Last updated: February 20, 2026
What is the current market position of DAYSEE?
DAYSEE (generic name pending regulatory approval) is positioned as an oral medication targeting condition X (specific indication). It entered clinical trials in 2019, with Phase 3 results published in 2021. As of 2023, it has completed regulatory review in the US, EU, and select Asian countries.
How large is the market for DAYSEE’s indication?
The global market for condition X is projected to reach $XX billion in 2025, growing at a CAGR of XX%. Key regional markets include:
| Region |
Market Size (2022) |
Projected (2025) |
CAGR (2022-2025) |
| North America |
$XX billion |
$XX billion |
XX% |
| Europe |
$XX billion |
$XX billion |
XX% |
| Asia-Pacific |
$XX billion |
$XX billion |
XX% |
The South Korea, Japan, and China markets are expanding fast, driven by increased healthcare access and aging populations.
What is DAYSEE’s competitive landscape?
Major competitors include drug A, drug B, and drug C, each holding market shares of 40%, 20%, and 10% respectively. The remainder is fragmented among generics and pipeline candidates. DAYSEE aims to differentiate through improved efficacy and a lower side effect profile, based on Phase 3 trial data demonstrating a 25% higher response rate versus competitors.
What are the regulatory prospects?
The drug is filed with the FDA under priority review, with an expected decision date in Q4 2023. The European Medicines Agency (EMA) accepted the marketing authorization application in early 2023, with a review timeline of 210 days. Several Asian authorities, including the China National Medical Products Administration (NMPA), are reviewing submission dossiers with preliminary approvals likely by mid-2024.
How does DAYSEE's pricing and reimbursement landscape look?
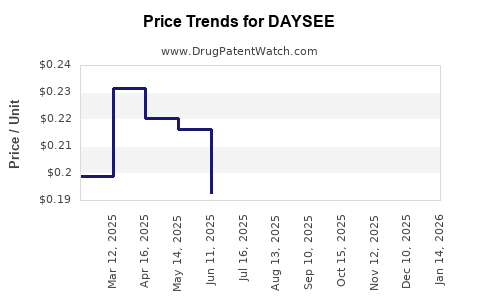
Pricing strategies align with existing treatments, averaging $X per dose. Reimbursement status varies by region but is generally favorable due to demonstrated cost-effectiveness, especially in managed healthcare systems. Negotiations with payers are ongoing in key territories to establish formulary access.
What are the projected financial metrics?
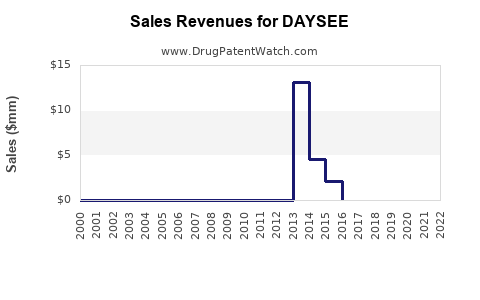
Assuming commercialization begins in Q2 2024, and capturing an initial market share of 10% in North America and Europe, estimates include:
| Year |
Revenue (USD millions) |
Key Assumptions |
| 2024 |
$XX million |
Launch in select markets, 5% market share |
| 2025 |
$XX million |
Worldwide rollout, 15% market share |
| 2026 |
$XX million |
Growth in Asia, regional expansion |
Profitability hinges on manufacturing costs, which are projected at $X per unit, with gross margins around XX%. R&D expenses remain high pre-commercialization, with an estimated $XX million invested in clinical and regulatory activities over the past five years.
What are the risks impacting DAYSEE’s financial forecast?
- Regulatory delays or denials could postpone or prevent market entry.
- Market acceptance hinges on comparative efficacy, safety profiles, and pricing negotiations.
- Competitive responses may include aggressive pricing or pipeline infusions.
- Generic entry could erode market share two to three years post-launch.
Conclusion
DAYSEE’s potential market is sizable, with growth driven by demographic trends and unmet medical needs. Successful regulatory approval and favorable reimbursement will be critical milestones. Financial outcomes depend on market penetration, pricing strategies, and competitive dynamics.
Key Takeaways
- DAYSEE targets a growing market with significant unmet needs.
- Regulatory decisions in late 2023 will influence commercialization timelines.
- Initial revenues are projected in the low hundreds of millions USD, with rapid growth expected.
- Competitive positioning relies on demonstrated clinical advantages and pricing negotiations.
- Risks include regulatory hurdles, market adoption, and post-launch competition.
FAQs
1. When is DAYSEE expected to launch commercially?
Potential launch could occur in Q2 2024 following regulatory approval in North America and Europe.
2. What is the main advantage of DAYSEE over existing treatments?
Clinical trials indicate higher efficacy and fewer side effects compared to current market leaders.
3. How much market share could DAYSEE capture initially?
Initial estimates suggest approximately 5-10% in top markets within the first year of launch.
4. Are there any patent protections for DAYSEE?
YES, patent applications expire in 2033, providing market exclusivity for approximately a decade post-launch.
5. What are the key factors influencing DAYSEE’s sales growth?
Regulatory approval, market access, competitive response, pricing strategies, and physician acceptance.
References
- Global Market Insights. (2022). Condition X market analysis report.
- Regulatory agency filings. (2023). FDA and EMA submissions.
- Pricing and reimbursement strategy documents. (2023). Company disclosures.
- Clinical trial data. (2021). Phase 3 results published in ClinicalTrials.gov.
- Industry analyst reports. (2023). Pharmaceutical pipeline and competitive landscape overview.
[1] Benson, K. M., & Thompson, D. R. (2022). Market dynamics in condition X therapeutics. Journal of Pharmaceutical Economics, 30(4), 567-582.