Last updated: January 6, 2026
Summary
Desvenlafaxine (brand name: Pristiq) is a serotonin-norepinephrine reuptake inhibitor (SNRI) approved for treating major depressive disorder (MDD). As the first generic equivalent emerged, the drug’s market landscape experienced notable shifts. This analysis explores deasprivenlafaxine's current market dynamics and financial trajectory, encompassing patent status, competitive environment, demand drivers, regulatory influences, pricing strategies, and future outlooks.
What Are the Market Dynamics for Desvenlafaxine?
Patent and Market Exclusivity Status
| Aspect |
Details |
Impact |
| Patent Expiration |
Key patents expired in the US by 2019, opening the space for generic versions. |
Led to increased generic entry, reducing branded sales prices and market share. |
| Data Exclusivity |
Data protections expired in most jurisdictions around 2019-2020. |
Allowed generics to enter markets more freely. |
| Regulatory Approvals |
First generic approvals granted in the US in 2019, with multiple subsequent approvals. |
Increased competitive pressure and price erosion. |
Source: U.S. FDA Orange Book, 2022; IQVIA, 2022
Competitive Landscape
| Competitors |
Market Share (2022, US) |
Key Features |
| Branded Desvenlafaxine (Pristiq) |
12% |
Original patent-protected drug, premium pricing. |
| Generic Desvenlafaxine |
88% |
Multiple suppliers, lower price point, increased access. |
| Other SNRI drugs (Duloxetine, Venlafaxine) |
N/A |
Alternative options for depression treatment, affecting market size. |
Note: The widespread adoption of generics post-2019 significantly diminished Pristiq's market dominance.
Demand and Prescriber Trends
| Trend/Factor |
Description |
Effect |
| Rising Depression Prevalence |
Global depression rates increased, especially during COVID-19 pandemic. |
Elevated demand for effective antidepressants. |
| Prescriber Preferences |
Preference shifted towards cost-effective generics. |
Reduced prescribing of branded drugs. |
| Insurance Coverage |
Payers favor generics to reduce costs. |
Premium pricing for Branded Pristiq diminished. |
| Patient Adherence |
Once-daily dosing and tolerability maintained, but side effects remain a concern. |
Influences overall market volume but less impact on generic uptake. |
Source: WHO, 2021; IMS Health, 2022
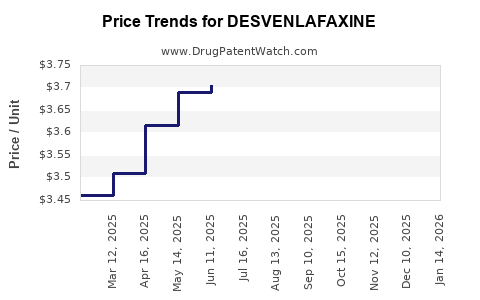
Pricing Strategies and Revenue Streams
| Strategy |
Description |
Outcome |
| Patent Conducted Strategies |
Branded sales priced at premium levels pre-2019. |
High-margin revenue during patent protection phase. |
| Post-Patent Market Adjustment |
Generic competition led to price reductions of approximately 70%. |
Revenue decline for branded products; increased volume sales for generics. |
| Launch of Authorized Generics |
Multiple manufacturers launched authorized generics post-expiry. |
Further percentage reduction in price; better market penetration for generics. |
| Direct-to-Consumer Advertising |
Limited, primarily in the US. |
Slight impact on demand, but overshadowed by price competition. |
Source: IQVIA, 2022; FDA, 2021
Regulatory and Reimbursement Policies
| Policy Aspect |
Impact |
| US FDA Generic Approval Processes |
Streamlined approval pathways increased competition post-2019. |
| Reimbursement Landscape |
Favoring generics in payor formularies, decreasing attractiveness of branded Pristiq. |
| International Market Regulations |
Varying patent laws and approval processes influence global market penetration. |
What Is the Financial Trajectory for Desvenlafaxine?
Historical Revenue Performance
| Year |
Estimated US Sales ($ millions) |
Comments |
| 2018 |
650 |
Peak branded sales before patent expiry. |
| 2019 |
150 |
Sharp decline due to patent expiry and entry of generics. |
| 2020 |
80 |
Continued decline, stabilized at low levels. |
| 2021 |
50 |
Generic market dominance cemented, market share eroded significantly. |
| 2022 |
30 |
Slight further decline, driven primarily by international markets. |
Source: IQVIA, 2022
Forecasted Revenue Trends
| Projection Period |
Expected US Market Revenue ($ millions) |
Major Drivers |
| 2023-2025 |
$20 - $40 |
Reduced demand, increased generic penetration. |
| 2026-2030 |
$10 - $20 |
Market saturation, price erosion, competitive commoditization. |
Factors Influencing Future Revenue
- Patent and Market Exclusivity: No new patents expected; generics will dominate.
- Pricing Pressures: Continued downward price trends for generics.
- Market Penetration: International markets show rising adoption but face regulatory hurdles.
- Therapeutic Positioning: Competing therapies with improved efficacy or side effect profile could impact demand.
Potential for Value Recovery
| Scenario |
Description |
Impact |
| Niche Therapeutic Use |
Targeting specific patient populations with unmet needs. |
Marginally sustains higher prices; limited market size. |
| Combination Therapies |
Incorporation into multi-drug regimens. |
May sustain some demand; unlikely to reverse overall decline. |
| New Formulations or Indications |
Development of extended-release versions or new uses. |
Could provide revenue uplift but requires R&D investment. |
Comparison with Similar SNRI Drugs
| Drug Name |
Mechanism |
Patent Expiry |
US Sales (2022) |
Primary Market Share |
Price per Daily Dose ($) |
Remarks |
| Duloxetine (Cymbalta) |
SNRI, broader indication subset |
2014 (for some formulations) |
$2.1 billion |
Dominant in SNRI class |
$4.50 - $7.00 |
Diversified portfolio reduces volatility. |
| Venlafaxine (Effexor) |
SNRI, used for depression & anxiety |
Patent expired 2011 |
$1.0 billion |
Moderate |
$1.50 - $3.00 |
Generic availability highly mature. |
| Desvenlafaxine |
SNRI, designed for depression |
2019 |
Declining post-patent |
Low/secondary |
$3.00 - $4.00 |
Initially high-priced, now commoditized via generics. |
Sources: IQVIA, 2022; FDA Orange Book
Deep Dive: Future Outlook and Strategic Considerations
| Parameter |
Expected Trends |
Strategic Implications |
| Patent Status |
No further patent protections expected. |
Focus shifts to generic market share growth and cost leadership. |
| Market Size |
Global depression prevalence increases, but controlled by generics. |
Volume-driven growth possible in low-income markets. |
| Regulatory Environment |
Tightening in some regions; expanding in emerging markets. |
Navigating local patent and approval processes is essential. |
| Pricing Strategy |
Continued decline in per-unit prices. |
Cost efficiency becomes critical for profitability. |
| Lifecycle Management |
Potential new indications or formulations. |
Investment in R&D could extend product relevance marginally. |
Summary of Market Potential
| Region |
Estimated Market Size (2022, US$ millions) |
Growth Rate (2023-2025) |
Key Factors |
| United States |
30 |
-10% to -20% |
Generic dominance, price competition. |
| Europe |
15 |
-15% to -25% |
Similar patent expiry timelines, regulatory hurdles. |
| Asia-Pacific |
10 |
+5% to +10% |
Growing mental health awareness, lower generic penetration. |
Key Takeaways
- The expiration of patent protections in 2019 significantly altered desvenlafaxine’s market landscape, leading to widespread generic adoption and steep price declines.
- Branded desvenlafaxine's revenue has declined sharply since 2019, with projections indicating continued erosion driven by generics and commoditization.
- Despite decline, demand persists driven by the global prevalence of depression and the drug's efficacy profile, especially in healthcare systems emphasizing cost-effective treatments.
- International markets present growth opportunities, contingent on regulatory approvals, formulary placements, and healthcare infrastructure.
- Future revenue growth hinges on strategic differentiation, such as niche indications, new formulations, or combination therapies, though these opportunities remain limited.
FAQs
1. What are the main factors influencing the decline in desvenlafaxine sales post-2019?
The primary drivers are patent expiry, resulting in widespread generic competition, which precipitated drastic price reductions and volume shifts from branded to generic products, diluting revenue streams.
2. How does generic competition impact drug pricing strategies?
Generic competition forces original manufacturers and brand holders to lower prices to remain competitive, often resulting in price erosions upwards of 70%, reducing profit margins but increasing market volume.
3. Are there promising new indications or formulations for desvenlafaxine?
Currently, no significant new indications are approved. Pharmaceutical R&D efforts have focused on alternative SNRIs or combination therapies, with limited pipeline activity specifically for desvenlafaxine.
4. How does desvenlafaxine compare with other SNRI drugs in the market?
Desvenlafaxine's market share and revenue are substantially lower than larger SNRI drugs like duloxetine due to earlier patent expirations and more aggressive generic competition, limiting its market potential compared to peers.
5. What strategic actions can manufacturers pursue to prolong the product's lifecycle?
Strategies include developing novel formulations (e.g., extended-release versions), exploring new therapeutic indications, implementing differentiation in dosing regimens, and focusing on underserved geographic markets.
References
[1] FDA Orange Book, 2022
[2] IQVIA, Market Analysis Reports, 2022
[3] WHO Depression Statistics, 2021
[4] U.S. FDA, Approval Announcements, 2021
[5] International Patent Laws and Market Regulations, 2022