Metronidazole - Generic Drug Details
✉ Email this page to a colleague
What are the generic sources for metronidazole and what is the scope of patent protection?
Metronidazole
is the generic ingredient in twenty branded drugs marketed by Pfizer, Able, Alembic, Chartwell Rx, Galderma Labs Lp, Aurobindo Pharma Ltd, Cosette, Fougera Pharms, Zydus Lifesciences, Bausch, Encube, Taro, Glenmark Pharms Ltd, Padagis Israel, Sciegen Pharms, Solaris Pharma Corp, Chemo Research Sl, Teva Pharms, Baxter Hlthcare, B Braun, Abbott, Abraxis Pharm, Hikma, Intl Medication, Watson Labs, Amneal, Baxter Hlthcare Corp, Gland, Hospira, Inforlife, Rising, Saptalis Pharms, Labs Af, Alembic Pharms Ltd, Cadila, Cadila Pharms Ltd, Chartwell Molecules, Flamingo Pharms, Fosun Pharma, Halsey, Innogenix, Ivax Sub Teva Pharms, LNK, Lupin Ltd, Mutual Pharm, Novitium Pharma, Pliva, Somerset Theraps Llc, Strides Pharma, Superpharm, Teva Pharms Usa, Unichem, Watson Labs Inc, Ortho Mcneil Pharm, and Savage Labs, and is included in ninety-two NDAs. There are nine patents protecting this compound and two Paragraph IV challenges. Additional information is available in the individual branded drug profile pages.Metronidazole has twenty-two patent family members in nine countries.
There are eighteen drug master file entries for metronidazole. Sixty-eight suppliers are listed for this compound.
Summary for metronidazole
| International Patents: | 22 |
| US Patents: | 9 |
| Tradenames: | 20 |
| Applicants: | 55 |
| NDAs: | 92 |
| Drug Master File Entries: | 18 |
| Finished Product Suppliers / Packagers: | 68 |
| Raw Ingredient (Bulk) Api Vendors: | 119 |
| Clinical Trials: | 589 |
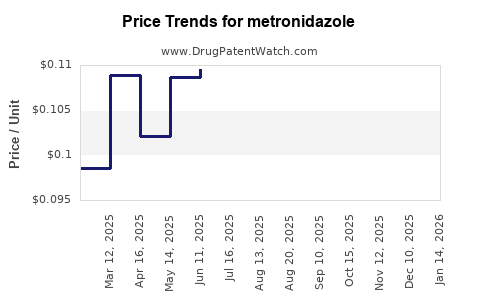
| Drug Prices: | Drug price trends for metronidazole |
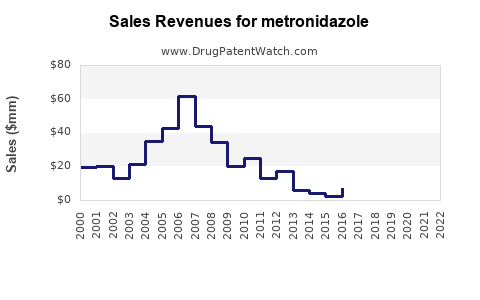
| Drug Sales Revenues: | Drug sales revenues for metronidazole |
| Patent Litigation and PTAB cases: | See patent lawsuits and PTAB cases for metronidazole |
| What excipients (inactive ingredients) are in metronidazole? | metronidazole excipients list |
| DailyMed Link: | metronidazole at DailyMed |
Recent Clinical Trials for metronidazole
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| Universitt Luzern | PHASE4 |
| Faiz ur rahman | NA |
| Medical College of Wisconsin | EARLY_PHASE1 |
Pharmacology for metronidazole
| Drug Class | Nitroimidazole Antimicrobial |
Medical Subject Heading (MeSH) Categories for metronidazole
Anatomical Therapeutic Chemical (ATC) Classes for metronidazole
Paragraph IV (Patent) Challenges for METRONIDAZOLE
| Tradename | Dosage | Ingredient | Strength | NDA | ANDAs Submitted | Submissiondate |
|---|---|---|---|---|---|---|
| LIKMEZ | Oral Suspension | metronidazole | 500 mg/5 mL | 216755 | 1 | 2025-11-13 |
| NUVESSA | Vaginal Gel | metronidazole | 1.30% | 205223 | 1 | 2022-03-30 |
| METROGEL | Topical Gel | metronidazole | 1% | 021789 | 1 | 2008-10-21 |
| METROGEL-VAGINAL | Vaginal Gel | metronidazole | 0.75% | 020208 | 1 | 2004-09-02 |
US Patents and Regulatory Information for metronidazole
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Abbott | METRONIDAZOLE | metronidazole | INJECTABLE;INJECTION | 018889-001 | Nov 18, 1983 | DISCN | No | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | ||||
| Pfizer | FLAGYL | metronidazole | TABLET;ORAL | 012623-001 | Approved Prior to Jan 1, 1982 | DISCN | Yes | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | ||||
| Galderma Labs Lp | METROGEL | metronidazole | GEL;TOPICAL | 021789-001 | Jun 30, 2005 | AB | RX | Yes | Yes | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | |||
| Novitium Pharma | METRONIDAZOLE | metronidazole | TABLET;ORAL | 208162-001 | May 25, 2016 | AB | RX | No | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | |||
| Zydus Lifesciences | METRONIDAZOLE | metronidazole | GEL;TOPICAL | 220095-001 | Feb 13, 2026 | AB | RX | No | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | |||
| Strides Pharma | METRONIDAZOLE | metronidazole | TABLET;ORAL | 070040-001 | Jan 29, 1985 | DISCN | No | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | ||||
| Bausch | METROGEL-VAGINAL | metronidazole | GEL;VAGINAL | 020208-001 | Aug 17, 1992 | DISCN | Yes | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | ||||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for metronidazole
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Teva Pharms | VANDAZOLE | metronidazole | GEL;VAGINAL | 021806-001 | May 20, 2005 | ⤷ Start Trial | ⤷ Start Trial |
| Galderma Labs Lp | METROGEL | metronidazole | GEL;TOPICAL | 021789-001 | Jun 30, 2005 | ⤷ Start Trial | ⤷ Start Trial |
| Galderma Labs Lp | METROGEL | metronidazole | GEL;TOPICAL | 019737-001 | Nov 22, 1988 | ⤷ Start Trial | ⤷ Start Trial |
| Bausch | METROGEL-VAGINAL | metronidazole | GEL;VAGINAL | 020208-001 | Aug 17, 1992 | ⤷ Start Trial | ⤷ Start Trial |
| Bausch | METROGEL-VAGINAL | metronidazole | GEL;VAGINAL | 020208-001 | Aug 17, 1992 | ⤷ Start Trial | ⤷ Start Trial |
| Bausch | METROGEL-VAGINAL | metronidazole | GEL;VAGINAL | 020208-001 | Aug 17, 1992 | ⤷ Start Trial | ⤷ Start Trial |
| Pfizer | FLAGYL ER | metronidazole | TABLET, EXTENDED RELEASE;ORAL | 020868-001 | Nov 26, 1997 | ⤷ Start Trial | ⤷ Start Trial |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
International Patents for metronidazole
| Country | Patent Number | Title | Estimated Expiration |
|---|---|---|---|
| China | 107823123 | 高剂量粘膜粘附甲硝唑水基凝胶制剂及其治疗细菌性阴道病的用途 (High dosage mucoadhesive metronidazole aqueous-based gel formulations and use to treat bacterial vaginosis) | ⤷ Start Trial |
| World Intellectual Property Organization (WIPO) | 2013003646 | ⤷ Start Trial | |
| World Intellectual Property Organization (WIPO) | 2019140516 | ⤷ Start Trial | |
| Mexico | 2014000066 | FORMULACIONES MUCOADHESIVAS EN GEL DE ALTA DOSIFICACIÓN DE METRONIDAZOL DE BASE ACUOSA Y SU USO PARA TRATAR LA VAGINOSIS BACTERIANA. (HIGH DOSAGE MUCOADHESIVE METRONIDAZOLE AQUEOUS-BASED GEL FORMULATIONS THEIR USE TO TREAT BACTERIAL VAGINOSIS.) | ⤷ Start Trial |
| Japan | 5986632 | ⤷ Start Trial | |
| China | 107823123 | ⤷ Start Trial | |
| Japan | 2017014248 | 高用量粘膜接着性メトロニダゾール水系ゲル製剤および細菌性膣炎を治療するためのそれらの使用 (HIGH-DOSE MUCOADHESIVE METRONIDAZOLE AQUEOUS GEL PREPARATION, AND USE OF THE SAME FOR TREATING BACTERIAL VAGINOSIS) | ⤷ Start Trial |
| >Country | >Patent Number | >Title | >Estimated Expiration |
Supplementary Protection Certificates for metronidazole
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 0328535 | 96C0021 | Belgium | ⤷ Start Trial | PRODUCT NAME: LANSOPRAZOLE + CLARITHROMYCINE + METRONIDAZOLE; REGISTRATION NO/DATE IN FRANCE: K 27 17033R DU 19960209; REGISTRATION NO/DATE AT EEC: K 27 17033R DU 19960209 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Make Better Decisions: Try a trial or see plans & pricing
Drugs may be covered by multiple patents or regulatory protections. All trademarks and applicant names are the property of their respective owners or licensors. Although great care is taken in the proper and correct provision of this service, thinkBiotech LLC does not accept any responsibility for possible consequences of errors or omissions in the provided data. The data presented herein is for information purposes only. There is no warranty that the data contained herein is error free. We do not provide individual investment advice. This service is not registered with any financial regulatory agency. The information we publish is educational only and based on our opinions plus our models. By using DrugPatentWatch you acknowledge that we do not provide personalized recommendations or advice. thinkBiotech performs no independent verification of facts as provided by public sources nor are attempts made to provide legal or investing advice. Any reliance on data provided herein is done solely at the discretion of the user. Users of this service are advised to seek professional advice and independent confirmation before considering acting on any of the provided information. thinkBiotech LLC reserves the right to amend, extend or withdraw any part or all of the offered service without notice.