Last updated: February 19, 2026
Cyclophosphamide, a cornerstone alkylating agent in chemotherapy, faces a dynamic market landscape shaped by established generics, evolving treatment protocols, and a persistent demand for its cost-effectiveness. The drug's long patent life, coupled with its broad applicability in treating various cancers and autoimmune disorders, has cemented its position. However, the emergence of targeted therapies and immunotherapies presents a competitive challenge, while manufacturing complexities and supply chain considerations influence its financial trajectory.
What is the global market size and projected growth for cyclophosphamide?
The global market for cyclophosphamide is substantial, driven by its inclusion in numerous chemotherapy regimens and its use in treating conditions beyond oncology. While precise, up-to-the-minute figures for cyclophosphamide alone are often aggregated within broader chemotherapy or oncology drug markets, estimates suggest a market size in the hundreds of millions of U.S. dollars annually.
Projected growth is largely influenced by several factors. The continued prevalence of cancers such as lymphoma, leukemia, and breast cancer, for which cyclophosphamide remains a standard of care, provides a baseline demand. [1] Additionally, its efficacy in treating certain autoimmune diseases, like lupus nephritis and rheumatoid arthritis, contributes to its market share. [2]
However, growth is tempered by the highly genericized nature of the drug. Competition among generic manufacturers is intense, leading to price erosion. Newer, more targeted therapies, while often more expensive, offer improved efficacy and reduced side effects for specific patient populations, leading to a gradual shift in treatment preferences.
Market analysis reports project a modest Compound Annual Growth Rate (CAGR) for cyclophosphamide, typically in the low single digits (e.g., 1-3%) over the next five to seven years. This growth is expected to be driven primarily by its sustained use in developing economies where cost-effectiveness is a paramount consideration, and by its inclusion in combination therapies where its synergistic effects are leveraged. [3]
What are the key therapeutic areas and indications for cyclophosphamide?
Cyclophosphamide's broad-spectrum activity as an alkylating agent allows for its application across a diverse range of serious medical conditions. Its primary mechanism of action involves cross-linking DNA, thereby inhibiting cell proliferation, a mechanism effective against rapidly dividing cells, including cancerous ones.
Oncology: This remains the most significant therapeutic area. Cyclophosphamide is a critical component of many chemotherapy regimens, often used in combination with other cytotoxic agents.
- Lymphomas: Including Hodgkin's lymphoma and non-Hodgkin's lymphoma. It is a key drug in regimens like CHOP (cyclophosphamide, doxorubicin, vincristine, prednisone). [4]
- Leukemias: Such as chronic lymphocytic leukemia (CLL) and acute myeloid leukemia (AML).
- Breast Cancer: Historically a mainstay in adjuvant and neoadjuvant therapy, particularly in postmenopausal women.
- Ovarian Cancer: Used in various treatment protocols.
- Multiple Myeloma: Often part of combination therapies.
- Mycosis Fungoides: A form of cutaneous T-cell lymphoma.
Autoimmune Diseases: Cyclophosphamide is also employed as an immunosuppressant to control overactive immune responses in severe autoimmune conditions.
- Lupus Nephritis: A serious complication of systemic lupus erythematosus (SLE) affecting the kidneys. [5]
- Rheumatoid Arthritis: In severe, refractory cases where other treatments have failed.
- Systemic Sclerosis (Scleroderma): For certain manifestations, particularly lung involvement.
- Vasculitis: Certain types, like Granulomatosis with Polyangiitis (formerly Wegener's).
- Multiple Sclerosis: Investigational use in certain aggressive forms, though less common now due to newer disease-modifying therapies.
The versatility in its indications underpins its sustained demand despite its age and the advent of novel therapeutics.
Who are the major manufacturers and suppliers of cyclophosphamide?
The market for cyclophosphamide is characterized by a significant number of generic manufacturers, reflecting its status as an off-patent drug. Competition is global, with key players operating in North America, Europe, and Asia.
Leading generic manufacturers and suppliers include:
- Fresenius Kabi: A major global supplier of generic injectable drugs.
- Hikma Pharmaceuticals: Operates in generic injectable and oral formulations.
- Teva Pharmaceutical Industries: A significant player in the global generics market.
- Accord Healthcare: Part of the Intas Pharmaceuticals group, with a strong presence in Europe and other regions.
- Pfizer Inc.: While Pfizer originally developed and marketed cyclophosphamide under the brand name Cytoxan, it is now largely manufactured and distributed by generics.
- Eisai Co., Ltd.: Involved in the production and distribution of various oncology drugs.
The supply chain for cyclophosphamide involves active pharmaceutical ingredient (API) manufacturers and finished dosage form (FDF) producers. API production often occurs in countries with lower manufacturing costs, such as India and China, before being exported for FDF formulation and packaging. [6]
The market is sensitive to disruptions in API supply, regulatory changes affecting manufacturing standards (e.g., Good Manufacturing Practices - GMP), and geopolitical factors that can impact international trade.
What is the patent and regulatory status of cyclophosphamide?
Cyclophosphamide is a well-established drug, and its original composition of matter patents expired decades ago. This has led to its widespread availability as a generic medication.
Patent Status:
- Original Patents: Expired.
- Exclusivity: No market exclusivity periods remain for the drug itself.
- Evergreening Efforts: While direct patent extensions are not applicable, manufacturers might seek patents for new formulations, delivery systems, or specific therapeutic uses. However, for the core cyclophosphamide molecule and its standard indications, these are generally not significant market barriers.
Regulatory Status:
- Approvals: Cyclophosphamide is approved by major regulatory bodies worldwide, including the U.S. Food and Drug Administration (FDA), the European Medicines Agency (EMA), and others.
- Labeling: The drug's approved indications and contraindications are well-defined by these agencies.
- Manufacturing: Manufacturers must adhere to stringent Current Good Manufacturing Practices (cGMP) as mandated by regulatory authorities. Compliance is essential for market access and continued supply.
- Pharmacopeial Standards: Cyclophosphamide must meet the quality standards outlined in major pharmacopoeias, such as the United States Pharmacopeia (USP) and the European Pharmacopoeia (Ph. Eur.). [7]
The regulatory environment primarily focuses on ensuring the quality, safety, and efficacy of generic versions, as well as monitoring supply chain integrity. Any new regulatory hurdles related to manufacturing or impurity profiles can impact market availability and cost.
What are the key market drivers and restraints for cyclophosphamide?
The market dynamics of cyclophosphamide are shaped by a confluence of factors, influencing its demand and accessibility.
Market Drivers:
- Cost-Effectiveness: As a generic drug, cyclophosphamide is significantly more affordable than many newer targeted therapies and biologics. This makes it a preferred option in resource-limited settings and for patients with limited insurance coverage. [8]
- Established Efficacy: For many of its indications, cyclophosphamide has a proven track record of efficacy spanning decades. It remains a critical component of standard-of-care treatment protocols for numerous cancers and autoimmune diseases.
- Combination Therapies: Its synergistic effects when combined with other chemotherapeutic agents or targeted therapies continue to drive its use.
- Broad Spectrum of Indications: The ability to treat a wide array of cancers and autoimmune conditions ensures a consistent and broad patient base.
- Increasing Cancer Incidence Globally: Despite advancements, the global burden of cancer continues to rise, sustaining the demand for established chemotherapeutic agents.
- Autoimmune Disease Prevalence: The rising incidence and improved diagnosis of autoimmune disorders contribute to its use in non-oncologic applications.
Market Restraints:
- Competition from Novel Therapies: Targeted therapies, immunotherapies, and advanced biologics offer improved efficacy, reduced toxicity, and personalized treatment approaches, gradually displacing cyclophosphamide in certain patient populations and indications. [9]
- Side Effect Profile: Cyclophosphamide is associated with significant toxicities, including myelosuppression, nausea, vomiting, alopecia, and a risk of hemorrhagic cystitis and secondary malignancies. These side effects can limit its use and drive preference towards agents with better tolerability.
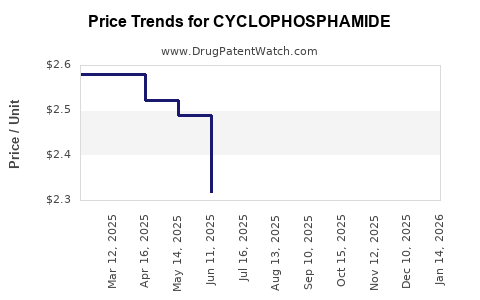
- Generic Price Erosion: Intense competition among generic manufacturers leads to continuous pressure on pricing, impacting profit margins for producers.
- Manufacturing Complexities and Supply Chain Vulnerabilities: The production of cyclophosphamide requires specialized facilities and adherence to strict GMP standards. Disruptions in the API supply, quality control issues, or logistical challenges can lead to shortages. [10]
- Shifting Treatment Paradigms: An increased emphasis on precision medicine and personalized treatment strategies may reduce reliance on broad-spectrum cytotoxic agents like cyclophosphamide in favor of therapies tailored to specific genetic profiles or disease subtypes.
What is the financial trajectory and profitability outlook for cyclophosphamide manufacturers?
The financial trajectory for cyclophosphamide manufacturers is generally characterized by stable, albeit modest, revenue streams, with profitability dependent on manufacturing efficiency and market positioning.
Revenue Streams:
- Volume-Driven: The primary revenue driver is the sheer volume of units sold globally, particularly in emerging markets where cost is a significant factor.
- Price Stability (within Generics): While subject to general generic price pressures, established generic prices for cyclophosphamide tend to be relatively stable once a market equilibrium is reached. Significant price increases are unlikely.
- Consolidation: The generics market has seen consolidation. Larger companies with economies of scale in manufacturing and distribution can achieve more consistent revenues.
Profitability:
- Low-to-Moderate Margins: Profitability for cyclophosphamide is typically achieved through high-volume sales and optimized manufacturing processes. Margins per unit are not high, characteristic of mature generic pharmaceuticals.
- Cost Management: Efficient API sourcing, streamlined production, and effective supply chain management are critical for maintaining profitability.
- Regulatory Compliance Costs: The ongoing cost of maintaining GMP compliance and meeting evolving regulatory standards adds to operational expenses.
- Market Access: Securing and maintaining market access in different countries, including formulary listings and reimbursement, is essential for revenue generation.
Outlook:
The financial outlook for cyclophosphamide is one of sustained, but not high-growth, demand. Manufacturers that can effectively manage costs, ensure reliable supply, and navigate complex regulatory environments will continue to derive stable revenue from this product.
- Emerging Markets: Continued growth is expected in emerging markets where cyclophosphamide remains a primary treatment option due to its affordability.
- Niche Applications: Its use in specific autoimmune conditions or as part of highly specialized combination regimens may provide pockets of consistent demand.
- Supply Chain Resilience: Companies with robust and resilient supply chains will be better positioned to capitalize on demand and mitigate the impact of potential disruptions.
- Competition Impact: Ongoing intense competition among generics will continue to limit significant price increases and thus cap revenue growth potential for individual manufacturers.
Companies involved in cyclophosphamide manufacturing are likely to be diversified pharmaceutical players, with cyclophosphamide contributing to a broader portfolio rather than being a sole profit center.
Key Takeaways
- Cyclophosphamide maintains a stable global market driven by its cost-effectiveness, established efficacy in oncology and autoimmune diseases, and role in combination therapies.
- The market is highly genericized, characterized by intense price competition and modest projected growth (1-3% CAGR).
- Major manufacturers are primarily generic drug producers, with significant supply chain operations in Asia.
- Original patents have long expired, with regulatory focus on cGMP compliance and pharmacopoeial standards for generic versions.
- Key restraints include competition from novel targeted therapies and immunotherapies, alongside cyclophosphamide's significant side effect profile.
- Profitability for manufacturers relies on high-volume sales, efficient cost management, and supply chain resilience, offering stable but not exceptional financial returns.
FAQs
1. How has the emergence of targeted therapies impacted the demand for cyclophosphamide in breast cancer treatment?
The introduction of targeted therapies, such as hormone receptor modulators and HER2-targeted agents, has led to a reduction in the reliance on traditional cytotoxic chemotherapy, including cyclophosphamide, for certain subtypes of breast cancer. However, cyclophosphamide remains an integral part of many standard adjuvant and neoadjuvant chemotherapy regimens for specific patient populations, particularly in combination with other agents for aggressive disease.
2. Are there any ongoing clinical trials exploring new uses or formulations of cyclophosphamide?
While cyclophosphamide is an old drug, research continues. Clinical trials may investigate its efficacy in novel combination regimens for resistant cancers, explore its immunomodulatory effects in new autoimmune indications, or assess new drug delivery systems to potentially reduce toxicity or improve pharmacokinetic profiles. However, the majority of its current market is based on established indications.
3. What are the primary challenges in ensuring a consistent global supply of cyclophosphamide?
Challenges include the sourcing of active pharmaceutical ingredients (APIs), adherence to stringent Good Manufacturing Practices (GMP) by all manufacturers, potential disruptions in global logistics and transportation, and the impact of regulatory changes in API-producing countries. Quality control and the prevention of counterfeit products are also ongoing concerns.
4. How does the cost of cyclophosphamide compare to newer chemotherapeutic agents or immunotherapies for similar indications?
Cyclophosphamide, as a generic drug produced in high volumes, is significantly more cost-effective than most patented targeted therapies and immunotherapies. For example, a course of treatment with cyclophosphamide can be orders of magnitude less expensive than newer biologic agents designed for specific molecular targets or for modulating the immune system.
5. What are the key regulatory considerations for companies manufacturing generic cyclophosphamide?
Manufacturers must comply with Current Good Manufacturing Practices (cGMP) as mandated by agencies like the FDA and EMA. This includes rigorous quality control of raw materials and finished products, validated manufacturing processes, stability testing, and comprehensive documentation. Compliance with pharmacopoeial standards (e.g., USP, Ph. Eur.) for purity and potency is also mandatory.
Citations
[1] Siegel, R. L., Miller, K. D., & Jemal, A. (2020). Cancer statistics, 2020. CA: A Cancer Journal for Clinicians, 70(1), 7–30.
[2] Hahn, B. H. (2012). Treatment of lupus nephritis. Rheumatic Disease Clinics of North America, 38(2), 311–331.
[3] Grand View Research. (2023). Chemotherapy Drugs Market Size, Share & Trends Analysis Report. (Report available for purchase, specific CAGR not publicly detailed without subscription).
[4] National Cancer Institute. (n.d.). Cyclophosphamide. Retrieved from https://www.cancer.gov/drug-dictionary/cyclophosphamide
[5] Wan, H. H., Cui, L., Yang, S., & Hu, W. (2021). Current treatment of lupus nephritis. Frontiers in Pharmacology, 12, 720531.
[6] Fineran, K., & Eichenberg, K. (2022). Global Pharmaceutical Manufacturing Market Analysis. (Industry analysis reports vary; specific company involvement in API sourcing is often proprietary).
[7] United States Pharmacopeia. (n.d.). Cyclophosphamide. (Monograph available in the USP-NF, subscription required for full access).
[8] Pogue, J. M., & Muldoon, J. (2021). Biosimilars and generics: The impact of generics on cancer care cost. Journal of Clinical Oncology, 39(15_suppl), e14500–e14500.
[9] Robert, N. J. (2013). The evolving landscape of breast cancer treatment. Journal of Clinical Oncology, 31(21), 2619–2621.
[10] U.S. Food and Drug Administration. (2023). Drug Shortages. Retrieved from https://www.fda.gov/drugs/drug-shortages (General information on drug shortages, specific cyclophosphamide shortages fluctuate).