Last updated: February 19, 2026
Dexlansoprazole, a proton pump inhibitor (PPI) used to treat gastroesophageal reflux disease (GERD) and other acid-related disorders, faces a mature market following patent expirations. Its primary active ingredient, dexlansoprazole, is the R-enantiomer of lansoprazole. The drug is formulated for delayed-release to optimize its efficacy. This analysis outlines the patent landscape, market competition, and financial implications for dexlansoprazole.
What is the Patent Status of Dexlansoprazole?
The principal U.S. patents protecting dexlansoprazole have expired, opening the market to generic competition. The original New Drug Application (NDA) for dexlansoprazole was filed by Takeda Pharmaceutical Company.
- Core Composition of Matter Patent: U.S. Patent No. 5,492,938, which covered the compound dexlansoprazole itself, expired in 2010.
- Formulation and Method of Use Patents: Several secondary patents related to specific formulations and methods of use also expired in subsequent years. For example, patents covering the delayed-release formulations have also expired.
- Exclusivity Periods: While patents represent the primary barrier to entry, other regulatory exclusivities also played a role. The drug achieved New Chemical Entity (NCE) exclusivity, which provided an initial period of market protection independent of patent terms.
The expiration of these patents and exclusivities has fundamentally altered the competitive landscape, transitioning the market from a branded monopoly to a multi-player generic environment.
Who Are the Key Manufacturers and Competitors in the Dexlansoprazole Market?
The market for dexlansoprazole is now characterized by the presence of multiple generic manufacturers. This has led to significant price erosion and increased market fragmentation.
- Branded Manufacturer: Takeda Pharmaceutical Company was the original innovator and marketer of dexlansoprazole under the brand name Dexilant.
- Major Generic Manufacturers: Following patent expiry, a number of pharmaceutical companies have entered the market with generic versions of dexlansoprazole. These include:
- Teva Pharmaceuticals Industries Ltd.
- Aurobindo Pharma Ltd.
- Dr. Reddy's Laboratories Ltd.
- Sun Pharmaceutical Industries Ltd.
- Mylan N.V. (now part of Viatris Inc.)
- Major generic players frequently compete on price, volume, and market access agreements with pharmacy benefit managers and large healthcare systems.
The presence of these diverse manufacturers intensifies competition, driving down average selling prices (ASPs) for dexlansoprazole formulations.
What is the Market Size and Growth Trajectory for Dexlansoprazole?
The market for dexlansoprazole has experienced a decline in total revenue due to genericization, despite stable or growing unit sales.
- Market Size Data: Prior to significant generic entry, branded dexlansoprazole generated substantial annual revenues. For instance, in its peak years, Dexilant sales exceeded \$1 billion in the U.S. market.
- Post-Genericization Trends: With the advent of generic competition, the aggregate market revenue for dexlansoprazole has contracted considerably. This is a standard dynamic in the pharmaceutical industry following patent expiry.
- Growth Projections: Overall market growth for dexlansoprazole, in terms of revenue, is projected to be negative or flat. Unit sales may remain stable or see modest increases driven by healthcare cost management initiatives that favor generics, but this will not translate into revenue growth for the market as a whole.
- Therapeutic Area Context: The market for PPIs as a class remains large due to the high prevalence of acid-related disorders. However, dexlansoprazole competes not only with its own generics but also with other established PPIs (e.g., omeprazole, lansoprazole, pantoprazole, esomeprazole) and newer treatment modalities.
The financial trajectory is dictated by the aggressive pricing strategies of generic manufacturers and the diminished pricing power of any remaining branded entities.
What are the Key Dosing and Formulation Aspects of Dexlansoprazole?
Dexlansoprazole is available in specific oral dosage forms designed for delayed release, targeting improved patient outcomes through optimized drug delivery.
- Active Pharmaceutical Ingredient (API): Dexlansoprazole.
- Dosage Strengths:
- 30 mg (delayed-release capsules)
- 60 mg (delayed-release capsules)
- Formulation Technology: The drug utilizes a dual delayed-release (DDR) capsule technology. This involves two distinct layers of enteric-coated beads, which release dexlansoprazole at different times within the gastrointestinal tract. This dual release is designed to provide a more sustained plasma concentration of the drug.
- Indications:
- Healing of erosive esophagitis
- Maintenance of healed erosive esophagitis
- Treatment of heartburn associated with diagnosed GERD
- Administration: Dexlansoprazole capsules are typically taken once daily. They can be taken with or without food.
The specific formulation is a key differentiator that was protected by patents, but the underlying API is now available generically.
What is the Competitive Positioning of Dexlansoprazole Against Other PPIs?
Dexlansoprazole's competitive positioning is defined by its unique dual delayed-release formulation and its place within the broader PPI market.
- Within the PPI Class: Dexlansoprazole competes directly with other PPIs, including:
- Omeprazole: The first PPI approved in the U.S.
- Lansoprazole: The racemate of which dexlansoprazole is the R-enantiomer.
- Esomeprazole: The S-enantiomer of omeprazole.
- Pantoprazole: Another widely prescribed PPI.
- Rabeprazole: Known for its rapid onset of action.
- DEXILANT® vs. Other PPIs: The branded product, Dexilant, positioned itself as offering advantages in symptom control and healing rates due to its dual release mechanism. However, the cost-effectiveness of these perceived advantages is often questioned in the face of generic competition for other PPIs.
- Generic Dexlansoprazole vs. Other Generic PPIs: Generic dexlansoprazole now competes on price with generics of all other major PPIs. The therapeutic equivalence is established by regulatory bodies, and the primary decision-making factor for purchasers and prescribers becomes cost.
- Market Dynamics: The overall PPI market is mature and highly competitive. Pricing pressures are significant across the board. Dexlansoprazole's unique delivery system is less of a barrier to entry for generics once the patents expire, as generic companies can develop bioequivalent formulations.
The differentiation for dexlansoprazole has largely been neutralized by generic entry, making it a price-sensitive commodity within the PPI class.
What are the Financial Implications of Generic Competition for Dexlansoprazole?
The financial implications for stakeholders in the dexlansoprazole market are substantial and primarily negative for originators, while creating opportunities for generic manufacturers.
- Revenue Decline for Originator: Takeda experienced a precipitous decline in sales of branded Dexilant following the introduction of generic dexlansoprazole. This is a common and predictable outcome.
- Profitability for Generic Manufacturers: Generic manufacturers can achieve significant profitability by producing dexlansoprazole at lower costs and selling it at prices substantially below the branded originator, while still maintaining healthy profit margins. The market share captured by generics is directly correlated with their ability to offer competitive pricing.
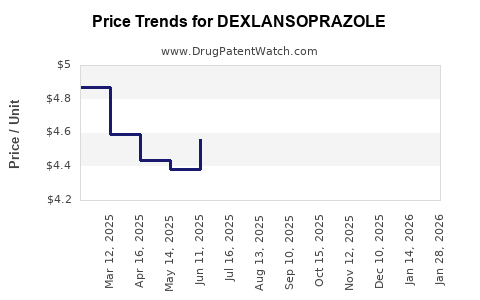
- Pricing Pressure: The introduction of multiple generic players intensifies price competition. The average selling price (ASP) for dexlansoprazole has fallen dramatically since the first generic approvals.
- Market Access and Reimbursement: Generic manufacturers focus on securing favorable formulary placement with payers, including Medicare Part D plans, Medicaid, and commercial insurers. This involves negotiating rebates and discounts.
- Investment Opportunities: For investors, the dexlansoprazole market represents opportunities in companies that have successfully developed and commercialized generic versions. The financial returns are tied to market share capture and efficient manufacturing.
- Decline in R&D Investment: As the market matures and profitability shifts to generics, the incentive for further R&D investment in dexlansoprazole itself diminishes. Focus shifts to lifecycle management of branded products or development of new chemical entities.
The financial narrative is one of value transfer from the innovator to the generic industry, driven by patent expiration and regulatory pathways for generic approval.
What is the Regulatory Landscape for Dexlansoprazole Generics?
The regulatory pathway for generic dexlansoprazole in the United States is governed by the Food and Drug Administration (FDA) under the Hatch-Waxman Act.
- Abbreviated New Drug Application (ANDA): Generic manufacturers must file an ANDA with the FDA.
- Bioequivalence: The core requirement for ANDA approval is demonstrating bioequivalence between the generic product and the branded reference listed drug (RLD), Dexilant. This means the generic must deliver the same amount of active ingredient into the bloodstream in the same amount of time.
- Dosage Forms and Strengths: The ANDA must demonstrate equivalence for all approved dosage forms and strengths (30 mg and 60 mg delayed-release capsules).
- Patent Certifications (Paragraph IV): Generic companies often file "Paragraph IV" certifications in their ANDAs, asserting that the patents covering the RLD are invalid, unenforceable, or will not be infringed by the generic product. This can trigger patent litigation.
- 180-Day Exclusivity: The first generic company to successfully challenge a patent through a Paragraph IV certification can be eligible for 180 days of market exclusivity, during which no other ANDA can be approved. This exclusivity period can significantly impact market share and profitability for the first mover.
- FDA Approval Process: The FDA reviews the ANDA for scientific, technical, and regulatory compliance before granting approval. This includes reviewing manufacturing processes, quality control, labeling, and stability data.
The regulatory framework facilitates generic entry post-patent expiration, driving competition and reducing drug costs.
Key Takeaways
Dexlansoprazole, formerly a high-revenue branded pharmaceutical, has transitioned to a genericized market following the expiration of its key patents. This has led to a substantial decline in overall market revenue for the drug class, with value shifting to generic manufacturers. Multiple competitors, including Teva Pharmaceuticals, Aurobindo Pharma, and Dr. Reddy's Laboratories, now offer generic dexlansoprazole. The drug's dual delayed-release formulation, once a key differentiator, is now replicated by generic versions. Financial implications are characterized by reduced originator sales and increased profitability for generic producers through volume and aggressive pricing. The U.S. regulatory framework, primarily the ANDA process and patent challenge provisions, facilitates this competitive entry.
Frequently Asked Questions
1. When did the primary patent for dexlansoprazole expire?
The core composition of matter patent for dexlansoprazole, U.S. Patent No. 5,492,938, expired in 2010.
2. How does dexlansoprazole's formulation contribute to its market dynamics?
Dexlansoprazole's dual delayed-release formulation was a key innovation that provided market exclusivity for the branded product. Upon patent expiration, generic companies focused on developing bioequivalent formulations, neutralizing this differentiation and enabling broad generic competition.
3. What is the typical impact of generic entry on the price of a drug like dexlansoprazole?
Generic entry typically leads to a significant reduction in the average selling price (ASP) of a drug. For dexlansoprazole, this price erosion has been substantial as multiple generic manufacturers entered the market, intensifying competition.
4. Which companies are the major generic competitors for dexlansoprazole?
Key generic competitors include Teva Pharmaceuticals Industries Ltd., Aurobindo Pharma Ltd., Dr. Reddy's Laboratories Ltd., Sun Pharmaceutical Industries Ltd., and Viatris Inc. (formed from the merger of Mylan and Upjohn).
5. What is the role of bioequivalence in the approval of generic dexlansoprazole?
Bioequivalence is a critical requirement for the FDA's approval of generic dexlansoprazole. Generic manufacturers must demonstrate that their product performs in the body identically to the reference listed drug, Dexilant, in terms of absorption rate and extent.
6. How has the overall revenue of the dexlansoprazole market changed since generic entry?
The overall market revenue for dexlansoprazole has significantly declined following generic entry. While unit sales may remain stable or increase due to cost-saving measures favoring generics, the drastically lower prices per unit result in lower aggregate market revenue.
7. Are there any remaining exclusivities that protect dexlansoprazole?
As of the current market landscape, the primary patent and NCE exclusivities have expired. Any remaining market protection would be through secondary patents on specific formulations or methods of use, which are typically challenged by generic applicants.
8. What are the different dosage strengths available for dexlansoprazole?
Dexlansoprazole is available in delayed-release capsules in 30 mg and 60 mg strengths.
Citations
[1] U.S. Food and Drug Administration. (n.d.). Orange Book: Approved Drug Products with Therapeutic Equivalence Evaluations. Retrieved from https://www.accessdata.fda.gov/scripts/cder/ob/ (Specific patent and exclusivity information can be accessed through searches on this database.)
[2] Takeda Pharmaceutical Company Limited. (2023). Annual Reports. (Historical financial data for branded Dexilant sales can be found in past annual reports.)
[3] Various Pharmaceutical Industry Market Research Reports. (Data on generic market share, pricing, and competitor landscape is often compiled in specialized industry reports.)
[4] U.S. Patent and Trademark Office. (n.d.). Patent Search. Retrieved from https://patft.uspto.gov/ (For details on specific U.S. patent numbers, such as U.S. Patent No. 5,492,938.)