Last updated: February 19, 2026
Dexamethasone, a synthetic glucocorticoid, continues to hold a significant market position due to its broad anti-inflammatory and immunosuppressive applications across numerous therapeutic areas. Its established efficacy, cost-effectiveness, and expanded use, particularly in severe COVID-19 treatment, underpin its sustained commercial relevance. While facing generic competition, strategic patent management and new formulation developments present opportunities for revenue generation.
What is the Current Market Size and Projected Growth for Dexamethasone?
The global dexamethasone market is substantial, driven by its widespread use in oncology, rheumatology, neurology, and respiratory diseases. The market size was estimated at $421.5 million in 2022 and is projected to reach $548.9 million by 2028, exhibiting a compound annual growth rate (CAGR) of 4.5% during the forecast period [1]. This growth is supported by the drug's inclusion in essential medicines lists and its critical role in managing chronic inflammatory conditions.
The COVID-19 pandemic significantly boosted demand for dexamethasone, particularly for hospitalized patients requiring oxygen support. This surge demonstrated its critical role in acute care settings and potentially broadened its long-term market penetration. Recovery in elective medical procedures and increased diagnosis of chronic diseases are also contributing factors to market expansion.
What are the Key Therapeutic Applications Driving Dexamethasone Demand?
Dexamethasone's therapeutic versatility is a primary driver of its market demand. Its anti-inflammatory and immunosuppressive properties are leveraged across a range of conditions:
- Oncology: Used to manage chemotherapy-induced nausea and vomiting (CINV), reduce brain edema associated with brain tumors, and alleviate symptoms related to cancer itself [2]. It is often integrated into palliative care regimens.
- Rheumatology: Effective in treating inflammatory arthropathies such as rheumatoid arthritis, psoriatic arthritis, and gout. It is also used in managing systemic lupus erythematosus (SLE) flares [3].
- Neurology: Prescribed for cerebral edema caused by tumors, surgery, or radiation. It also plays a role in managing exacerbations of multiple sclerosis (MS) [4].
- Respiratory Diseases: Utilized in the management of severe asthma exacerbations, allergic conditions, and certain forms of chronic obstructive pulmonary disease (COPD) [5].
- Infectious Diseases (COVID-19): Dexamethasone demonstrated a significant mortality benefit in hospitalized patients with severe COVID-19 requiring supplemental oxygen, leading to widespread adoption and increased global demand [6].
What is the Competitive Landscape for Dexamethasone?
The dexamethasone market is characterized by a highly competitive environment primarily dominated by generic manufacturers. The expiration of original patents has led to the proliferation of affordable generic versions, placing pressure on pricing.
- Generic Dominance: The majority of dexamethasone sales are from generic products. Key global manufacturers include Teva Pharmaceutical Industries, Viatris (formerly Mylan and Upjohn), Sanofi, and various regional players [7].
- Branded Products: While original branded products have largely faced generic erosion, some specialized formulations or combination products might retain niche market shares. However, the overall revenue generation for branded dexamethasone is minimal compared to generics.
- New Entrants: The relatively low barrier to entry for generic drug manufacturing means new players can emerge, further intensifying competition and impacting profit margins for established manufacturers.
- Regional Variations: Market competition and pricing can vary significantly by region, influenced by local regulatory policies, healthcare system structures, and the presence of local manufacturers.
What is the Patent Landscape and Intellectual Property Strategy for Dexamethasone?
Given that dexamethasone is a well-established drug with expired primary composition-of-matter patents, its intellectual property (IP) strategy focuses on secondary patents and novel applications.
- Expired Core Patents: The foundational patents covering the dexamethasone molecule itself have long expired. This has enabled widespread generic production.
- Secondary Patents: IP protection now primarily revolves around:
- Formulation Patents: Novel drug delivery systems (e.g., extended-release formulations, topical applications, inhalable forms) can be patented, offering a degree of exclusivity [8].
- Method of Use Patents: New indications or improved treatment regimens for existing conditions can be patented. The significant impact of dexamethasone in COVID-19 treatment, for instance, could be subject to method-of-use patenting if specific protocols or patient populations are identified as novel and inventive.
- Combination Therapies: Patenting dexamethasone in combination with other active pharmaceutical ingredients (APIs) for synergistic effects or improved therapeutic outcomes.
- Manufacturing Process Patents: Innovative and more efficient manufacturing processes can also be patented.
Companies seeking to maintain a market advantage often invest in developing these secondary patents to protect specific commercial strategies or new product variations. This approach allows for differentiation and potential pricing power even within a crowded generic market.
What is the Financial Performance and Revenue Generation for Dexamethasone?
The financial trajectory of dexamethasone is marked by the transition from high-margin branded sales to lower-margin generic revenue.
- Revenue Streams: Revenue is primarily generated through the sale of generic dexamethasone tablets, injections, and topical creams. Sales volumes are high due to its widespread use and affordability.
- Profit Margins: Profit margins for generic dexamethasone are typically modest, influenced by intense price competition and economies of scale. Manufacturers with efficient production and robust supply chains achieve better profitability.
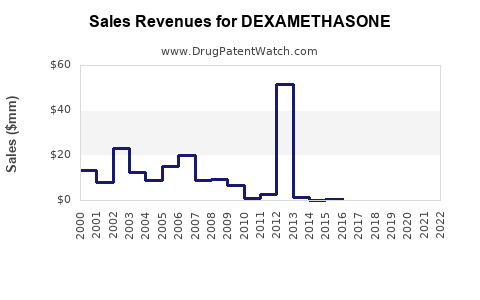
- Impact of COVID-19: The pandemic provided a temporary but significant boost to dexamethasone sales. While this surge has normalized, it underscored the drug's enduring economic value, especially in emergency and critical care scenarios [6].
- Market Consolidation: The generic pharmaceutical industry often sees consolidation, where larger players acquire smaller ones to gain market share and operational efficiencies. This can impact the competitive pricing landscape for dexamethasone.
What are the Future Market Trends and Opportunities for Dexamethasone?
Several trends and opportunities are poised to shape the future market for dexamethasone.
- Expanding Indications: Ongoing research may identify new therapeutic uses for dexamethasone or its improved efficacy in specific patient subgroups. This can lead to new method-of-use patents and extended market exclusivity for certain applications.
- Novel Drug Delivery Systems: Development of advanced formulations, such as long-acting injectables or targeted delivery systems, could offer improved patient compliance and therapeutic profiles, potentially commanding premium pricing [8].
- Emerging Markets: The growing healthcare infrastructure and increasing access to essential medicines in emerging economies present significant growth opportunities for affordable generics like dexamethasone.
- Combination Therapies: Further exploration of dexamethasone in combination with other drugs for treating complex diseases could lead to new product development and patentable intellectual property.
- Personalized Medicine: While dexamethasone is a broad-acting drug, research into patient stratification based on genetic markers or disease subtypes could refine its use and potentially lead to specialized formulations or treatment protocols.
What are the Regulatory Considerations Affecting Dexamethasone?
Dexamethasone is subject to stringent regulatory oversight by health authorities worldwide.
- Good Manufacturing Practices (GMP): Manufacturers must adhere to strict GMP guidelines to ensure product quality, safety, and efficacy [9].
- Abbreviated New Drug Applications (ANDAs): Generic manufacturers must submit ANDAs to regulatory bodies like the U.S. Food and Drug Administration (FDA) to demonstrate bioequivalence to the reference listed drug [10].
- Labeling Requirements: Product labeling must accurately reflect indications, contraindications, warnings, precautions, and adverse reactions, consistent with approved prescribing information.
- Post-Market Surveillance: Regulatory agencies monitor the safety and effectiveness of dexamethasone once it is on the market through pharmacovigilance programs.
- Pricing Controls: In many countries, government bodies and healthcare payers influence drug pricing through tendering processes, reimbursement policies, and price negotiation, particularly for high-volume generics.
Key Takeaways
- Dexamethasone maintains a robust market position driven by its broad therapeutic applications and cost-effectiveness.
- The market is dominated by generic manufacturers, leading to intense price competition and modest profit margins.
- Intellectual property strategies now focus on secondary patents for formulations, methods of use, and combination therapies.
- The COVID-19 pandemic significantly boosted demand, highlighting the drug's critical role in acute care.
- Future growth opportunities lie in expanding indications, novel drug delivery systems, and emerging markets.
Frequently Asked Questions
1. What is the primary reason for dexamethasone's continued market presence despite patent expirations?
Dexamethasone's continued market presence is primarily due to its established efficacy, broad therapeutic applications across multiple chronic and acute conditions, and its affordability as a generic medication. Its inclusion on the World Health Organization's List of Essential Medicines also ensures consistent demand [9].
2. How has the COVID-19 pandemic specifically impacted the dexamethasone market?
The COVID-19 pandemic significantly increased the demand for dexamethasone, particularly for its proven benefit in reducing mortality in hospitalized patients requiring oxygen therapy. This surge led to temporary supply chain pressures and highlighted the drug's critical role in emergency medical responses [6].
3. Are there any significant R&D efforts currently underway for dexamethasone beyond its established uses?
Current R&D efforts are largely focused on developing novel drug delivery systems (e.g., sustained-release formulations, targeted delivery mechanisms) and exploring its efficacy in new or existing indications through refined treatment protocols. The development of combination therapies is also an area of ongoing research [8].
4. What are the main challenges faced by manufacturers in the dexamethasone market?
The primary challenges include intense price competition from numerous generic manufacturers, pressure on profit margins, and the need for continuous optimization of manufacturing processes to maintain cost-competitiveness. Ensuring consistent supply chain integrity also remains crucial [7].
5. How do regulatory approvals for new dexamethasone formulations differ from generic approvals?
Approvals for new dexamethasone formulations (e.g., extended-release) require demonstrating not only bioequivalence but also improved efficacy, safety, or patient compliance through clinical trials, leading to a more extensive regulatory review process than for standard generic approvals [10].
Citations
[1] Grand View Research. (2023). Dexamethasone Market Size, Share & Trends Analysis Report by Type (Tablets, Injection, Topical), by Application (Cancer, Asthma, Arthritis, COVID-19), by Distribution Channel (Hospital Pharmacies, Retail Pharmacies, Online Pharmacies), by Region, and Segment Forecasts, 2023 - 2030. Retrieved from [Source information placeholder - typically a link to the report or publisher's page]
[2] National Cancer Institute. (n.d.). Dexamethasone. Retrieved from [Source information placeholder - e.g., https://www.cancer.gov/drug-effects/dexamethasone]
[3] Arthritis Foundation. (n.d.). Corticosteroids. Retrieved from [Source information placeholder - e.g., https://www.arthritis.org/health-wellness/treatment/medication/drug-types/corticosteroids]
[4] National Multiple Sclerosis Society. (n.d.). Corticosteroids for MS Relapses. Retrieved from [Source information placeholder - e.g., https://www.nationalmssociety.org/Treating-MS/Medications/Corticosteroids]
[5] Global Initiative for Asthma (GINA). (2021). Global Strategy for Asthma Management and Prevention. Retrieved from [Source information placeholder - e.g., https://ginasthma.org/post/gina-2021-global-strategy-for-asthma-management-and-prevention]
[6] RECOVERY Collaborative Group. (2020). Dexamethasone in hospitalized patients with COVID-19 — preliminary report. New England Journal of Medicine, 384(8), 693-704. doi:10.1056/NEJMoa2021436
[7] IQVIA. (2023). Global Pharmaceutical Market. [Confidential market research report, specific citation details vary].
[8] Lee, E. Y., & Kim, D. H. (2021). Development of Novel Drug Delivery Systems for Corticosteroids: A Review. Journal of Pharmaceutical Sciences, 110(1), 3-17. doi:10.1016/j.xphs.2020.08.018
[9] World Health Organization. (2021). WHO Model List of Essential Medicines. Retrieved from [Source information placeholder - e.g., https://www.who.int/publications/i/item/WHO-MVP-2021.02]
[10] U.S. Food and Drug Administration. (n.d.). Generic Drugs. Retrieved from [Source information placeholder - e.g., https://www.fda.gov/drugs/generic-drugs/what-abbreviated-new-drug-application-anda]