Last updated: April 25, 2026
How big is the vancomycin market and how is it moving?
Vancomycin is a mature, off-patent antibiotic with persistent demand driven by hospital infection control needs, rising antimicrobial resistance pressure, and ongoing treatment of serious Gram-positive infections. Commercially, the market tracks the hospital environment more than new-product innovation cycles.
Demand anchors
- Hospital and long-term care consumption: Vancomycin use is concentrated in inpatient settings (ICU, surgery, oncology, transplant units) and in institutions managing high-risk patients.
- Resistance and stewardship: MRSA prevalence and hospital infection control programs keep baseline utilization steady, while stewardship policies shape dosing and duration.
- Core clinical positioning: Vancomycin remains a standard-of-care option for serious suspected or confirmed Gram-positive infections where empiric coverage or MRSA risk exists.
Competitive structure
- Multiple authorized generic manufacturers: Pricing power is limited, and the market typically clears at competitive generic levels.
- Biosafety and supply stability considerations: Contract manufacturing capacity and sterile injectables supply can affect short-term availability and pricing.
What market forces are shaping pricing and share?
1) Generic erosion and tender-driven pricing
The largest determinant of financial trajectory for vancomycin is channel pricing in hospitals through:
- Group purchasing organization (GPO) contracts
- Institutional tenders
- Automatic substitution and formulary preference rules for injectables
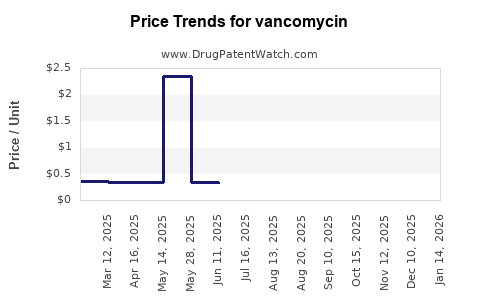
As more competitors participate and hospitals lock in multi-quarter procurement contracts, net price usually compresses unless supply constraints or regulatory actions intervene.
2) Formulation mix: vial size, concentration, and therapeutic substitution
Vancomycin’s financial profile depends on:
- Injection presentations (single-dose vials, vial strengths)
- Hospital pharmacy handling preferences
- Substitution by alternative agents when local antimicrobial guidelines shift (for example, when linezolid or daptomycin is preferred in specific scenarios)
3) Safety and dosing governance
Clinical governance can influence volume and duration:
- Protocol-driven dosing, therapeutic drug monitoring, and renal dosing adjustments can affect utilization patterns.
- Shifts in stewardship targets may reduce unnecessary courses but still preserve demand for severe infections.
4) Regulatory and manufacturing continuity
Vancomycin’s mature status means financial outcomes are sensitive to supply disruptions:
- Sterile injectable manufacturing interruptions can spike pricing temporarily.
- Regulatory issues affecting specific sites or product lots can change effective supply and raise short-term procurement costs.
How does product lifecycle affect vancomycin’s financial trajectory?
Vancomycin’s lifecycle is defined by:
- Low brand premium (limited patent-driven pricing)
- High-volume, low-margin procurement economics
- Continuous competitive replenishment by generics
This creates a financial trajectory pattern common to off-patent sterile injectables:
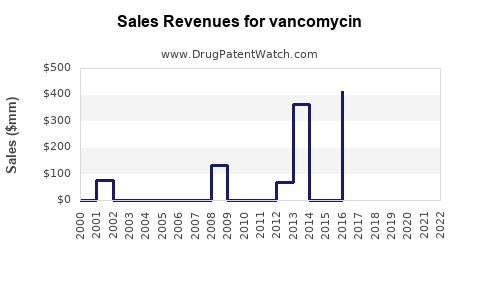
- Revenue tends to track volume (infection burden and hospital admissions)
- Profit pools shift to efficient manufacturing and contracting rather than differentiation
- Price compression dominates topline growth, unless volume expands faster than pricing declines
What are the key revenue and cost drivers?
Revenue drivers
- US and EU hospital discharge and ICU occupancy levels
- MRSA case mix and empiric therapy practices
- Infection control initiatives that increase antibiotic use for suspected bacterial infections
- Formulary status and contract renewals
Cost drivers
- Sterile manufacturing overhead (cleanroom, aseptic process controls, QC release)
- Raw materials and utility costs
- Quality systems and batch testing
- Regulatory compliance costs (inspection readiness)
- Working capital and logistics for cold-chain-adjacent storage requirements (where applicable)
How do financial outcomes typically vary by geography and channel?
US
- Large generic competitive set and strong contracting dynamics.
- Hospital formulary practices and PBM/GPO structures drive net pricing.
- Demand is stable with modest cyclical influence from admission rates.
Europe
- More frequent price regulation and tender-based procurement can create tighter net-price ranges.
- Uptake depends on reimbursement mechanisms and national hospital formularies.
Developing markets
- Lower price ceilings and procurement irregularities can create volume volatility.
- Supply chain quality and distributor networks materially affect continuity.
What is the competitive and substitution landscape?
Direct competitive alternatives
Vancomycin is routinely compared against other Gram-positive agents depending on:
- severity and site of infection (skin/soft tissue, bacteremia, pneumonia)
- MRSA risk
- renal impairment
- treatment monitoring capability
Common comparators in hospital protocols include linezolid, daptomycin, telavancin (market access dependent), and other beta-lactams for MRSA where appropriate.
Substitution pressure
Substitution does not eliminate demand for vancomycin because:
- many guidelines still include vancomycin for initial empiric coverage in severe infection
- stewardship practices often retain vancomycin as a fallback or confirmatory therapy
The net effect is that vancomycin typically faces share volatility at the margins, but it retains baseline volume.
What financial trajectory indicators matter for stakeholders?
For investors, commercial planners, and R&D portfolio owners, the following indicators typically drive valuation and planning for vancomycin-like sterile injectables:
| Indicator |
What it signals |
Typical direction for mature products |
| Net price vs list |
Tender and GPO outcomes |
Downward or flat over time |
| Volume (doses) |
Infection burden and hospital use |
Stable to modest growth |
| Contract renewal timing |
Procurement cycle impacts |
Quarter-to-quarter volatility |
| Supply continuity |
Plant uptime and lot release |
Can swing revenue profitability |
| Compliance events |
FDA/EMA warning letters, recalls |
Short-term disruption, longer-term margin pressure |
How does vancomycin’s market compare to newer Gram-positive antibiotics?
Newer agents usually command premium pricing when patents or exclusivity exist. Vancomycin instead behaves like a “base layer” antibiotic:
- Lower pricing but persistent absolute demand
- Less product differentiation
- Higher sensitivity to procurement economics and supply continuity
This means vancomycin’s financial trajectory is more correlated with hospital purchasing and supply logistics than with science breakthroughs.
What does the regulatory and brand history imply for long-term economics?
Vancomycin has longstanding clinical use and broad availability. In mature antibiotics:
- Patent expiry and generic entry compress prices structurally
- Long-term growth relies on volume expansion and manufacturer execution rather than innovation-driven premiumization
That structure tends to stabilize demand but caps margins.
Key commercial scenario outcomes
Base case
- Competitive pressure holds net prices steady-to-down
- Volume stays stable with healthcare utilization
- Profitability depends on manufacturing efficiency and contract position
Upside
- Supply constraints or lot availability issues temporarily strengthen pricing
- Infection burden rises and empiric use increases
- Contracting dynamics favor higher-volume producers
Downside
- Price cuts intensify due to additional generic competition
- Shift toward alternative agents reduces share in specific indications
- Compliance or manufacturing disruptions cause loss of tender participation
What is the practical financial trajectory for vancomycin manufacturers?
For stakeholders owning manufacturing or distribution rights in off-patent vancomycin, the financial trajectory typically looks like:
- Revenue: stable to slowly declining per unit price; total revenue driven by dose throughput and contract win rates
- Gross margin: under sustained pressure; episodic improvement when supply tightness or fewer available competitors align
- Operating margin: sensitive to QC costs, batch failures, and regulatory risk events
This is consistent with sterile injectable generics where the major differentiator is reliability of supply at the contracted cost, not product novelty.
What should investors and strategists monitor next?
- Tender calendar and contract renewals in major hospital systems
- Site-level manufacturing performance (lot release rates and inspection outcomes)
- Net pricing trend versus list and versus primary generic competitors
- Therapeutic guideline updates that shift empiric coverage recommendations
- MRSA epidemiology changes impacting empiric dosing decisions
Key Takeaways
- Vancomycin is a mature, off-patent hospital antibiotic whose market is driven primarily by hospital infection patterns and procurement contracting, not by innovation cycles.
- Net pricing compresses structurally under generic competition; financial performance depends on volume execution and supply continuity.
- Revenue tends to track dosing demand in inpatient settings, while margins hinge on aseptic manufacturing efficiency, QC reliability, and regulatory outcomes.
- Competitive pressure from newer Gram-positive agents typically creates marginal share volatility rather than market elimination.
FAQs
-
Why does vancomycin demand remain resilient despite generic erosion?
Because inpatient protocols retain vancomycin for serious Gram-positive infections, particularly when MRSA risk is present, and usage is concentrated in hospitals with persistent high-acuity care.
-
What has the biggest impact on vancomycin net price in the US?
GPO and hospital tender contracting dynamics, which often set net price below list and drive ongoing erosion among competing suppliers.
-
Do alternative antibiotics materially reduce vancomycin volume?
They can shift share in specific indications or patient profiles, but vancomycin generally maintains baseline demand because it remains in empiric and confirmatory pathways for severe infections.
-
What operational factors most influence profitability for vancomycin manufacturers?
Aseptic manufacturing yield, lot release throughput, QC costs, and inspection outcomes that affect the ability to supply contracted demand.
-
How sensitive is vancomycin to supply disruptions?
High. Sterile injectable shortages can quickly change effective supply and alter short-term pricing and contract allocations.
References
[1] FDA. (n.d.). Vancomycin hydrochloride injection drug safety and labeling resources. U.S. Food and Drug Administration. https://www.fda.gov/
[2] EMA. (n.d.). Vancomycin information and product documentation. European Medicines Agency. https://www.ema.europa.eu/
[3] IDSA. (n.d.). Guidelines on the treatment of MRSA and other Gram-positive infections. Infectious Diseases Society of America. https://www.idsociety.org/