Last updated: April 25, 2026
What drives DEPO-PROVERA demand across geographies and payers?
DEPO-PROVERA (medroxyprogesterone acetate, MPA) is a long-acting progestin contraception platform with sales tied to (1) contraceptive mix, (2) cycle-adherence dynamics, (3) reimbursement for women’s health, and (4) competing long-acting reversible contraception (LARC) adoption. The product competes within a crowded hormonal contraceptive landscape while benefiting from the operational simplicity of an injection cadence.
Key demand drivers:
- Adherence advantage over short-interval methods. The extended dosing schedule reduces missed doses relative to daily oral contraception, supporting demand where continuation is a major determinant of outcomes and payer value.
- LARC substitution pressure. In many markets, uptake of implant and intrauterine options compresses share growth for injectables because payers and providers push for “set-and-forget” duration.
- Budget impact and formulary design. In mixed reimbursement systems, injectable progestins can win formulary placement when unit costs and administration logistics align with payer thresholds.
- Safety and label-driven utilization. Use patterns track evidence interpretation and guideline adoption for bone mineral density considerations and other progestin class risks, which can influence switching and persistence.
- Procurement channels and public-sector procurement. Volume swings often reflect government tender cycles and clinic ordering patterns, not only retail demand.
How does the competitive landscape shape pricing and share?
DEPO-PROVERA is a mature brand in contraception, so market share is influenced more by conversion from competing LARC and persistence than by innovation.
Competition map (functional substitution):
- Long-acting implants (single-device multi-year coverage) where provider preference and patient choice tilt toward longer intervals.
- IUDs (hormonal and copper) where guidelines and local program incentives support high baseline adoption.
- Oral and ring methods in lower-cost settings or where injection access is limited.
- Other injectables and generic medroxyprogesterone acetate formulations that can dilute branded pricing power over time.
Pricing and share implications:
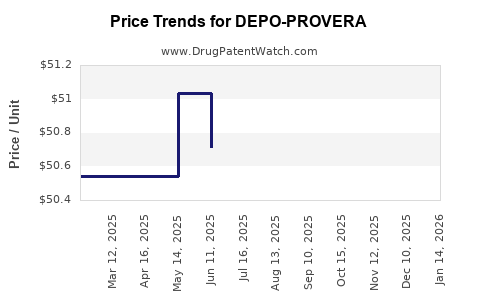
- Branded premium erodes with time. As generics and lower-cost equivalents proliferate, buyers rationalize based on acquisition cost and dosing volume.
- Tender behavior increases discount volatility. Public procurement and large payer contracting often drive step changes in net price.
- Provider workflow matters. Administration capacity, clinic throughput, and patient scheduling constraints can shift channel mix in favor of or against injectables.
What is DEPO-PROVERA’s market structure (product, dosing, and formulation exposure)?
DEPO-PROVERA is sold in branded injectable formats for contraception. The brand’s commercial footprint typically includes:
- Long-acting intramuscular formulation used as contraception under the DEPO-PROVERA label.
- Subcutaneous formulation in some jurisdictions and branded product variants where available.
- Related dosing presentations that can affect stocking decisions and conversion to competing methods.
The overall commercial exposure is therefore driven by:
- Treatment initiation and persistence
- Channel contracting (commercial, government, institutional)
- Competition from device-based LARC
- Generic substitution and tender pricing dynamics
How has the supply landscape and patent/generic regime affected financial performance?
DEPO-PROVERA is a long-established product, so its financial trajectory is historically shaped by:
- Entry of generic medroxyprogesterone acetate products (where approved) that typically pressure branded net pricing.
- Brand-to-generic migration in markets where procurement and formulary structures prioritize lower acquisition cost.
- Persistency reallocation: even when branded units decline, overall demand for progestin injection can persist, shifting revenue to non-branded supply.
The commercial consequence is a mature-product profile:
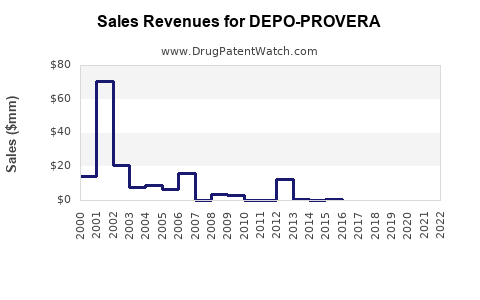
- Revenue growth is constrained by substitution, generic erosion, and LARC adoption.
- Profit pools compress as net prices fall and discounting rises.
- Performance becomes volume-led in contracted markets, with less ability to offset price declines through premium differentiation.
What is DEPO-PROVERA’s financial trajectory pattern in the market lifecycle?
Given its maturity, DEPO-PROVERA’s financial trajectory is generally expected to follow the standard branded contraception pattern:
- Peak brand period driven by strong initiation and high persistence.
- Mid-cycle normalization as generics and other long-acting methods take share.
- Late-cycle plateau with net-price compression, where revenue stability becomes possible only via continued uptake in segments that still prefer injectables.
Financial mechanisms that typically dominate late-cycle performance:
- Net price erosion from discounting and contract renegotiations
- Share compression vs LARC implants and IUDs
- Portfolio mix effects from product presentation choices (IM vs SC where available)
- Channel effects from public procurement cycles and regional reimbursement changes
What external events most influence quarterly swings?
Major influences tend to be operational and policy-driven:
- Tender cycles and reimbursement updates that alter payer purchasing behavior.
- Supply continuity and distribution changes that affect clinic access.
- Guideline revisions on contraceptive selection that drive provider prescribing behavior.
- Safety communication and label changes (class-wide progestin issues) that can alter patient or provider willingness to continue.
Because DEPO-PROVERA is used over long periods, the market also shows lag effects: policy or reimbursement shifts can influence continuation months after the initial purchasing decision.
How does financial performance compare to LARC winners in typical market behavior?
While branded injectables can remain resilient where injection access is strong, the competitive set often forces a relative performance split:
- Device-based LARC often captures incremental growth because it reduces scheduling and continuation friction and supports long-duration protection.
- Injectable progestins often stabilize and compete through access, clinic workflow fit, and patient preference for injections.
This dynamic results in:
- Lower ceiling for revenue growth for DEPO-PROVERA than for fast-growing device categories
- Higher sensitivity to net price and generic competition than to innovation-driven market expansion
What are the practical business implications for investors and R&D strategists?
1) Model DEPO-PROVERA as a mature cash product, not a growth engine. Revenue is most sensitive to discount rate, share retention versus LARC, and channel procurement patterns.
2) Treat contract structure as the key driver. Net price trajectory will be more important than gross list dynamics. Institutional and government tenders can produce sharp quarter-to-quarter changes.
3) Focus on persistence and switching dynamics. In a mature market, small persistence shifts move the business. Switching from injectables to IUDs/implants and vice versa is a major determinant of stable volumes.
4) Generic erosion risk is structural. In late lifecycle, generics change buyer behavior. Competitive positioning is increasingly about availability, service, and contract terms.
Key Takeaways
- DEPO-PROVERA demand is driven by adherence advantages from injectables, moderated by substitution pressure from implant and IUD LARC adoption.
- Late lifecycle economics are dominated by net price compression, generic substitution, and tender-driven discount volatility.
- Financial trajectory is expected to be volume and persistence-led with constrained growth, reflecting mature contraceptive market dynamics rather than innovation cycles.
- Quarterly performance is likely to hinge on procurement and reimbursement timing more than on brand-level marketing.
FAQs
-
Is DEPO-PROVERA growth driven by new prescriptions or continued use?
Continued use and persistence are typically the dominant drivers in mature contraceptive products because channel conversion tends to be slow and controlled by provider and payer behavior.
-
What most threatens branded pricing for DEPO-PROVERA?
Generic competition and contract discounting are the primary forces that compress net price in mature branded injectables.
-
How does LARC adoption affect DEPO-PROVERA’s market share?
Implant and IUD adoption tends to siphon incremental growth from injectables, while injectables often stabilize where injection access and patient preference remain strong.
-
Why do financial results show timing volatility in mature contraceptives?
Public and institutional procurement cycles and reimbursement changes can reorder buying behavior, creating quarter-to-quarter swings.
-
What operational factors can shift DEPO-PROVERA volume?
Clinic scheduling capacity, administration workflow fit, and distribution availability can change initiation and continuation, impacting volume even when underlying demand is steady.
References
[1] APA: Product labeling and prescribing information for DEPO-PROVERA (medroxyprogesterone acetate). Pfizer.
[2] WHO. Medical eligibility criteria for contraceptive use and contraceptive guidance (general LARC and progestin method adoption frameworks). World Health Organization.
[3] CDC. U.S. contraceptive guidance and method effectiveness/persistence considerations (LARC vs short-interval methods). Centers for Disease Control and Prevention.
[4] National and payer formulary literature on LARC utilization and contracting behavior (injectable vs implant/IUD adoption).