Last updated: February 19, 2026
What is CIPRO and its Therapeutic Area?
CIPRO is an antibiotic belonging to the fluoroquinolone class. Its primary therapeutic use is in the treatment of a wide range of bacterial infections. These infections include, but are not limited to, urinary tract infections, respiratory tract infections, skin and soft tissue infections, bone and joint infections, and certain types of gastrointestinal infections. CIPRO is effective against a broad spectrum of Gram-positive and Gram-negative bacteria, including many strains resistant to other antibiotics. The active pharmaceutical ingredient is ciprofloxacin.
What is the Patent Landscape for CIPRO?
The original patents for ciprofloxacin, developed by Bayer, have long expired. Key patents covering the composition of matter and method of use for ciprofloxacin began to expire in the late 1990s and early 2000s. For instance, U.S. Patent No. 4,670,444, which claimed the compound itself, expired in December 2003 [1]. Other foundational patents related to its synthesis and specific indications also expired subsequently.
Despite the expiration of primary patents, secondary patents and evergreening strategies have been employed by originator companies and generic manufacturers. These can include patents on:
- New salt forms or polymorphs: Altering the crystalline structure or salt form of ciprofloxacin can lead to new patentable entities with potentially improved bioavailability or stability.
- New formulations: Extended-release formulations, intravenous preparations, or combination therapies involving ciprofloxacin may be subject to separate patent protection.
- New indications: Discoveries of ciprofloxacin's efficacy against previously unrecognized bacterial strains or in novel disease states could be patented.
- Manufacturing processes: Novel or improved methods for synthesizing ciprofloxacin or its intermediates can also be patented.
As of late 2023, the core composition of matter patents have expired globally, allowing for widespread generic competition. However, vigilance regarding specific formulation or process patents is necessary for market participants.
Who are the Key Manufacturers and Competitors?
The market for ciprofloxacin is characterized by significant generic competition. The originator, Bayer, previously held the primary market share with its brand name product, Cipro. Following patent expirations, numerous generic manufacturers have entered the market.
Key players in the ciprofloxacin market include:
- Bayer AG: The originator, still maintains some presence with its branded product, though generic competition has significantly eroded its market share in many regions.
- Teva Pharmaceutical Industries Ltd.: A major global generic pharmaceutical company with a significant ciprofloxacin offering.
- Dr. Reddy's Laboratories: An Indian multinational pharmaceutical company with a broad portfolio of generic antibiotics, including ciprofloxacin.
- Sun Pharmaceutical Industries Ltd.: Another leading Indian pharmaceutical company with a substantial generic drug pipeline and market presence.
- Cipla Ltd.: An Indian multinational pharmaceutical company that is a significant producer of generic medicines, including ciprofloxacin.
- Mylan N.V. (now part of Viatris): A global healthcare company with a broad generic portfolio.
- Lupin Limited: An Indian multinational pharmaceutical company with a strong focus on generics.
These companies compete primarily on price, formulation (e.g., immediate release vs. extended release), and market access. The availability of multiple generic suppliers leads to intense price competition.
What is the Current Market Size and Projected Growth?
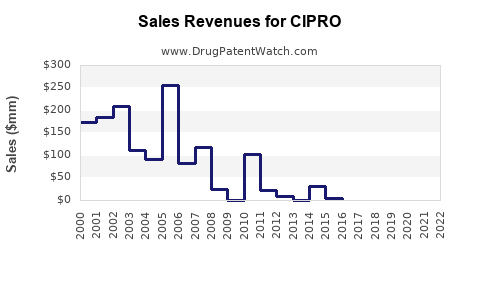
The global market for ciprofloxacin, while mature, remains substantial due to its broad utility and cost-effectiveness as a broad-spectrum antibiotic. Precise market size figures fluctuate based on reporting methodologies and currency conversions, but industry estimates place the global market for ciprofloxacin in the hundreds of millions of U.S. dollars annually.
Projected Growth:
The growth trajectory for ciprofloxacin is generally modest to flat, with potential for slight decline in developed markets. This is due to several factors:
- Genericization: The market is largely dominated by generics, leading to price erosion.
- Antibiotic Stewardship Programs: Increased focus on responsible antibiotic use to combat antimicrobial resistance (AMR) can lead to more targeted prescribing, potentially reducing overall ciprofloxacin usage for less severe or specific infections.
- Emergence of Newer Antibiotics: Development of novel antibiotics with broader spectra, improved safety profiles, or efficacy against resistant strains can displace older antibiotics like ciprofloxacin in certain clinical settings.
- Resistance Development: Increasing rates of bacterial resistance to fluoroquinolones, including ciprofloxacin, can limit its effectiveness and lead to its replacement with alternative therapies.
However, growth in emerging markets may offset declines in developed regions. Factors driving this include:
- Increasing Access to Healthcare: Improved healthcare infrastructure and affordability in developing countries lead to greater demand for essential medicines.
- Higher Prevalence of Bacterial Infections: Endemic infectious diseases may continue to drive demand.
- Cost-Effectiveness: Ciprofloxacin remains a cost-effective treatment option for many common infections, making it a preferred choice where budget is a constraint.
Estimated Market Size (Global Ciprofloxacin):
- 2023 Estimate: \$400 million - \$600 million USD [2]
- Projected CAGR (2024-2028): -1% to +2% [3]
This subdued growth reflects the mature nature of the drug and the challenges posed by AMR and newer therapeutic options.
What are the Key Market Drivers and Restraints?
Market Drivers:
- Broad-Spectrum Efficacy: Ciprofloxacin's effectiveness against a wide range of Gram-negative and some Gram-positive bacteria makes it a go-to treatment for numerous infections.
- Cost-Effectiveness: As a widely available generic, ciprofloxacin is an affordable treatment option, particularly crucial in resource-limited settings.
- Established Clinical Track Record: Decades of use have provided extensive clinical data on its efficacy and safety profiles.
- High Prevalence of Bacterial Infections: The persistent global burden of bacterial infections ensures ongoing demand.
- Growth in Emerging Markets: Expanding healthcare access and prevalence of infectious diseases in developing nations contribute to sustained demand.
- Use in Specific Indications: Its role in treating complicated urinary tract infections, prostatitis, and certain sexually transmitted infections continues to be significant.
Market Restraints:
- Antimicrobial Resistance (AMR): Growing resistance to fluoroquinolones limits ciprofloxacin's utility and necessitates the use of alternative, often more expensive, treatments.
- Adverse Event Profile: Fluoroquinolones are associated with serious adverse events, including tendinopathy, peripheral neuropathy, and central nervous system effects. Regulatory agencies have issued warnings and restrictions on their use for less severe infections.
- Antibiotic Stewardship Initiatives: Programs promoting judicious antibiotic use to preserve efficacy can lead to decreased prescription rates.
- Availability of Newer Antibiotics: The development of newer agents with improved safety, efficacy against resistant strains, or more targeted action can displace ciprofloxacin.
- Competition from Other Fluoroquinolones: While ciprofloxacin is a leader, other fluoroquinolones (e.g., levofloxacin) compete for similar indications.
- Regulatory Scrutiny: Increased regulatory oversight regarding the risks and benefits of fluoroquinolones can impact prescribing patterns.
What is the Pricing and Reimbursement Landscape?
The pricing of ciprofloxacin is largely driven by the generic market and is subject to intense competition.
- Generic Pricing: The price of generic ciprofloxacin is significantly lower than its originator brand. Prices vary by manufacturer, dosage form (tablets, oral suspension, intravenous), strength, and quantity. Generic ciprofloxacin tablets can range from \$0.05 to \$0.50 per tablet for common dosages, depending on the supplier and volume. Intravenous formulations are typically more expensive.
- Reimbursement: Ciprofloxacin is a widely reimbursed drug by most public and private health insurance plans globally. It is typically classified as a formulary drug.
- United States: Medicare, Medicaid, and most commercial insurers cover generic ciprofloxacin. Preferred status on formularies is common due to its cost-effectiveness.
- Europe: National health services and private insurers reimburse ciprofloxacin based on national guidelines and drug pricing regulations.
- Emerging Markets: Government health programs and out-of-pocket payments are common. Affordability is a key factor.
The pricing strategy for generic manufacturers focuses on achieving high volume through competitive bidding for tenders (e.g., hospital contracts, government supply agreements) and broad distribution. Price erosion is a continuous challenge.
What are the Regulatory Considerations?
Regulatory considerations for ciprofloxacin are multifaceted, focusing on safety, efficacy, and appropriate use due to concerns about antimicrobial resistance and adverse events.
- Antimicrobial Resistance (AMR): Regulatory bodies worldwide (e.g., FDA, EMA) have intensified their focus on AMR. This has led to:
- Restrictions on Use: For example, the U.S. FDA has advised against using fluoroquinolones for uncomplicated infections (e.g., sinusitis, acute bronchitis, uncomplicated urinary tract infections) when other treatment options are available, due to the risk of serious side effects outweighing the benefits [4].
- Labeling Changes: Updates to drug labels reflecting new safety information and recommended uses.
- Safety Warnings and Black Box Warnings: Fluoroquinolones, including ciprofloxacin, carry warnings regarding serious side effects:
- Tendinitis and Tendon Rupture: The risk of tendinitis and tendon rupture is a significant concern.
- Peripheral Neuropathy: Permanent nerve damage can occur.
- Central Nervous System Effects: Including seizures, dizziness, and psychiatric reactions.
- Aortic Aneurysm and Dissection: Recent studies and regulatory reviews have highlighted an increased risk of aortic aneurysm and dissection with fluoroquinolone use [5].
- Generic Drug Approval: Generic manufacturers must demonstrate bioequivalence to the reference listed drug (RLD) and meet quality standards.
- Manufacturing and Quality Control: Strict adherence to Good Manufacturing Practices (GMP) is required for all ciprofloxacin manufacturing facilities.
- Pharmacovigilance: Ongoing monitoring of adverse events is crucial. Manufacturers are required to report post-market safety data to regulatory authorities.
- Antibiotic Stewardship Mandates: Public health initiatives and regulatory pressures encourage healthcare systems to implement antibiotic stewardship programs, which can influence prescribing patterns for ciprofloxacin.
What are the Future Market Outlook and Strategic Opportunities?
The future market for ciprofloxacin will be shaped by the interplay of its established utility and the growing challenges of AMR and evolving regulatory landscapes.
Future Market Outlook:
- Continued Demand in Specific Niches: Ciprofloxacin will likely retain its position for certain severe or complicated infections where it remains a cost-effective and efficacious option, particularly in regions with high bacterial infection rates.
- Decline in Uncomplicated Infections: Its use in uncomplicated infections is expected to continue to decline in markets with strong antibiotic stewardship and access to alternative therapies.
- Geographic Divergence: Demand will likely be more robust in emerging markets due to lower cost and persistent infectious disease burdens, while developed markets may see a plateau or decline.
- Focus on Resistance Management: The long-term viability will depend on strategies to mitigate resistance, which may involve more targeted use or combination therapies.
Strategic Opportunities:
- Optimized Formulations for Emerging Markets: Developing and distributing affordable, stable formulations suitable for challenging supply chain environments in emerging economies.
- Combination Therapies: Exploring and developing ciprofloxacin-based combination therapies to enhance efficacy against resistant strains or reduce the risk of resistance development.
- Enhanced Diagnostic Integration: Partnering with diagnostic companies to develop rapid diagnostics that can identify ciprofloxacin-susceptible infections, enabling more targeted prescribing.
- Cost-Efficient Manufacturing: Continuous improvement in manufacturing processes to maintain competitive pricing in the highly commoditized generic market.
- Pediatric Formulations: While ciprofloxacin is used in pediatric populations, ensuring availability of palatable and appropriate pediatric formulations can be a niche opportunity.
- Market Access in Underserved Regions: Establishing strong distribution networks and partnerships to ensure availability in regions with high unmet needs for essential antibiotics.
Key Takeaways
Ciprofloxacin is a mature, broad-spectrum antibiotic facing significant market challenges from antimicrobial resistance (AMR) and evolving regulatory landscapes, alongside robust generic competition. While its use in uncomplicated infections is declining in developed markets due to safety concerns and stewardship programs, it retains substantial value for treating specific severe infections, particularly in emerging economies where cost-effectiveness and broad applicability remain critical drivers. The market size is in the hundreds of millions of dollars, with modest to flat projected growth globally, though specific regional trends may diverge. Strategic opportunities lie in optimizing formulations for emerging markets, exploring combination therapies, enhancing diagnostic integration, and maintaining cost-efficient manufacturing.
FAQs
1. Has the patent on CIPRO (ciprofloxacin) expired?
Yes, the original composition of matter patents for ciprofloxacin expired many years ago, primarily in the late 1990s and early 2000s. This has led to widespread generic availability.
2. What are the primary concerns regarding CIPRO's safety?
Key safety concerns include tendinitis and tendon rupture, peripheral neuropathy, central nervous system effects, and a potential increased risk of aortic aneurysm and dissection.
3. Why is antimicrobial resistance (AMR) a significant challenge for CIPRO?
AMR reduces the effectiveness of ciprofloxacin, necessitating the use of alternative treatments and contributing to its restricted use for less severe infections.
4. Is CIPRO still prescribed for common infections like UTIs?
While still used for some urinary tract infections, regulatory bodies and stewardship programs advise against its use for uncomplicated UTIs when other options are available, due to safety concerns and resistance.
5. What is the economic outlook for CIPRO in emerging markets?
The economic outlook in emerging markets is more positive than in developed regions, driven by affordability, continued prevalence of bacterial infections, and expanding healthcare access.
Citations
[1] U.S. Patent No. 4,670,444. (1987). Fluoroquinolone carboxylic acids. Bayer Aktiengesellschaft.
[2] Grand View Research. (2023). Ciprofloxacin Market Size, Share & Trends Analysis Report By Type, By Application, By Region, And Segment Forecasts, 2023-2030. Data compiled from industry reports.
[3] Market Research Future. (2023). Ciprofloxacin Market: Global Key Trends, Share, Industry Demand, Growth, Opportunities and Forecast to 2030. Data compiled from industry reports.
[4] U.S. Food and Drug Administration. (2018, December 11). FDA Drug Safety Communication: FDA advises restricting fluoroquinolone antibiotic use for uncomplicated sinusitis, acute bacterial exacerbation of chronic bronchitis, and acute bacterial rhinosinusitis. Retrieved from https://www.fda.gov/drugs/drug-safety-and-availability/fda-drug-safety-communication-fda-advises-restricting-fluoroquinolone-antibiotic-use-uncomplicated
[5] U.S. Food and Drug Administration. (2018, December 11). FDA Drug Safety Communication: FDA updates warnings for fluoroquinolone antibiotics. Retrieved from https://www.fda.gov/drugs/drug-safety-and-availability/fda-updates-warnings-fluoroquinolone-antibiotics