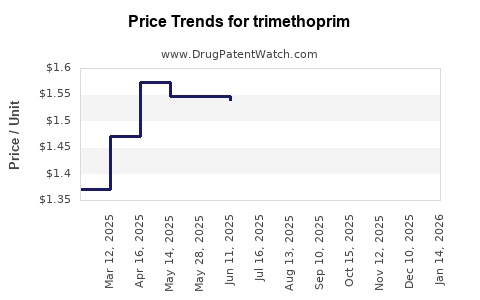
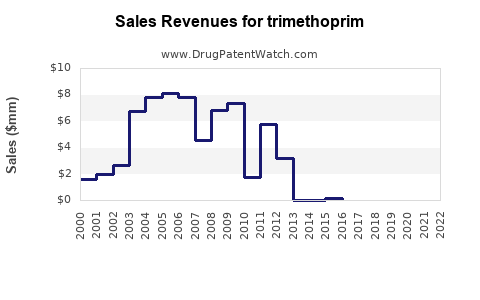
Last updated: April 25, 2026
TRIMETHOPRIM: Market Dynamics and Financial Trajectory
Trimethoprim is an old, off-patent antibacterial used in oral and injectable regimens and most commonly in fixed-dose combinations (notably with sulfamethoxazole). The commercial landscape is shaped by patent expiration, aggressive generic competition, ongoing stewardship and resistance pressure, and steady but uneven demand driven by UTI and respiratory-infection treatment patterns.
This analysis covers supply and demand mechanics, pricing and margin structure, revenue trajectory signals across major geographies, and how portfolio behavior (monotherapy vs combination) affects financial outcomes.
How does the trimethoprim market work in practice?
Demand drivers
- Uncomplicated UTI treatment patterns
- Trimethoprim is used in routine outpatient management of urinary tract infections, either as monotherapy in selected markets or more often as fixed-dose combination with sulfamethoxazole for broader antimicrobial coverage.
- Respiratory and other bacterial indications in combination protocols
- Trimethoprim-based regimens show persistent use in specific clinical pathways where clinician familiarity and local guideline alignment persist.
- Resistance and stewardship constraints
- Growing antimicrobial resistance changes prescriber behavior. This does not remove demand, but it can shift mix away from trimethoprim in regions where susceptibility rates have declined.
- Healthcare access and formularies
- Low-cost antibiotics maintain place on formularies, but adoption rises and falls with local policy updates, clinical guidelines, and antimicrobial-use restrictions.
Supply drivers
- Generic-dominant economics
- Trimethoprim is long past patent exclusivity in core markets. Supply is fragmented across multiple generics and manufacturers, which typically compresses price.
- Combination products concentrate volume
- In many geographies, fixed-dose combinations (trimethoprim/sulfamethoxazole) capture a large share of treated patients. That concentration affects revenue distribution, pricing power, and manufacturing scale.
- Regulatory and quality compliance costs
- Ongoing GMP compliance and inspection outcomes impact cost structure across the generic supply chain, influencing exit/entry behavior and gross margin variability.
What does the financial trajectory look like for trimethoprim over time?
Base-case trajectory (generic antibiotic profile)
Trimethoprim’s financial path follows the typical “patent-out then commoditize” pattern:
- Revenue stability to slow erosion after major competitive entries.
- Price compression as additional ANDA-style entrants or supply expansions hit market pricing.
- Volume sensitivity to guideline shifts and resistance rates.
- Margin volatility driven by input cost cycles (APIs and excipients), contract manufacturing competition, and inspection-related disruptions.
Key inflection points
- Guideline and resistance-driven mix shift
- Even where total antibiotic use stays steady, susceptibility thresholds can reduce trimethoprim’s role, especially in empiric therapy.
- Combination vs monotherapy substitution
- When combinations remain favored for coverage and clinical outcomes, they can stabilize total trimethoprim-related demand even as monotherapy use declines.
- Stewardship restrictions
- Restrictions tend to reduce broad use, but they can also preserve narrow “last-line” utilization that sustains low but persistent revenue.
Revenue behavior you can model
Because trimethoprim is off-patent, the practical financial drivers are:
- Net price decline vs. volume retention
- Market share rotation among generics
- Geographic heterogeneity where local guidance and resistance differ
- Regulatory constraints that periodically tighten access and raise effective barriers
How competitive is the market, and what does that do to pricing?
Competition intensity
- Trimethoprim is widely available as a generic in multiple formats (most notably oral tablets/capsules and as part of combinations).
- The competitive set is large and pricing is typically benchmarked to low-cost generics.
Pricing mechanics
- Low differentiation: efficacy is constrained by bioequivalence requirements rather than innovation-led differentiation.
- Tender and hospital formulary effects: institutional procurement tends to shift volumes to the lowest-cost compliant supplier.
- Price floor behavior: multiple entrants reduce the ability for any single manufacturer to push pricing sustainably unless supply is constrained.
Margin structure
- Gross margin compression is a persistent feature.
- Operating margin depends more on execution (manufacturing yields, compliance cost control, distribution contracts) than on pricing power.
- Product format matters: combinations often stabilize demand because they fit established dosing regimens.
What is the role of combinations in trimethoprim’s financial performance?
Why combinations matter
Fixed-dose trimethoprim/sulfamethoxazole products typically:
- Capture a broader patient base under empiric treatment frameworks.
- Reduce “brand switching” risk for generics because combination dosing is a standard clinical choice.
- Create a revenue stream that is more resilient than monotherapy when resistance patterns remain unfavorable to one agent but acceptable for the combination.
Financial impact
- Volume stabilization: combinations can offset monotherapy declines.
- Pricing ceiling is capped: competition among generics still drives net price down.
- Supply chain scale favors incumbents: high-volume manufacturers can often sustain better unit economics.
How do key geographies shape the revenue outlook?
North America
- Off-patent availability and generic substitution typically create:
- Low net pricing
- Multiple supplier overlap
- Procurement-driven demand volatility
- Revenue tends to track treated-patient demand more than innovation.
Europe
- Formularies and antimicrobial stewardship affect prescribing mix by country.
- Generic intensity remains high; pricing is controlled by tendering and health system reimbursement policies.
Rest of world
- Growth tends to be demand-led (population, healthcare access) rather than value-led.
- Regulatory pathways and local supply stability can drive short-term market swings.
What are the market risks that change the financial trajectory?
1) Resistance and guideline changes
- Resistance reduces empirical suitability, shifting use toward narrower indications.
- That can reduce total treated volume or shift it to alternative antibiotic classes.
2) Safety, tolerability, and clinician switching
- Adverse-event profiles influence clinician and institutional preferences.
- This affects market share versus other low-cost antibiotic classes.
3) Supply disruptions
- Generic manufacturing is sensitive to:
- API supply constraints
- Inspection outcomes
- Batch failures or recalls
- These events can cause temporary pricing spikes but usually reverse when supply normalizes.
4) Regulatory tightening
- Bioequivalence and quality requirements are baseline; enforcement intensity can change effectively available supply.
How does innovation pressure show up despite trimethoprim being off-patent?
Even without new patents on trimethoprim itself, the broader antibacterial market influences financial performance:
- New classes can displace trimethoprim in some indications.
- Stewardship and antibiogram-driven protocols shift empiric pathways away from older agents in some settings.
- As a result, trimethoprim’s financial outcome can deteriorate even while absolute antibiotic use stays stable.
Financial trajectory summary: where revenue goes and why
Expected direction
- Net revenue: stable to declining
- Unit volume: modest swings
- Net price: structurally downward over long horizons
- Operating profitability: constrained by low pricing, offset by scale and cost control
What can improve outcomes
- Channel-level execution:
- secure hospital and pharmacy contracts
- win tender cycles
- maintain consistent supply
- Portfolio mix management:
- focus on combination formats where clinical pathways remain stable
- Cost leadership:
- API sourcing and manufacturing efficiency
What typically hurts outcomes
- Resistance-driven preference shifts
- Loss of formulary position
- Supplier exits that trigger temporary shortages can raise short-term pricing but do not permanently reverse the commoditization trend
Key Takeaways
- Trimethoprim is an off-patent, generic-dominant antibacterial; pricing power is structurally limited by commoditization and procurement dynamics.
- Financial performance is driven primarily by volume retention (patient-treated demand) and mix between monotherapy and fixed-dose combination products.
- Resistance patterns and antimicrobial stewardship shift indications and empiric use, creating uneven demand even when total antibiotic use is steady.
- Revenue typically shows slow erosion or stability with margin volatility driven by generic supply chain cost and compliance execution.
FAQs
-
Is trimethoprim revenue growth likely to come from new approvals?
No. Commercial upside is primarily supply, channel execution, and mix (especially combination products), not innovation-led expansion.
-
Why do fixed-dose combinations matter more than monotherapy for financial stability?
Combination dosing fits established prescribing pathways and tends to preserve treated-patient volume even when monotherapy use declines.
-
What is the biggest driver of price changes in trimethoprim?
Generic competition plus procurement and tender pricing typically determine net price more than independent manufacturer strategy.
-
How do antimicrobial resistance trends affect financial trajectory?
Resistance can reduce empirical appropriateness and shift patient flow to alternative antibiotics, lowering volumes or changing mix toward narrower use.
-
What determines profitability for generic trimethoprim suppliers?
Unit economics come from manufacturing yield and cost control, compliance execution, and contract position in hospital and retail channels.
References
[1] World Health Organization. (2023). Antibacterial agents in clinical use: resistance data and stewardship considerations. WHO. https://www.who.int/health-topics/antimicrobial-resistance
[2] FDA. (n.d.). Drug shortages: trimethoprim-containing products and supply issues (database). U.S. Food and Drug Administration. https://www.fda.gov/drugs/drug-shortages
[3] European Medicines Agency. (n.d.). Guidelines on the quality and bioequivalence of generic medicinal products. EMA. https://www.ema.europa.eu/en/human-regulatory/marketing-authorisation/generics
[4] CDC. (2024). Antibiotic resistance threats in the United States. Centers for Disease Control and Prevention. https://www.cdc.gov/drugresistance/threat-report/