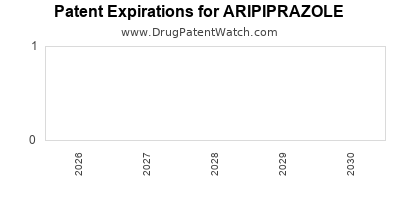
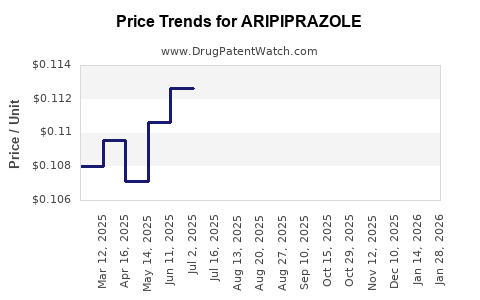
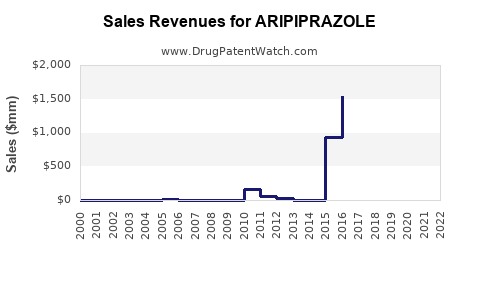
ARIPIPRAZOLE Drug Patent Profile
✉ Email this page to a colleague
Which patents cover Aripiprazole, and what generic alternatives are available?
Aripiprazole is a drug marketed by Mylan, Amneal Pharms, Apotex, Aurobindo Pharma Ltd, Chartwell Rx, Hetero Labs Ltd Iii, Lannett Co Inc, Quagen, Rubicon Research, Vistapharm Llc, Alembic, Aurobindo Pharma, Orbion Pharms, Sciegen Pharms, Square Pharms, Accord Hlthcare, Aiping Pharm Inc, Ajanta Pharma Ltd, Alkem Labs Ltd, Hetero Labs Ltd V, Lupin, Macleods Pharms Ltd, Prinston Inc, Rising, Sunshine, Teva Pharms Usa, Torrent, Unichem, and Zydus Pharms. and is included in thirty-seven NDAs.
The generic ingredient in ARIPIPRAZOLE is aripiprazole. There are forty-nine drug master file entries for this compound. Forty-nine suppliers are listed for this compound. Additional details are available on the aripiprazole profile page.
DrugPatentWatch® Litigation and Generic Entry Outlook for Aripiprazole
A generic version of ARIPIPRAZOLE was approved as aripiprazole by ALEMBIC on April 28th, 2015.
AI Deep Research
Questions you can ask:
- What is the 5 year forecast for ARIPIPRAZOLE?
- What are the global sales for ARIPIPRAZOLE?
- What is Average Wholesale Price for ARIPIPRAZOLE?
Summary for ARIPIPRAZOLE
Recent Clinical Trials for ARIPIPRAZOLE
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| University Hospital, Strasbourg, France | PHASE2 |
| University of California, San Francisco | PHASE1 |
| University of Washington | PHASE1 |
Pharmacology for ARIPIPRAZOLE
| Drug Class | Atypical Antipsychotic |
Medical Subject Heading (MeSH) Categories for ARIPIPRAZOLE
Anatomical Therapeutic Chemical (ATC) Classes for ARIPIPRAZOLE
Paragraph IV (Patent) Challenges for ARIPIPRAZOLE
| Tradename | Dosage | Ingredient | Strength | NDA | ANDAs Submitted | Submissiondate |
|---|---|---|---|---|---|---|
| ABILIFY MAINTENA KIT | Extended-release Injectable Suspension | aripiprazole | 300 mg/vial and 400 mg/vial | 202971 | 1 | 2021-12-20 |
| ABILIFY | Oral Solution | aripiprazole | 1 mg/mL | 021713 | 1 | 2007-12-20 |
| ABILIFY | Tablets | aripiprazole | 2 mg, 5 mg, 10 mg, 15 mg, 20 mg and 30 mg | 021436 | 8 | 2006-11-15 |
| ABILIFY | Orally Disintegrating Tablets | aripiprazole | 10 mg, 15 mg, 20 mg and 30 mg | 021729 | 1 | 2006-11-15 |
US Patents and Regulatory Information for ARIPIPRAZOLE
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Hetero Labs Ltd V | ARIPIPRAZOLE | aripiprazole | TABLET;ORAL | 205064-001 | Apr 28, 2015 | AB | RX | No | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | |||
| Prinston Inc | ARIPIPRAZOLE | aripiprazole | TABLET;ORAL | 205363-006 | Dec 4, 2017 | AB | RX | No | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | |||
| Ajanta Pharma Ltd | ARIPIPRAZOLE | aripiprazole | TABLET;ORAL | 206174-006 | Sep 12, 2016 | AB | RX | No | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | |||
| Aiping Pharm Inc | ARIPIPRAZOLE | aripiprazole | TABLET;ORAL | 091279-003 | Jan 9, 2017 | DISCN | No | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | ||||
| Macleods Pharms Ltd | ARIPIPRAZOLE | aripiprazole | TABLET;ORAL | 204111-003 | Oct 7, 2016 | AB | RX | No | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | |||
| Sunshine | ARIPIPRAZOLE | aripiprazole | TABLET;ORAL | 213037-001 | Oct 2, 2023 | AB | RX | No | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | |||
| Alkem Labs Ltd | ARIPIPRAZOLE | aripiprazole | TABLET;ORAL | 207105-002 | Feb 21, 2019 | AB | RX | No | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | |||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
EU/EMA Drug Approvals for ARIPIPRAZOLE
| Company | Drugname | Inn | Product Number / Indication | Status | Generic | Biosimilar | Orphan | Marketing Authorisation | Marketing Refusal |
|---|---|---|---|---|---|---|---|---|---|
| Mylan Pharmaceuticals Limited | Aripiprazole Mylan Pharma (previously Aripiprazole Pharmathen) | aripiprazole | EMEA/H/C/003803Aripiprazole Mylan Pharma is indicated for the treatment of schizophrenia in adults and in adolescents aged 15 years and older.Aripiprazole Mylan Pharma is indicated for the treatment of moderate to severe manic episodes in Bipolar I Disorder and for the prevention of a new manic episode in adults who experienced predominantly manic episodes and whose manic episodes responded to aripiprazole treatment.Aripiprazole Mylan Pharma is indicated for the treatment up to 12 weeks of moderate to severe manic episodes in Bipolar I Disorder in adolescents aged 13 years and older. | Authorised | yes | no | no | 2015-06-30 | |
| Otsuka Pharmaceutical Netherlands B.V. | Abilify | aripiprazole | EMEA/H/C/000471Abilify is indicated for the treatment of schizophrenia in adults and in adolescents aged 15 years and older.Abilify is indicated for the treatment of moderate to severe manic episodes in Bipolar I Disorder and for the prevention of a new manic episode in adults who experienced predominantly manic episodes and whose manic episodes responded to aripiprazole treatment.Abilify is indicated for the treatment up to 12 weeks of moderate to severe manic episodes in Bipolar I Disorder in adolescents aged 13 years and older. | Authorised | no | no | no | 2004-06-04 | |
| Otsuka Pharmaceutical Netherlands B.V. | Abilify Maintena | aripiprazole | EMEA/H/C/002755Maintenance treatment of schizophrenia in adult patients stabilised with oral aripiprazole. | Authorised | no | no | no | 2013-11-14 | |
| Accord Healthcare S.L.U. | Aripiprazole Accord | aripiprazole | EMEA/H/C/004021Aripiprazole Accord is indicated for the treatment of schizophrenia in adults and in adolescents aged 15 years and older., , Aripiprazole Accord is indicated for the treatment of moderate to severe manic episodes in Bipolar I Disorder and for the prevention of a new manic episode in adults who experienced predominantly manic episodes and whose manic episodes responded to aripiprazole treatment., , Aripiprazole Accord is indicated for the treatment up to 12 weeks of moderate to severe manic episodes in Bipolar I Disorder in adolescents aged 13 years and older., | Authorised | yes | no | no | 2015-11-15 | |
| Zentiva, k.s. | Aripiprazole Zentiva | aripiprazole | EMEA/H/C/003899Aripiprazole Zentiva is indicated for the treatment of schizophrenia in adults and in adolescents aged 15 years and older.Aripiprazole Zentiva is indicated for the treatment of moderate to severe manic episodes in Bipolar I Disorder and for the prevention of a new manic episode in adults who experienced predominantly manic episodes and whose manic episodes responded to aripiprazole treatment.Aripiprazole Zentiva is indicated for the treatment up to 12 weeks of moderate to severe manic episodes in Bipolar I Disorder in adolescents aged 13 years and older. | Authorised | yes | no | no | 2015-06-25 | |
| Sandoz GmbH | Aripiprazole Sandoz | aripiprazole | EMEA/H/C/004008Aripiprazole Sandoz is indicated for the treatment of schizophrenia in adults and in adolescents aged 15 years and older., , Aripiprazole Sandoz is indicated for the treatment of moderate to severe manic episodes in Bipolar I Disorder and for the prevention of a new manic episode in adults who experienced predominantly manic episodes and whose manic episodes responded to aripiprazole treatment., , Aripiprazole Sandoz is indicated for the treatment up to 12 weeks of moderate to severe manic episodes in Bipolar I Disorder in adolescents aged 13 years and older., | Authorised | yes | no | no | 2015-08-20 | |
| >Company | >Drugname | >Inn | >Product Number / Indication | >Status | >Generic | >Biosimilar | >Orphan | >Marketing Authorisation | >Marketing Refusal |
Market Dynamics and Financial Trajectory for Aripiprazole
More… ↓