Last updated: February 19, 2026
Clindamycin phosphate, a widely prescribed lincosamide antibiotic, exhibits stable market performance driven by its established efficacy against anaerobic and gram-positive bacteria. The drug's generic availability and broad therapeutic applications in treating skin infections, bone and joint infections, and pelvic inflammatory disease contribute to consistent demand. While the market is mature, ongoing research into novel delivery systems and combination therapies presents opportunities for incremental growth and differentiation. Financial performance is characterized by steady revenue streams from generic sales, with profitability influenced by manufacturing efficiencies and pricing pressures from competitive generic manufacturers.
What is the Current Market Size and Growth Rate for Clindamycin Phosphate?
The global clindamycin phosphate market is a mature segment within the broader antibiotic sector. Precise current market size figures are proprietary and vary by market research firm. However, industry analysis indicates a market valuation in the hundreds of millions of U.S. dollars annually. Growth rates are generally low, typically in the low single digits (1-3% compound annual growth rate – CAGR). This subdued growth is characteristic of well-established generic drugs with limited patent exclusivity. The primary drivers of demand remain consistent, stemming from its utility in treating common bacterial infections.
The market is segmented by application, with dermatological applications (e.g., acne treatment) representing a significant portion of usage. Other key applications include intra-abdominal infections, bone and joint infections, and gynecological infections. The pharmaceutical industry's focus on novel antibiotic development for resistant pathogens means that growth for older antibiotics like clindamycin phosphate is more likely to come from increased generic penetration in emerging markets or minor formulation improvements rather than breakthrough clinical advancements.
What are the Key Therapeutic Applications Driving Demand?
Clindamycin phosphate's therapeutic utility is diverse, underpinning its sustained market presence. The primary indications include:
- Dermatological Infections: This is a major application, particularly in the topical treatment of acne vulgaris. Clindamycin phosphate lotions, gels, and solutions effectively target Cutibacterium acnes bacteria.
- Bone and Joint Infections: Intravenous and oral formulations are used to treat osteomyelitis and septic arthritis, often in combination with other antibiotics, particularly when Staphylococcus aureus is implicated.
- Intra-abdominal Infections: It is a cornerstone in treating serious infections such as peritonitis and appendicitis, especially when anaerobic bacteria are suspected. It is frequently used in combination with antibiotics that cover aerobic gram-negative bacilli.
- Pelvic Inflammatory Disease (PID): Intravenous clindamycin phosphate is a standard of care for PID, often combined with an agent that provides coverage against gram-negative organisms.
- Respiratory Tract Infections: While less common than other indications, it can be used for certain types of pneumonia and empyema, especially those caused by anaerobic bacteria.
- Bacterial Vaginosis: Vaginal formulations are used for the treatment of bacterial vaginosis.
The prevalence of these conditions globally, coupled with the drug's established safety and efficacy profile at a lower cost point compared to newer agents, ensures a consistent demand.
What is the Competitive Landscape and Key Players?
The competitive landscape for clindamycin phosphate is characterized by a high degree of genericization. Following patent expiry, numerous pharmaceutical manufacturers globally produce and market clindamycin phosphate products. This leads to intense price competition.
Key players in the clindamycin phosphate market include:
- Generic Manufacturers: A large number of companies worldwide produce generic clindamycin phosphate. Examples include Apotex Inc., Mylan N.V. (now Viatris), Teva Pharmaceutical Industries Ltd., Sun Pharmaceutical Industries Ltd., and Zydus Lifesciences Ltd. These companies compete primarily on price and market reach.
- Branded Manufacturers (Limited): While branded versions exist, they typically hold a smaller market share compared to generics. The original innovator may still market a branded product, but its pricing power is significantly limited by the generic competition.
- Formulation Specialists: Some companies may specialize in specific formulations (e.g., topical, injectable) and compete on product quality, delivery system innovation, and regulatory approvals in specific regions.
The market is highly fragmented due to the large number of generic suppliers. Pricing is a critical differentiator, and manufacturers with efficient production processes and established supply chains have a competitive advantage. Regulatory approvals in various global markets (e.g., FDA in the U.S., EMA in Europe) are essential for market access.
What are the Regulatory and Patent Considerations?
Clindamycin phosphate is an off-patent drug in most major markets. The original patents for clindamycin expired decades ago. This has led to the widespread availability of generic versions.
- Patent Status: Generally, no active patents cover the basic clindamycin phosphate molecule or its standard therapeutic uses. This means that any company can manufacture and sell generic clindamycin phosphate as long as they meet regulatory requirements.
- Regulatory Approvals: Manufacturers must obtain marketing authorization from regulatory bodies in each country where they wish to sell the drug. In the U.S., this typically involves submitting an Abbreviated New Drug Application (ANDA) to the Food and Drug Administration (FDA). In Europe, similar approvals are sought from the European Medicines Agency (EMA) or national competent authorities.
- Formulation Patents: While the core drug is off-patent, companies may pursue patents on novel formulations, delivery systems (e.g., extended-release versions, specific topical preparations), or combination therapies. These patents, if granted and defensible, can provide a period of market exclusivity for a specific product variation. However, these are typically incremental and do not significantly alter the overall market dynamics for the generic drug.
- Manufacturing Standards: Regulatory bodies enforce stringent Good Manufacturing Practice (GMP) standards for all pharmaceutical production, including clindamycin phosphate. Compliance is a prerequisite for market access.
The absence of significant patent protection for the base compound means that competition is driven by manufacturing cost, quality, and marketing rather than intellectual property exclusivity.
What are the Financial Performance Drivers and Challenges?
The financial performance of clindamycin phosphate products is shaped by several factors:
Drivers:
- Consistent Demand: As a widely prescribed antibiotic for common infections, demand remains relatively stable.
- Generic Market Access: The low cost of generic clindamycin phosphate makes it accessible in a wide range of healthcare settings, including low- and middle-income countries.
- Manufacturing Efficiencies: Companies that can optimize their manufacturing processes to reduce production costs can achieve higher profit margins.
- Established Supply Chains: Robust and reliable supply chains are crucial for consistent product availability, which is a key factor for healthcare providers.
Challenges:
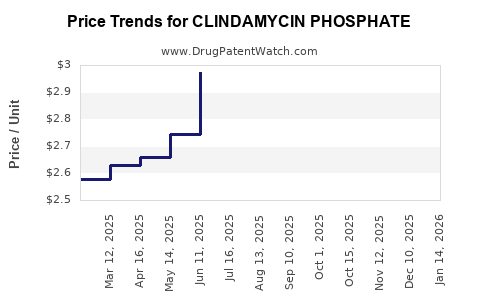
- Price Erosion: Intense competition among numerous generic manufacturers leads to significant price pressure. The price per unit of clindamycin phosphate has steadily declined over the years.
- Volume-Based Sales: Profitability relies heavily on sales volume rather than high per-unit margins.
- Antibiotic Resistance: The increasing threat of antibiotic resistance can impact the long-term utility of older antibiotics and may necessitate a shift towards newer, more expensive agents in certain cases. However, clindamycin phosphate remains effective against many susceptible bacterial strains.
- Regulatory Hurdles: While the drug is off-patent, obtaining and maintaining regulatory approvals in various global markets requires ongoing compliance and investment.
- Raw Material Costs: Fluctuations in the cost of active pharmaceutical ingredients (APIs) and excipients can impact manufacturing costs.
The financial trajectory is one of steady, albeit low, revenue generation. Companies focus on maintaining high production volumes and controlling costs to remain competitive in this mature generic market.
What are the Future Market Trends and Opportunities?
The future market for clindamycin phosphate will likely see continued stability with potential for niche growth.
- Emerging Markets: Increased access to healthcare and rising incomes in emerging economies may lead to higher demand for essential antibiotics like clindamycin phosphate.
- Combination Therapies: Research into novel combination therapies, where clindamycin phosphate is paired with other agents to enhance efficacy or broaden spectrum of activity, could create opportunities for specific product lines.
- Advanced Delivery Systems: Innovations in drug delivery, such as improved topical formulations or extended-release injectables, could offer differentiation and command slightly higher prices.
- Dermatology Focus: The significant use of clindamycin phosphate in topical acne treatments suggests that developments in dermatological formulations will remain a key area for potential product evolution.
- Stewardship Programs: Increased emphasis on antibiotic stewardship may lead to more targeted prescribing, but clindamycin phosphate is likely to retain its role in appropriate indications due to its efficacy and cost-effectiveness.
- Competition from Newer Agents: While clindamycin phosphate will persist, the ongoing development of novel antibiotics targeting resistant strains could gradually reduce its market share in certain complex or resistant infections.
The market is unlikely to experience rapid expansion but will remain a reliable component of the global antibiotic supply, driven by its established role in treating common bacterial infections.
Key Takeaways
- Clindamycin phosphate is a mature antibiotic with stable market demand driven by its broad efficacy in treating common bacterial infections, particularly dermatological and anaerobic infections.
- The market is characterized by intense generic competition, leading to low single-digit CAGR and price erosion.
- Key therapeutic applications include acne treatment, bone and joint infections, and intra-abdominal infections.
- Most patents on clindamycin phosphate have expired, allowing numerous generic manufacturers to compete.
- Financial performance relies on high sales volumes and efficient manufacturing to offset low profit margins per unit.
- Future growth opportunities lie in emerging markets, novel formulations, and combination therapies, though competition from newer antibiotics presents a long-term challenge.
Frequently Asked Questions
-
What is the typical lifespan of a generic drug's market dominance before significant price erosion occurs?
Generic drugs, once off-patent, typically face immediate and substantial price erosion due to multiple manufacturers entering the market. The dominance is characterized by volume rather than price, with significant pressure beginning as soon as the first generic competitor gains approval.
-
Are there any new clinical trials for clindamycin phosphate currently underway that could significantly impact its market?
While extensive research has been conducted on clindamycin phosphate, large-scale Phase III trials for new indications or breakthrough formulations are uncommon for well-established generic drugs. Most ongoing research focuses on incremental improvements or combination therapies rather than entirely new uses for the base molecule.
-
How does the rise of antibiotic resistance specifically affect the market for clindamycin phosphate?
The rise of antibiotic resistance necessitates careful antibiotic stewardship. For clindamycin phosphate, this means its use is increasingly guided by susceptibility testing. While resistance can limit its effectiveness in some severe or persistent infections, it remains a valuable option for susceptible bacterial strains due to its established profile.
-
What are the primary manufacturing challenges for generic clindamycin phosphate producers?
Primary manufacturing challenges include maintaining consistent API quality from suppliers, optimizing synthesis processes for cost-efficiency, ensuring compliance with stringent global GMP standards, and managing the complexities of global distribution networks to ensure product availability and stability.
-
What is the typical profit margin for a generic clindamycin phosphate product compared to a novel patented antibiotic?
Profit margins for generic clindamycin phosphate are considerably lower, often in the single digits or low double digits, reflecting intense price competition and high volumes. In contrast, novel patented antibiotics can achieve profit margins of 30-50% or higher during their exclusivity period, due to pricing power and unmet medical need.
Citations
[1] U.S. Food and Drug Administration. (n.d.). Abbreviated New Drug Applications (ANDAs). Retrieved from https://www.fda.gov/drugs/abbreviated-new-drug-applications-andas
[2] European Medicines Agency. (n.d.). Applying for a medicine's authorisation. Retrieved from https://www.ema.europa.eu/en/human-regulatory/medicines/applying-medicines-authorisation
[3] World Health Organization. (2019). Antibiotic resistance. Retrieved from https://www.who.int/news-room/fact-sheets/detail/antibiotic-resistance
[4] Generic Pharmaceutical Association. (n.d.). The Value of Generic Medicines. Retrieved from https://www.pgenlife.org/ (Note: This link refers to the Pharmaceutical Research and Manufacturers of America, PhRMA, and its member companies. For specific generic association info, an alternative source may be needed if this link is not definitive.)
[5] Viatris Inc. (2023). Annual Report on Form 10-K. (Specific report for the relevant fiscal year would be cited here if available).