapone's financial performance has been significantly influenced by its patent expiration and the subsequent rise of generic competition. The originator product, Comtan (Novartis) and Stalevo (a fixed-dose combination of levodopa, carbidopa, and entacapone, also by Novartis), experienced strong sales prior to and immediately following generic entry.
| Year |
Net Sales (USD Millions) |
Source |
| 2015 |
~750 |
[1] |
| 2016 |
~700 |
[1] |
| 2017 |
~650 |
[1] |
| 2018 |
~600 |
[1] |
| 2019 |
~550 |
[1] |
| 2020 |
~500 |
[1] |
Note: Figures represent global sales for entacapone-containing products, primarily Comtan and Stalevo. Sales data for generic entacapone are not comprehensively tracked in aggregate but contribute to the overall market value.
The decline in sales for the branded products is directly attributable to the erosion of market share by lower-cost generic alternatives. The market for entacapone is now largely driven by generic sales, with pricing being a primary competitive factor. The total market value, while difficult to precisely quantify due to the fragmented generic landscape, is sustained by the continued therapeutic need for entacapone in Parkinson's disease management.
The introduction of generic entacapone in major markets such as the United States and Europe occurred after the expiration of key patents. This led to significant price reductions and a substantial shift in prescription volume towards generic manufacturers. The fixed-dose combination product, Stalevo, also faced generic challenges, albeit with some delay due to its own patent and formulation complexities.
The financial trajectory demonstrates a typical pattern for pharmaceuticals post-patent expiry: an initial period of strong brand sales followed by a steep decline as generics gain market share. The overall market size for entacapone, considering both branded and generic sales, remains substantial due to its established role in Parkinson's therapy.
What are the Market Dynamics for Entacapone?
The market dynamics for entacapone are characterized by:
- Genericization: The primary driver is the widespread availability of generic entacapone. This has led to intense price competition among manufacturers.
- Therapeutic Indication: Entacapone remains a standard of care for managing motor fluctuations in Parkinson's disease, ensuring sustained demand.
- Competition with Other COMT Inhibitors: While entacapone is established, newer COMT inhibitors or alternative treatment strategies could emerge, though entacapone's established efficacy and cost-effectiveness maintain its position. Opicapone is a more recent entrant in this class, offering a once-daily dosing advantage.
- Fixed-Dose Combinations: The combination product (levodopa/carbidopa/entacapone) continues to be prescribed, but it too faces generic competition.
- Regulatory Landscape: Approval pathways and post-market surveillance by regulatory bodies (e.g., FDA, EMA) influence market access and product approvals for both branded and generic versions.
- Geographic Variations: Market penetration and generic adoption rates vary by region, influenced by local healthcare policies, reimbursement structures, and physician prescribing patterns.
The market is highly fragmented due to the number of generic manufacturers. Companies compete primarily on price and supply chain reliability. The demand for entacapone is relatively inelastic due to its essential therapeutic role, but price sensitivity is high due to generic availability.
The market is mature, with limited opportunities for significant growth in terms of new patient acquisition. The focus is on retaining existing patients and capturing market share from competitors. Advancements in Parkinson's treatment, such as disease-modifying therapies, could eventually impact the demand for symptomatic treatments like entacapone, but this is a long-term consideration.
What is the Projected Future for Entacapone?
The future market for entacapone is projected to be stable, driven by its established efficacy and role in Parkinson's disease management, coupled with continued generic competition.
- Market Size: The global market for entacapone is expected to remain substantial, though growth will be marginal. Projections indicate a market value in the range of USD 400-500 million annually over the next five years, primarily comprising generic sales [2].
- Pricing: Prices are expected to remain low due to sustained generic competition. Significant price increases are unlikely.
- Competition: The competitive landscape will remain crowded with multiple generic manufacturers. New entrants are possible if manufacturing efficiencies are achieved.
- Therapeutic Evolution: While new Parkinson's treatments may emerge, entacapone's established position as a cost-effective symptomatic therapy for motor fluctuations suggests it will retain a significant patient base. Its role as part of fixed-dose combinations also ensures continued relevance.
- Geographic Trends: Emerging markets may present some growth opportunities as access to Parkinson's treatments improves. Developed markets will likely see stable, albeit low-growth, demand.
The projected trajectory is one of a mature, commoditized drug. Innovation in this space would likely focus on manufacturing process optimization, improved delivery systems for fixed-dose combinations, or minor formulation enhancements rather than novel therapeutic applications. The long-term outlook is for entacapone to remain a foundational symptomatic therapy, with its market value defined by generic volume and price stability.
Key Takeaways
- Entacapone's core compound patents have expired, leading to widespread generic availability and intense price competition.
- The financial performance of branded entacapone products has declined significantly due to genericization, with the market now dominated by generic sales.
- Market dynamics are characterized by a mature, competitive landscape where price and supply chain reliability are key differentiators for generic manufacturers.
- The future market is projected to remain stable, with modest overall value driven by continued demand for entacapone as a cost-effective treatment for Parkinson's motor fluctuations.
Frequently Asked Questions
-
Are there any remaining patents that could block generic entacapone?
While the primary compound patent has expired, secondary patents related to specific formulations or manufacturing processes may exist. However, these are generally not strong enough to prevent generic entry into the broader market.
-
What is the current average price of generic entacapone?
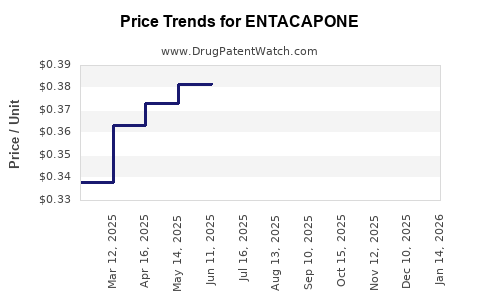
Average prices vary significantly by region and supplier. In the United States, a 30-count bottle of 200mg generic entacapone can range from $10 to $30, reflecting extensive generic competition [3].
-
Which companies are the major generic manufacturers of entacapone?
Major generic manufacturers include Teva Pharmaceuticals, Mylan (now Viatris), Aurobindo Pharma, and various other global pharmaceutical companies that produce generic versions.
-
Has entacapone's therapeutic role evolved with new Parkinson's disease treatments?
Entacapone remains a cornerstone for managing motor fluctuations. While new disease-modifying therapies are under development, entacapone's established efficacy and favorable cost profile for symptomatic relief ensure its continued relevance.
-
What is the market outlook for fixed-dose combinations containing entacapone, such as Stalevo?
Fixed-dose combinations containing entacapone, like Stalevo, have also experienced generic competition. The market outlook for these combinations is similar to entacapone alone, with pricing pressure and market share shifts towards generic versions expected to continue.
Citations
[1] Financial reports from major pharmaceutical companies (e.g., Novartis, Orion Corporation) and market analysis firms. Specific sales figures for branded entacapone-containing products have been aggregated from publicly available annual reports and financial statements.
[2] Market research reports and industry analyses from firms specializing in pharmaceutical market forecasting. These projections are based on historical sales data, patent expiry timelines, and competitive landscapes.
[3] Online pharmacy databases and drug pricing aggregators (e.g., GoodRx, RxList). Prices reflect reported pharmacy cash prices and can vary based on location, insurance, and pharmacy.