BREYNA Drug Patent Profile
✉ Email this page to a colleague
When do Breyna patents expire, and what generic alternatives are available?
Breyna is a drug marketed by Mylan and is included in one NDA.
The generic ingredient in BREYNA is budesonide; formoterol fumarate dihydrate. There are twenty-two drug master file entries for this compound. Four suppliers are listed for this compound. Additional details are available on the budesonide; formoterol fumarate dihydrate profile page.
AI Deep Research
Questions you can ask:
- What is the 5 year forecast for BREYNA?
- What are the global sales for BREYNA?
- What is Average Wholesale Price for BREYNA?
Summary for BREYNA
| US Patents: | 0 |
| Applicants: | 1 |
| NDAs: | 1 |
| Finished Product Suppliers / Packagers: | 1 |
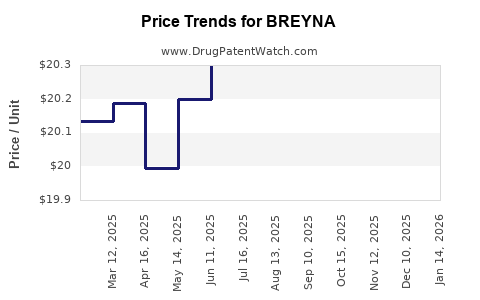
| Drug Prices: | Drug price information for BREYNA |
| DailyMed Link: | BREYNA at DailyMed |
Pharmacology for BREYNA
| Drug Class | Corticosteroid beta2-Adrenergic Agonist |
| Mechanism of Action | Adrenergic beta2-Agonists Corticosteroid Hormone Receptor Agonists |
US Patents and Regulatory Information for BREYNA
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Mylan | BREYNA | budesonide; formoterol fumarate dihydrate | AEROSOL, METERED;INHALATION | 211699-001 | Mar 15, 2022 | AB | RX | No | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | |||
| Mylan | BREYNA | budesonide; formoterol fumarate dihydrate | AEROSOL, METERED;INHALATION | 211699-002 | Mar 15, 2022 | AB | RX | No | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | |||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
EU/EMA Drug Approvals for BREYNA
| Company | Drugname | Inn | Product Number / Indication | Status | Generic | Biosimilar | Orphan | Marketing Authorisation | Marketing Refusal |
|---|---|---|---|---|---|---|---|---|---|
| Teva Pharma B.V. | Budesonide/Formoterol Teva Pharma B.V. | budesonide, formoterol fumarate dihydrate | EMEA/H/C/004882Budesonide/Formoterol Teva Pharma B.V. is indicated in adults 18 years of age and older only.AsthmaBudesonide/Formoterol Teva Pharma B.V. is indicated in the regular treatment of asthma, where use of a combination (inhaled corticosteroid and long-acting β2 adrenoceptor agonist) is appropriate:-in patients not adequately controlled with inhaled corticosteroids and “as needed” inhaled short-acting β2 adrenoceptor agonists.or-in patients already adequately controlled on both inhaled corticosteroids and long-acting β2 adrenoceptor agonists.COPDSymptomatic treatment of patients with COPD with forced expiratory volume in 1 second (FEV1) | Authorised | no | no | no | 2020-04-03 | |
| Teva Pharma B.V. | Budesonide/Formoterol Teva Pharma B.V. | budesonide, formoterol fumarate dihydrate | EMEA/H/C/003953Budesonide/Formoterol Teva Pharma B.V. is indicated in adults 18 years of age and older only.AsthmaBudesonide/Formoterol Teva Pharma B.V. is indicated in the regular treatment of asthma, where use of a combination (inhaled corticosteroid and long-acting β2 adrenoceptor agonist) is appropriate: orin patients not adequately controlled with inhaled corticosteroids and “as needed” inhaled short-acting β2 adrenoceptor agonists.in patients already adequately controlled on both inhaled corticosteroids and long-acting β2 adrenoceptor agonists. | Withdrawn | no | no | no | 2014-11-19 | |
| >Company | >Drugname | >Inn | >Product Number / Indication | >Status | >Generic | >Biosimilar | >Orphan | >Marketing Authorisation | >Marketing Refusal |
Market Dynamics and Financial Trajectory for BREYNA
More… ↓