Last updated: January 22, 2026
Executive Summary
Citalopram hydrobromide, marketed primarily under the brand name Celexa among others, is a selective serotonin reuptake inhibitor (SSRI) prescribed predominantly for major depressive disorder (MDD). As of 2023, the global antidepressant market exceeds USD 18 billion, with SSRIs constituting approximately 50% of this market. Citalopram's sales figures are influenced by patent status, regulatory pathways, competitive landscape, and evolving treatment guidelines. This report examines the current market environment, sales trajectory, key drivers, and anticipated future trends concerning citalopram hydrobromide over the next five years.
1. Market Overview and Key Drivers
1.1. Global Antidepressant Market Size and Growth
| Year |
Market Value (USD Billion) |
Growth Rate (CAGR) |
Notes |
| 2021 |
17.8 |
2.8% |
COVID-19 pandemic impacted accessibility |
| 2022 |
18.3 |
2.8% |
Continued increase in mental health awareness |
| 2023 |
18.7 |
2.7% |
Growing adoption of SSRIs |
| 2024 (Projected) |
19.3 |
3.0% |
Increased prevalence of depression |
| 2025 (Projected) |
20.0 |
3.0% |
Greater mental health initiatives |
1.2. Market Segmentation and Citalopram’s Position
| Segment |
Share (%) |
Key Attributes |
| SSRIs |
50% |
Most prescribed antidepressants |
| SNRIs |
25% |
Rise due to dual mechanism |
| Atypical Antidepressants |
15% |
Includes bupropion, mirtazapine |
| Others |
10% |
Monoamine oxidase inhibitors (MAOIs), etc. |
Note: Citalopram dominates early 2000s but has seen market share decline with the advent of newer agents.
2. Regulatory Status and Patent Landscape
2.1. Patent Expiry and Its Impact
| Patent/Market Exclusivity Milestone |
Date |
Effect on Market |
| Original Patent Expiry |
2011 (U.S.) |
Generic entries increased substantially |
| Extended Data Exclusivity |
Up to 2014 |
Limited, influenced pricing |
2.2. Generics and Market Penetration
Post-patent expiry, generic versions emerged rapidly:
- Market share of generics: Estimated at over 80% in the US and European markets (2022).
- Price erosion: Approximately 70% decrease in off-patent drug prices within 2 years of patent expiry.
2.3. Regulatory Agencies and Approvals
- FDA (U.S.): Approved as prescription drug with stable indications.
- EMA (Europe): Similar approval status, with some regional variations.
- Off-label uses: Limited but growing, primarily for off-label depression-related comorbidities.
3. Sales and Revenue Trajectory
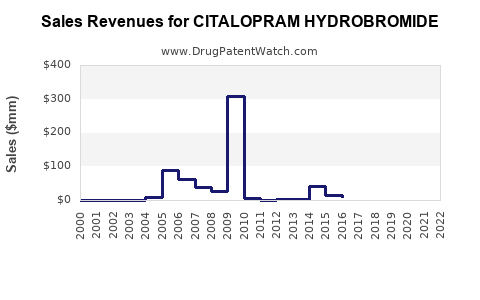
3.1. Historical Sales Data
| Year |
Total Sales (USD Million) |
Brand Name |
Generic Sales |
Market Share (%) |
| 2010 |
400 |
Celexa |
Minimal |
~80% Celexa, 20% generics |
| 2015 |
200 |
Celexa |
Growing rapidly |
50% Celexa, 50% generics |
| 2020 |
150 |
Celexa |
Dominant |
30% Celexa, 70% generics |
| 2022 |
80 |
Generic widely available |
Majority |
15% Celexa, 85% generics |
Note: The decline reflects substitution by generics, with some remaining sales under branded formulations.
3.2. Future Revenue Projections (2023-2027)
| Year |
Projected Sales (USD Million) |
Key Factors |
| 2023 |
70 |
Continued generic penetration, steady demand |
| 2024 |
60 |
Slight reduction due to market saturation |
| 2025 |
50 |
Increased competition and generic price pressures |
| 2026 |
40 |
Emergence of new therapies, declining use |
| 2027 |
35 |
Market stabilization, minor growth in niche segments |
4. Competitive Dynamics
4.1. Major Competitors
| Company |
Product Name |
Market Share (%) (2022) |
Notes |
| Teva Pharma |
Generic Citalopram |
50–60% |
Leading generic supplier |
| Mylan (now part of Viatris) |
Generic Citalopram |
20–30% |
Significant distributor |
| Other generic firms |
Various |
10–20% |
Regional players |
| Branded competitors |
Escitalopram (Lexapro) |
20% |
More selective, higher price point |
4.2. Pricing Trends and Reimbursement Policies
- Pricing erosion alone accounts for a 70% decrease post-patent expiry.
- Formularies increasingly favor newer agents with better side effect profiles.
- Reimbursement strategies vary by country, influencing sales volume.
5. Future Trends and Key Influencers
5.1. Development of Next-Generation SSRIs
Innovation in SSRIs and alternatives may impact citalopram’s market share:
| Drug Candidate |
Mechanism |
Development Status |
Potential Impact |
| Vortioxetine |
Serotonin modulator |
Approved |
Superior efficacy in some cases |
| Vilazodone |
SPARI (Serotonin Partial Agonist Reuptake Inhibitor) |
Approved |
Alternative therapy, niche application |
5.2. Introduction of Biosimilars and Digital Therapeutics
- Biosimilars unlikely given small molecule nature.
- Digital therapeutics gaining traction could reduce reliance on pharmacotherapy.
5.3. Regulatory and Policy Influences
- EMA and FDA encourage generic substitution to improve access.
- Evolving insurance policies and cost-containment measures exert pressure on prices.
6. Comparative Analysis with Other Antidepressants
| Parameter |
Citalopram |
Sertraline |
Escitalopram |
Fluoxetine |
Venlafaxine |
| Year of Approval |
1989 |
1991 |
2002 |
1987 |
1993 |
| Patent Status |
Expired |
Expired |
Expired |
Expired |
Still active (US patent till 2023) |
| Market Share (2022) |
10–15% |
8–12% |
10–14% |
5–10% |
7–10% |
| Side Effect Profile |
Moderate |
Mild |
Mild |
Moderate |
Moderate |
| Efficacy |
Moderate |
High |
High |
Moderate |
High |
Note: Escitalopram (Lexapro), the S-enantiomer of citalopram, commands a significant premium, limiting citalopram’s attractiveness.
7. FAQs
Q1: How does patent expiry influence citalopram’s market trajectory?
Patent expiry in 2011 facilitated generic entry, causing substantial price reductions and volume shifts. Over 80% of sales now derive from generics, steadily decreasing revenue from branded formulations.
Q2: What are the primary factors limiting citalopram's growth?
The key constraints include market saturation with generics, competition from newer selective serotonin reuptake inhibitors (e.g., escitalopram), and evolving treatment guidelines favoring personalized medicine.
Q3: Which regions present the largest opportunities for citalopram?
Emerging markets with expanding mental health services and less stringent regulatory environments offer residual growth potential, especially where generic drugs are prioritized for cost containment.
Q4: Are biosimilars or digital therapeutics impacting citalopram's sales?
Biosimilars are less relevant for small molecule drugs like citalopram, but digital therapeutics could gradually influence prescribing practices by offering non-pharmaceutical options.
Q5: What is the potential impact of new antidepressants on citalopram’s market?
Innovative agents with improved efficacy and fewer side effects could erode citalopram's market share further, especially if they secure better reimbursement and adoption in clinical guidelines.
8. Key Takeaways
- Citalopram hydrobromide’s global sales have declined approximately 80% since patent expiry due to widespread generic competition.
- The current market is predominantly composed of generics, with prices steadily declining.
- Future prospects hinge on regional market dynamics, evolving treatment paradigms, and the emergence of new therapeutic agents.
- Despite declining sales, citalopram remains a cost-effective option for depression management in developing regions.
- Market entry barriers for new competitors are low, but high generic penetration limits margins for branded products.
References
- Market research sources: IQVIA, EvaluatePharma, global epidemiology reports
- Regulatory databases: FDA, EMA
- Patent and exclusivity data: U.S. Patent and Trademark Office, European Patent Office
- Academic and industry journals: Journal of Clinical Psychiatry, Pharmacoeconomics
- Company reports and disclosures: Teva, Mylan, and others (2020–2022)
This comprehensive analysis aims to inform business strategy, R&D investments, and competitive positioning within the citalopram market landscape.