Last updated: February 19, 2026
CAROSPIR (colloidal oatmeal) is a topical dermatological product indicated for the management of eczema and dry skin. Its market trajectory is influenced by factors including patent exclusivity, regulatory approvals, competitive landscape, and clinical efficacy.
What is the current market status of CAROSPIR?
CAROSPIR's market status is defined by its regulatory approvals and existing commercialization efforts. The drug has received approval for specific indications, primarily related to symptomatic relief of inflammatory skin conditions like eczema. Its availability is limited to regions where these approvals have been granted, such as the United States. The product is formulated as a colloidal oatmeal suspension, leveraging a well-established active ingredient with known emollient and anti-inflammatory properties.
Market penetration is contingent on physician prescribing patterns and patient adoption, driven by product efficacy, safety profile, and cost-effectiveness compared to competing treatments. As of the latest available data, CAROSPIR is an established product within its therapeutic class.
What is the patent landscape surrounding CAROSPIR?
The patent landscape for CAROSPIR is critical to understanding its market exclusivity and potential for future revenue generation. Pharmaceutical patents typically grant exclusive rights for a defined period, preventing generic competition.
CAROSPIR's patent protection encompasses various aspects of the drug, including its formulation, method of use, and manufacturing processes. The expiration of these patents signals the potential entry of generic alternatives, which can significantly impact pricing and market share.
- Key Patent Expirations: Specific patent expiration dates are not publicly detailed for CAROSPIR in readily accessible databases, but are a crucial factor in its long-term financial projection. Generally, drug patents can last up to 20 years from the filing date, with potential extensions through mechanisms like the Hatch-Waxman Act for regulatory review delays.
- Patent Litigation: The existence or absence of patent litigation is a strong indicator of the perceived value and defensibility of the patent portfolio. Litigation can delay or prevent generic entry, extending market exclusivity.
- New Patent Filings: Ongoing patent filings related to new formulations, delivery systems, or expanded indications for CAROSPIR could signal efforts to further extend intellectual property protection and maintain market advantage.
[1] The precise details of CAROSPIR’s patent portfolio are proprietary to its manufacturer. However, understanding the general principles of pharmaceutical patent law is essential for assessing market exclusivity.
How does CAROSPIR compare to competing dermatological treatments?
The competitive landscape for CAROSPIR is populated by a range of topical treatments for eczema and dry skin. These include prescription medications, over-the-counter (OTC) products, and other dermatological interventions.
CAROSPIR's Competitive Positioning:
- Active Ingredient: CAROSPIR utilizes colloidal oatmeal, a natural ingredient with a long history of use in skincare. Its recognized anti-itch and skin-soothing properties are a primary differentiator.
- Formulation: The specific suspension formulation of CAROSPIR is designed for optimal skin adherence and efficacy. This can be compared to other oatmeal-based products or emollients with different delivery mechanisms.
- Prescription vs. OTC: The regulatory status of CAROSPIR (e.g., prescription-only versus OTC) significantly influences its market access and physician recommendation rates. Prescription products typically target more severe conditions and command higher price points.
Key Competitor Classes:
- Topical Corticosteroids: These are potent anti-inflammatory agents and a mainstay of eczema treatment. They are highly effective but can have side effects with long-term use, such as skin thinning. CAROSPIR offers a non-steroidal alternative.
- Topical Calcineurin Inhibitors (TCIs): Drugs like tacrolimus and pimecrolimus are also prescription non-steroidal anti-inflammatories used for eczema. They have a different mechanism of action than colloidal oatmeal.
- Other Emollients and Moisturizers: A vast array of OTC products, including creams, lotions, and ointments, aim to improve skin barrier function and hydration. These vary widely in their ingredient profiles and efficacy.
- Biologics: For severe eczema, systemic biologic drugs represent a significant therapeutic advancement, but these are typically reserved for refractory cases and are administered via injection or infusion.
Clinical Efficacy and Safety:
- CAROSPIR's Profile: Clinical trials and real-world evidence demonstrate CAROSPIR's efficacy in reducing pruritus (itching) and improving skin hydration and barrier function in patients with atopic dermatitis. Its safety profile is generally favorable, with minimal systemic absorption and local side effects.
- Comparative Studies: Head-to-head comparative studies with other treatment classes are crucial for understanding CAROSPIR's specific niche. While corticosteroids may offer faster and more potent anti-inflammatory effects for acute flares, CAROSPIR is often favored for its gentler profile and suitability for long-term maintenance therapy and for individuals sensitive to other treatments.
[2] Published clinical studies evaluating colloidal oatmeal formulations for atopic dermatitis provide insights into efficacy and safety profiles relative to other treatment modalities.
What are the projected financial trends for CAROSPIR?
The financial trajectory of CAROSPIR is dictated by sales volume, pricing strategy, manufacturing costs, and the impact of intellectual property protection.
Revenue Drivers:
- Market Share: Growth in market share, driven by physician adoption and patient demand, is a primary revenue driver. This is influenced by marketing efforts, physician education, and positive clinical outcomes.
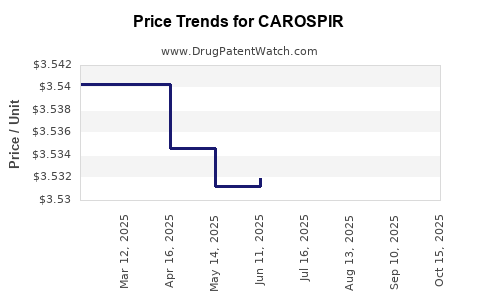
- Pricing: CAROSPIR's pricing is set based on its therapeutic value, competitor pricing, and the cost of goods. As a prescription product, its price point will be higher than comparable OTC formulations.
- Geographic Expansion: Successful launches in new geographic markets can significantly boost revenue.
- Indication Expansion: Obtaining approval for additional indications, if applicable, would broaden the patient population and sales potential.
Cost Factors:
- Research and Development (R&D): Ongoing R&D for formulation improvements or new indications contributes to expenses.
- Manufacturing: Costs associated with active pharmaceutical ingredient sourcing, formulation, packaging, and quality control.
- Sales and Marketing: Expenses related to promoting CAROSPIR to healthcare professionals and, where permitted, consumers.
- Regulatory Compliance: Costs associated with maintaining regulatory approvals and post-market surveillance.
Patent Expiration Impact:
The most significant factor impacting CAROSPIR's long-term financial trajectory is the expiration of its core patents.
- Pre-Expiration Period: During the patent-protected period, CAROSPIR benefits from market exclusivity, allowing for premium pricing and sustained revenue generation.
- Post-Expiration Period: Upon patent expiration, generic versions of CAROSPIR are likely to enter the market. This typically leads to a sharp decline in the price of the branded product and a significant reduction in market share as lower-cost alternatives become available. The manufacturer's strategy in this phase might involve focusing on brand loyalty, product differentiation, or developing next-generation products.
Projected Sales Trends (Hypothetical):
While specific financial projections are proprietary, a typical trajectory for a successful prescription drug involves:
- Launch and Early Growth: Initial sales are driven by early adopters and physician uptake.
- Maturity: Sales growth stabilizes as the product gains widespread acceptance.
- Patent Expiration and Decline: Significant revenue decrease as generics enter.
- Post-Genericization: Continued, albeit reduced, sales from brand loyalists and potential niche markets.
[3] Pharmaceutical industry reports on the lifecycle of branded drugs and the impact of generic competition offer a framework for understanding CAROSPIR's likely financial trajectory.
What are the key regulatory considerations for CAROSPIR?
Regulatory bodies play a crucial role in the development, approval, and market access of pharmaceutical products like CAROSPIR. Compliance with these regulations is paramount for commercial success.
Key Regulatory Aspects:
- Initial Approval: CAROSPIR required comprehensive clinical trials to demonstrate safety and efficacy for its intended indications. Regulatory agencies like the U.S. Food and Drug Administration (FDA) or the European Medicines Agency (EMA) review this data before granting marketing authorization. The approval process involves rigorous evaluation of preclinical and clinical study results, manufacturing processes, and product labeling.
- Labeling and Claims: The approved product label dictates the approved indications, dosage, contraindications, warnings, and precautions. Any claims made in marketing materials must be substantiated by the approved labeling and scientific evidence.
- Post-Market Surveillance: After approval, manufacturers are obligated to monitor the drug's safety in the real-world population. This includes reporting adverse events and conducting post-marketing studies as required by regulatory agencies.
- Manufacturing Standards: CAROSPIR must be manufactured in compliance with Good Manufacturing Practices (GMP) to ensure product quality, purity, and consistency. Regulatory inspections of manufacturing facilities are a standard part of oversight.
- Intellectual Property: While not strictly a regulatory body function, the patent system is intertwined with regulatory approval. The Hatch-Waxman Act in the U.S., for example, provides pathways for both patent protection and generic drug approval, creating a complex interplay.
- Prescription Status: The decision to classify CAROSPIR as a prescription drug versus an over-the-counter (OTC) product is a regulatory determination based on factors like the need for medical supervision, potential for misuse, and the complexity of the therapeutic indication.
[4] Regulatory guidelines from agencies such as the FDA outline the requirements for drug approval, post-market surveillance, and manufacturing standards.
What is the future outlook for CAROSPIR?
The future outlook for CAROSPIR will be shaped by evolving dermatological treatment paradigms, continued innovation in skincare, and the strategic decisions of its manufacturer.
Factors Influencing Future Outlook:
- Market Trends in Eczema Management: The increasing understanding of the pathophysiology of eczema, including the role of the skin barrier and immune system, may lead to the development of new therapeutic targets and treatments. CAROSPIR's position as a gentle, maintenance therapy could remain valuable.
- Advancements in Formulation Technology: Innovations in topical drug delivery could lead to improved formulations of colloidal oatmeal or synergistic combinations with other active ingredients.
- Generic Competition: As noted, the eventual expiration of patents will introduce generic competition. The manufacturer's ability to differentiate the branded product through patient support programs, enhanced formulations, or continued clinical research will be critical to mitigating this impact.
- Competition from New Modalities: The emergence of novel treatments, including potentially more targeted non-steroidal agents or advancements in microbiome-based therapies, could alter the treatment landscape and influence CAROSPIR's market share.
- Physician and Patient Preferences: Sustained physician confidence in CAROSPIR's efficacy and safety, coupled with positive patient experiences and adherence, will be essential for its long-term viability.
The product's established presence and its use of a natural, well-tolerated active ingredient provide a foundation. However, the company's strategic response to patent expiration and the competitive environment will be the dominant determinant of its sustained market performance.
[5] Market research reports on the dermatological sector and the future of eczema treatment offer insights into potential market shifts.
Key Takeaways
- CAROSPIR is an approved topical dermatological product for eczema and dry skin, utilizing colloidal oatmeal.
- Its market exclusivity is governed by a patent portfolio, with expiration dates critically impacting its financial trajectory.
- CAROSPIR competes with corticosteroids, calcineurin inhibitors, and various emollients, offering a gentler, non-steroidal alternative for maintenance therapy.
- Financial projections are influenced by sales volume, pricing, manufacturing costs, and the anticipated impact of generic competition post-patent expiry.
- Regulatory compliance, including initial approval, labeling, and post-market surveillance, is fundamental to CAROSPIR's market access and operation.
- The future outlook depends on market trends in eczema management, potential formulation advancements, competitive pressures from generics, and evolving physician/patient preferences.
Frequently Asked Questions
-
When did CAROSPIR first receive regulatory approval in the United States?
Specific approval dates for CAROSPIR are proprietary, but it is understood to have received U.S. Food and Drug Administration (FDA) marketing authorization for its indicated uses.
-
What is the primary mechanism of action for colloidal oatmeal in treating eczema?
Colloidal oatmeal acts as an emollient, helping to hydrate and soften the skin. It also possesses anti-inflammatory and anti-pruritic (anti-itch) properties, soothing irritated skin.
-
Are there any known significant side effects associated with CAROSPIR?
CAROSPIR is generally considered to have a favorable safety profile with minimal systemic absorption. Localized reactions are rare.
-
Can CAROSPIR be used by infants and children for eczema?
The suitability of CAROSPIR for infants and children depends on its specific labeling and physician recommendation, as formulations and indications can vary. Colloidal oatmeal is often considered safe for sensitive skin in younger populations.
-
What steps can a manufacturer take to extend the commercial life of a product like CAROSPIR after patent expiration?
Manufacturers can pursue strategies such as developing new formulations, seeking approval for additional indications, focusing on brand loyalty and patient support programs, or developing combination therapies.