Last updated: April 23, 2026
Aprepitant (oral/IV NK1 receptor antagonist) has a mature antiemetic market presence driven by chemotherapy-induced nausea and vomiting (CINV) protocols and guideline adherence. Commercial performance has shifted from broad early adoption toward life-cycle management across fixed-dose combinations, expanded regimen mix, and competitive pressure from generics and therapeutic alternatives.
What drives aprepitant demand and market access?
1) Clinical demand is anchored in CINV guideline algorithms
Aprepitant is used in multi-day and single-day CINV regimens, typically as part of NK1-receptor antagonist-based combinations with 5-HT3 receptor antagonists and corticosteroids. The commercial demand profile maps to chemotherapy utilization and the share of patients treated with emetogenic regimens where NK1-based prophylaxis is recommended.
2) Formulation and regimen fit affect payer adoption
Market access depends on formulary placement for:
- NK1 antagonist component in CINV pathways
- Route of administration for institutional workflows (IV vs oral), especially where outpatient infusion centers manage day-1 IV and subsequent oral dosing
3) Switching risk is structural
Aprepitant’s market is exposed to:
- Generic substitution after exclusivity expiry
- Therapeutic substitution from other CINV agents and evolving guideline preferences
- Biosimilar-like dynamics are not relevant, but small-molecule substitution and tender-led procurement are
How has the competitive landscape shaped pricing and share?
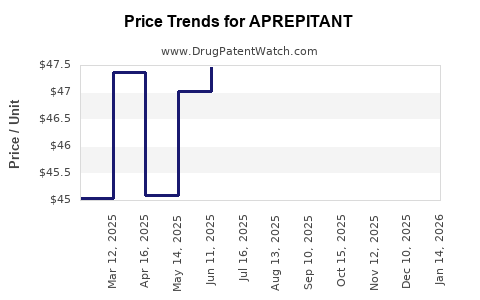
Generic entry and margin compression
Aprepitant’s trajectory follows the typical small-molecule antiemetic pattern:
- Early premium pricing during brand exclusivity
- Then price erosion post-generic entry
- Then revenue stabilization at lower price points driven by persistence of protocol usage and remaining brand differentiators (packaging, clinical familiarity, and administered regimen logistics)
Competitive alternatives
The CINV market includes multiple classes that can win specific patient or provider preferences:
- Other NK1 receptor antagonists and regimen structures
- 5-HT3 antagonists, dexamethasone regimens, and risk-adapted approaches
- Newer agents that can shift prescriber behavior by regimen simplicity, dosing convenience, or reimbursement economics
Net effect: brand unit demand often remains resilient due to guideline reinforcement, but realized revenue trends decline due to price compression.
What is the likely revenue trajectory across the life cycle?
Revenue path: exclusivity peak to stabilized low-price base
Aprepitant’s financial trajectory is best understood in two phases:
- Exclusivity and adoption phase: Revenue grows with clinical adoption as NK1-based prophylaxis becomes embedded in standard CINV regimens.
- Post-exclusivity stabilization phase: Units often persist, but realized revenue declines with generic entry and procurement-driven pricing.
Real-world revenue mechanics
In mature oncology supportive-care markets:
- Volume stays tied to chemotherapy mix and CINV prophylaxis penetration.
- Revenue follows a “price-first” adjustment after generic substitution, with volume declines often secondary unless substitution is aggressive.
What do global patent and exclusivity events imply for market timing?
Key patent framework (high-level)
Aprepitant is protected by a network of composition-of-matter and method/regimen-related intellectual property plus regulatory exclusivities in multiple jurisdictions. The commercial timing is typically governed by:
- Composition-of-matter expiration
- Secondary patents affecting formulations or specific dosing regimens
- Regulatory exclusivity (where applicable)
Practical implication
Once the legal barrier falls, price falls quickly in tender and national formulary systems. Aprepitant’s mature status means current market dynamics are primarily driven by:
- Generic share
- Contracting and pharmacy benefit manager (PBM) dynamics
- Continued protocol usage
Where does financial performance concentrate across channels?
1) Hospital and infusion center channels
Supportive oncology is procurement-heavy. Institutional formularies and conversion decisions are driven by:
- Tender economics
- Inventory and IV-to-oral switching workflow
- Budget impact analysis for antiemetic bundles
2) Specialty pharmacy and outpatient dispensing
Outpatient oncology typically relies on pharmacy networks and prior authorization patterns less than hospitals, but PBM contracting still drives net pricing:
- Brand vs generic tier placement
- Copay and coverage rules
- Step therapy where payer policies exist
3) International differences
Aprepitant market outcomes vary by:
- Date of generic approvals and local launches
- Reimbursement systems and tender regimes
- Local guideline adoption patterns
What financial outcomes typically follow for aprepitant brands?
Brand revenue tends to decline faster than unit demand after generics
For a mature antiemetic, the financial signature after generic introduction typically shows:
- Lower net sales due to steep price erosion
- Better-than-expected volume stability (protocol continuation)
- Increased reliance on remaining differentiation levers (pack size, physician familiarity, institutional contracting)
Pipeline and lifecycle tools
Companies usually respond with:
- Line extensions via dosing/formulation improvements
- Evidence packages supporting specific risk categories
- Marketing focus on regimen adherence and clinician trust
Market dynamics by segment: acute, delayed, and multi-day emesis control
Aprepitant’s clinical role extends across CINV phases (notably delayed nausea/vomiting in multi-day regimens). That matters for commercial performance because:
- Delayed CINV prophylaxis persistence is protocol-driven
- Multi-day regimen completion affects total patient exposure and repeat dispensing
- Any regimen redesign that reduces NK1 antagonist exposure can affect unit demand, even if initial prophylaxis remains
How does payer and reimbursement policy change realized pricing?
Net price compression mechanisms
Realized pricing for branded supportive oncology drugs commonly declines through:
- Formulary tier reclassification
- PBM rebate pressures
- Contracting tied to market share and generic parity
- Reference pricing in some reimbursement systems
Consequence for financial trajectory
Net sales usually track:
- A steep decline after generic entry
- A longer plateau at a lower price level if generic supply stabilizes and procurement locks in routine CINV pathway usage
What do investors and R&D teams look for now in aprepitant’s trajectory?
Key indicators
- Generic share in major markets (drives price floor)
- Uptake of protocol refinements that use NK1 antagonists in narrower or broader patient segments
- Tender price indexes in hospital systems
- Net-to-gross trends for the brand (rebates and contracting)
Commercial durability
Even as pricing declines, NK1 antagonists often retain a role because CINV prevention is a standard-of-care objective and chemotherapy providers face accountability for emesis outcomes.
Key Takeaways
- Aprepitant demand is anchored to CINV prophylaxis algorithms where NK1 antagonists remain part of standard multi-drug regimens.
- The financial trajectory is dominated by post-exclusivity pricing compression from generic entry, with unit volume typically more resilient than net pricing.
- Hospital procurement and PBM contracting determine realized price more than headline list pricing.
- Competitive alternatives influence share by regimen convenience and payer preference, but guideline reinforcement tends to sustain baseline NK1 antagonist utilization.
- Current market dynamics are best read through tender-led pricing, generic penetration, and protocol stability across delayed and multi-day emesis phases.
FAQs
-
Is aprepitant primarily driven by oncology supportive care rather than broader indications?
Yes. Commercial demand is most consistently tied to CINV prevention within chemotherapy supportive-care protocols.
-
What happens to revenue after generic entry for aprepitant?
Net sales usually decline faster than units because price compression drives revenue erosion, while protocol-driven use can keep volume relatively steadier.
-
Which channel most affects aprepitant’s realized pricing?
Hospital and infusion center procurement is typically the largest lever, followed by PBM and specialty pharmacy contracting.
-
Does regimen structure materially affect commercial performance?
Yes. Use across delayed and multi-day phases increases total exposure; any shift that narrows NK1 antagonist coverage can reduce total dosing.
-
What competitive forces matter most for aprepitant today?
Generic competition and tender/PBM contracting, plus therapeutic alternatives that can alter prescribing behavior within specific emetogenic risk categories.
References
[1] FDA. Emend (aprepitant) prescribing information. U.S. Food and Drug Administration. (Accessed via FDA labeling database).
[2] National Comprehensive Cancer Network (NCCN). Guidelines for Antiemesis. NCCN Clinical Practice Guidelines in Oncology.
[3] EMA. Product information for aprepitant-containing medicines. European Medicines Agency.